Abstract
As transcatheter aortic valve implantation (TAVI) is increasingly used in younger and lower-risk patients, it is important to understand the risk of valve thrombosis in TAVI prostheses and how this may contribute to bioprosthetic valve dysfunction and thromboembolic events. Subclassification of valve thrombosis by incidence or timing is challenged by differences in sensitivity and heterogeneity among the imaging modalities used for its detection. The long-term implications of valve thrombosis – particularly regarding stroke risk and prosthesis longevity – remain uncertain and debated. Current treatment options for valve thrombosis include temporary anticoagulation therapy, thrombolysis, and, in some cases, reintervention. To address these concerns, ongoing research is focused on improving the detection and treatment of valve thrombosis through standardised imaging protocols and the use of intensified antithrombotic regimens that balance the thrombotic and bleeding risks. Prevention of valve thrombosis remains a challenge requiring a better understanding of patient- and procedure-related risk factors. These insights will be essential to individualise antithrombotic therapy and improve prosthesis design. This review outlines current evidence on the mechanisms, prevalence, clinical relevance, and management of TAVI thrombosis and highlights ongoing clinical trials investigating this condition.
Transcatheter aortic valve implantation (TAVI) is a minimally invasive procedure for the treatment of severe aortic stenosis. Although the safety of the procedure continues to improve, the risk of thromboembolic complications remains high. Periprocedural complications include ischaemic stroke, transient ischaemic attack, acute myocardial infarction, and systemic thromboembolism, which are associated with increased morbidity and mortality1. Periprocedural emboli may consist of aortic valve tissue, aortic wall tissue, thrombus, and even catheter particles2. In later phases, (new-onset) atrial fibrillation (AF) is a more prevalent cause of thromboembolic events.
Valve leaflet thrombosis may also pose a risk of thromboembolic events and possibly lead to long-term structural valve deterioration34. The mechanisms behind the formation of valve thrombosis are not fully elucidated56. Valve thrombosis associated with obvious clinical sequelae is rare, with incidences similar to those for surgical biological aortic valve prostheses78910. Subclinical leaflet thrombosis, however, is common, with reported incidences varying by up to 40% with different imaging modalities, antithrombotic use, and timing of follow-up11. Still, the phenomenon is likely underdiagnosed as the routine follow-up is usually with transthoracic echocardiography (TTE)12. TAVI thrombosis, detected by cardiac computed tomography (CT), is known as hypoattenuated leaflet thickening (HALT). It may occur with or without reduced leaflet motion (RLM); when coexisting, it is described as hypoattenuation affecting motion (HAM). Different predictors of HALT have been identified, but the clinical impact, as well as the antithrombotic treatment, of HALT remains debated.
This review summarises the available evidence on the mechanisms, prevalence, clinical significance, and treatment of TAVI thrombosis and provides an oversight of upcoming clinical trials studying this phenomenon.
Definitions
Bioprosthetic valve dysfunction and failure
As the use of TAVI is rapidly increasing, now expanding to lower-risk and younger patients, durability of the biological prosthesis is of utmost importance. Bioprosthetic valve dysfunction can be divided into 4 aetiological groups comprising structural valve deterioration, non-structural valve deterioration, thrombosis, and endocarditis1314. While thrombosis is potentially a reversible cause, when it persists it can result in long-lasting or permanent bioprosthetic valve dysfunction. Bioprosthetic valve failure is “a patient-oriented clinical endpoint”, unlike bioprosthetic valve dysfunction, which is without clinical impact1314. Bioprosthetic valve failure was defined by the Valve Academic Research Consortium 3 (VARC-3) Committee as (i) any bioprosthetic valve dysfunction associated with clinically expressive criteria or irreversible haemodynamic valve deterioration, (ii) aortic valve reoperation or reintervention, or (iii) valve-related death14.
Valve thrombosis
As stated, different terms have been introduced to describe the following CT phenomena: HALT is referred to as HAM when RLM is also present1415. HALT can be visualised on cardiac CT as a non-contrast-enhanced thickening on the aortic side of the leaflet surface. HALT and RLM can be graded according to the proportion of thickening and reduced motion from the base to the edge of the leaflet, respectively (Table 1, Figure 1). Subclassifications of valve thrombosis related to TAVI can be based on the anatomical location, including (1) (sub)clinical leaflet thrombosis (HALT, which can result in reduced leaflet mobility [RLM]), (2) neosinus thrombosis, (3) subvalvular thrombosis, and (4) stent frame thrombosis16.
Whereas subclinical leaflet thrombosis can only be diagnosed with cardiac CT, clinically significant valve thrombosis is per definition diagnosed clinically, associated with worsened transprosthetic haemodynamics with TTE, and confirmed later by cardiac CT or transoesophageal echocardiography (TOE). Please refer to Table 1 for the VARC-3 definition of HALT/HAM and clinically significant valve thrombosis14.
Table 1. Characteristics of TAVI thrombosis including HALT/HAM and clinically significant valve thrombosis#.
| TAVI thrombosis | |
|---|---|
| Presentation | Not clinically significant: |
| No or mild haemodynamic changes and no symptoms or sequelae compatible with valve thrombosis or thromboembolism | |
| Clinically significant: | |
| Symptoms indicating worsening of valve stenosis or regurgitation, such as worsening dyspnoea, syncope, etc. and/or sequelae of a thromboembolic event such as ischaemic stroke, systemic thromboembolism, etc. | |
| Timing | Time from TAVI |
| Acute: 0 to ≤24 hours | |
| Subacute: >24 hours to ≤30 days | |
| Late: >30 days to ≤1 year | |
| Very late: >1 year | |
| Diagnosis | HALT/HAM: |
| Hypoattenuated leaflet thickening and typical meniscal configuration of one or more leaflets visually identified by computed tomography (2D multiplanar reconstructions or 3D volume-rendering), with or without RLM | |
| Clinically significant valve thrombosis: | |
| Definite: histopathological confirmation | |
| Probable: haemodynamic changes and imaging findings compatible with valve thrombosis, with resolution of haemodynamic changes and of imaging findings following anticoagulation therapy | |
| Possible: imaging compatible with leaflet thrombosis formation but either haemodynamic changes or imaging findings persist following anticoagulation therapy or anticoagulation therapy is not (yet) administered | |
| Characterisation | HALT/HAM: |
| The extent of HALT can be described as follows: | |
| ≤25% (limited to the base) | |
| >25% and ≤50% | |
| >50% and ≤75% | |
| >75% | |
| The extent of RLM is described as follows: | |
| No RLM | |
| <50% RLM | |
| ≥50% RLM | |
| Immobile leaflet | |
| Clinically significant valve thrombosis: | |
| In case of symptoms indicating worsening valve stenosis or regurgitation, severe haemodynamic valve deterioration* and confirmatory imaging must be present | |
| In case of sequelae of a thromboembolic event, moderate** or severe haemodynamic valve deterioration or confirmatory imaging must be present | |
| Response to anticoagulant therapy (≥3 months) | Resolved: partial or complete resolution of symptoms, imaging findings, and haemodynamic valve deterioration |
| Persistent: no improvement in symptoms, imaging findings, or haemodynamic valve deterioration | |
| Recurrent: recurrence of symptoms, imaging findings, or haemodynamic valve deterioration | |
| #Adapted from the VARC-3: updated endpoint definitions for aortic valve clinical research (modified according to14). Note: clinically significant valve thrombosis and HALT/HAM are not separate entities: clinically significant valve thrombosis is a clinical manifestation of HALT/HAM. *Severe haemodynamic valve deterioration is defined as an “increase in mean transvalvular gradient ≥20 mmHg resulting in mean gradient ≥30 mmHg with concomitant decrease in EOA ≥0.6 cm2 or ≥50% and/or decrease in Doppler velocity index ≥0.2 or ≥40% compared with echocardiographic assessment performed 1-3 months post-procedure, or new occurrence, or increase of ≥2 grades of intraprosthetic AR resulting in severe AR”14. **Moderate haemodynamic valve deterioration is defined as an “increase in mean transvalvular gradient ≥10 mmHg resulting in mean gradient ≥20 mmHg with concomitant decrease in EOA ≥0.3 cm2 or ≥25% and/or decrease in Doppler velocity index ≥0.1 or ≥20% compared with echocardiographic assessment performed 1-3 months post-procedure, or new occurrence or increase of ≥1 grade of intraprosthetic AR resulting in ≥moderate AR”14. 2D: two-dimensional; 3D: three-dimensional; AR: aortic regurgitation; EOA: effective orifice area; HALT: hypoattenuated leaflet thickening; HAM: hypoattenuation affecting motion; RLM: reduced leaflet motion; TAVI: transcatheter aortic valve implantation; VARC: Valve Academic Research Consortium | |
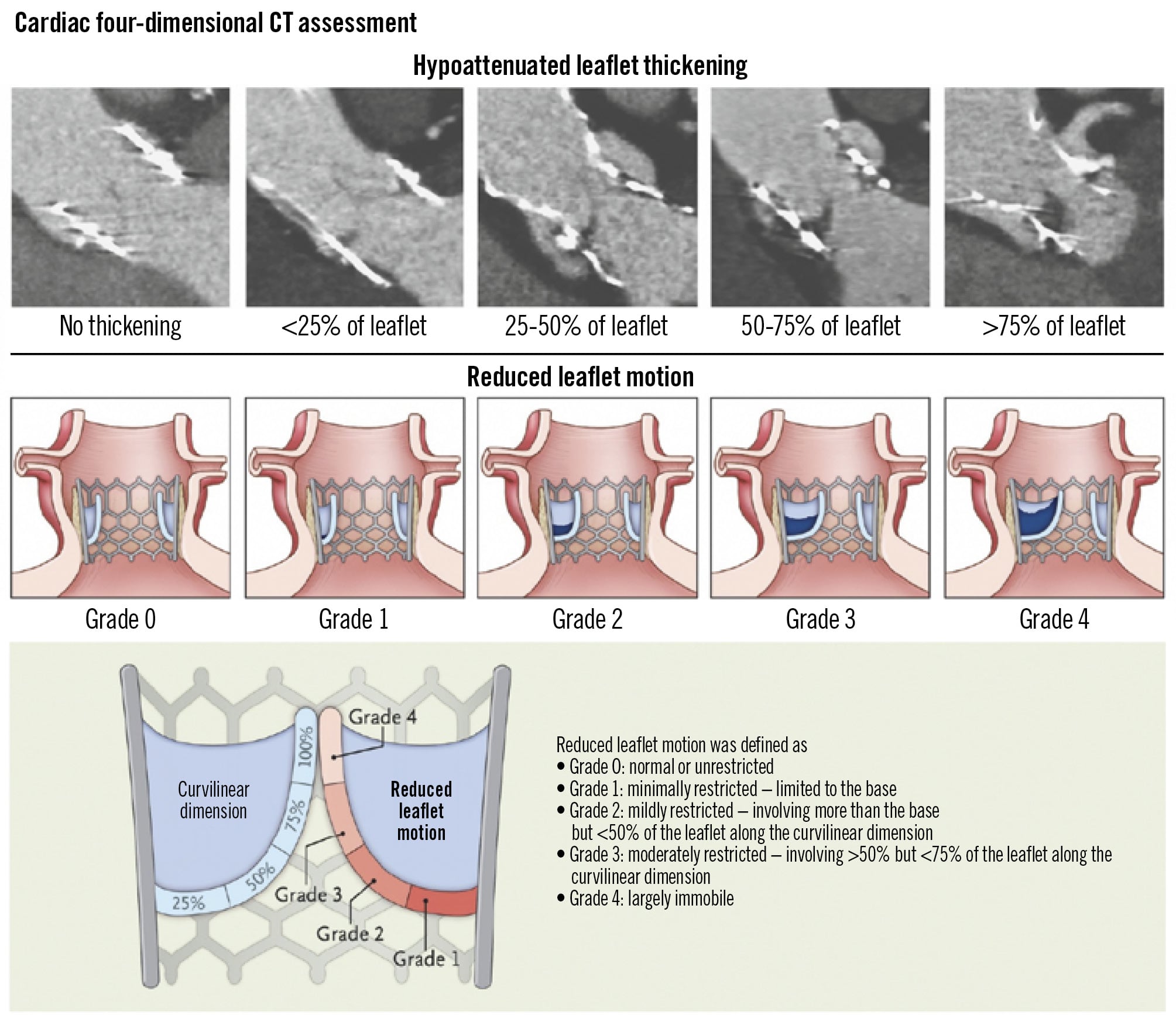
Figure 1. Assessment of HALT and RLM. Adapted with permission from15. The figure shows the different degrees of hypoattenuated leaflet thickening and reduced leaflet motion used in classifying the findings on four-dimensional CT and provides definitions of the different degrees of reduced leaflet motion. The term HALT specifically refers to thrombus formation on the leaflets and does not encompass other processes such as fibrosis or endocarditis. The terms subclinical leaflet thrombosis, subclinical valve thrombosis, and subclinical leaflet thickening have been used as descriptive synonyms of HALT; we will use the term subclinical leaflet thrombosis throughout. CT: computed tomography; HALT: hypoattenuated leaflet thickening; RLM: reduced leaflet motion
Incidences
Subclinical leaflet thrombosis is a common finding in patients undergoing TAVI, with a higher prevalence compared to patients undergoing surgical bioprosthetic aortic valve replacement11. The incidence of subclinical leaflet thrombosis in TAVI patients varies greatly according to the study setting, patient population, valve type, imaging modality, diagnostic criteria, antithrombotic regimen, and follow-up duration (Table 2). Reported rates range from 12% to 18% at 3 months and 25% to 30% at 1 year post-procedure61015171819. Subclinical leaflet thrombosis is dynamic in nature and can progress, persist, or regress56. In low surgical risk TAVI patients with subclinical leaflet thrombosis at 30 days (n=20), 50% of the patients had spontaneous resolution at 1 year without anticoagulant therapy10. Anticoagulation has demonstrated efficacy in both preventing and resolving subclinical leaflet thrombosis1020. In patients without an established indication for oral anticoagulation (OAC), 12.4% of patients randomised to an anticoagulant regimen and 32.4% of patients randomised to an antiplatelet regimen developed subclinical leaflet thrombosis15. In a registry of nearly 900 patients with TAVI or surgical aortic valve replacement, subclinical leaflet thrombosis resolved in 100% of patients treated with anticoagulants and persisted in 91% of patients without anticoagulants6.
Clinically significant valve thrombosis is rare with incidences ranging from <1% to 2.8%78910. While patients with significant valve thrombosis may present with symptoms of acute heart failure or thromboembolism, the majority report dyspnoea (65%) or no worsening in symptoms (31%) when diagnosed within a median time of 6 months from TAVI89.
Table 2. Subclinical leaflet thrombosis diagnosed by CT and clinical events in recent TAVI studies and trials.
| Design and population | Subclinical leaflet thrombosis | Clinical events | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Study | Year | Study design | Number of patients | Time to diagnosis | Incidence | Association with clinical events | Time to follow-up | Death | Stroke/TIA | Bleeding | Structural valve deterioration |
| Danish registry19 | 2016 | Registry | 460 consecutive TAVI patients | 1 to 3 months | 7% | None | 1 month | NA | 1%/NA | 4% | NA |
| 1 year | 17% | 4%/NA | NA | NA | |||||||
| RESOLVE and SAVORY registries6 | 2017 | Registry | 890 patients with TAVI or SAVR | 83 days (IQR 33-281) | 13% in TAVI 4% in SAVR | HR for stroke/TIA 3.27 (95% CI: 1.62-6.59) and for TIA 7.02 (95% CI: 2.35-20.91) | 540±413 days | 4% | 3%/1% | NA | 2% |
| SAVORY and Japanese registries17 | 2017 | Registry | 75 TAVI patients | 97 days (IQR 24-331) | 17% | None | NA | NA | NA/NA | NA | NA |
| OCEAN-TAVI registry26 | 2019 | Registry | 485 TAVI patients | Median 3 days | 9.3% | None | In-hospital | NA | 1%/NA | 4% | NA |
| 6 months | 7.1% | 2 years | 6.7% and 0% in patients with and without early SLT | 2.3% and 0% in patients with and without early SLT/NA | NA | NA | |||||
| 1 year | 11.3% | ||||||||||
| 2 years | 12.7% | ||||||||||
| 3 years | 16.9% | ||||||||||
| GALILEO CT substudy15 | 2019 | RCT | 231 TAVI patients | 90±15 days | 22.6% | None | Day 90 | 2% | 3%/NA | 2% | NA |
| PARTNER 3 cardiac CT substudy10 | 2020 | RCT | 435 low surgical risk patients with TAVI or SAVR | 30 days | 10% | None | Day 7 to 365 | 1% | <1%/1% | NA | NA |
| 365 days | 24% | ||||||||||
| Evolut Low Risk CT substudy18 | 2020 | RCT | 179 TAVI patients at low surgical risk | 30 days | 17.3% | None | Day 7 to 365 | 0% | 1%/1% | NA | NA |
| 365 days | 30.9% | ||||||||||
| German registry4 | 2022 | Registry | 804 TAVI patients | 5 days (IQR 4-6) | 16% | HR for symptomatic haemodynamic valve deterioration 6.10 (95% CI: 2.59-14.29) | 3.3 years (IQR 2.2-5.0) | 26.5%* | 4%/<1% | NA | 3% |
| Minneapolis registry21 | 2022 | Registry | 565 TAVI patients | 31 days (IQR 29-36) | 19% | HR for death 2.98 (95% CI: 1.57-5.63) and cardiac death 4.58 (95% CI: 1.81-11.6) | 1 year | 7% | 4% | 10% | NA |
| Minneapolis registry22 | 2022 | Registry | 856 TAVI patients | 30 days | 12.3% | HR for death 1.83 (95% CI: 1.13-2.97) | 2.2 years (IQR 1.5-3.2) | 21% | 5%/1% | 15.5% | NA |
| LRT trial 4-year outcomes24 | 2023 | RCT | 200 TAVI patients | 30 days | 14% | None | 4 years | 11.9% | 7.5% | 5.6% | 5.8% |
| EFFORT study27 | 2024 | Registry | 101 TAVI patients | 105 days (IQR 98-129) | 13% | NA | NA | NA | NA/NA | NA | NA |
| *1−Kaplan-Meier estimate for survival. CI: confidence interval; CT: computed tomography; HR: hazard ratio; IQR: interquartile range; NA: not available; RCT: randomised controlled trial; SAVR: surgical aortic valve replacement; SLT: subclinical leaflet thrombosis; TAVI: transcatheter aortic valve implantation; TIA: transient ischaemic attack | |||||||||||
Outcomes
The clinical significance of subclinical leaflet thrombosis is largely unknown. There is discordant evidence as to whether or not subclinical leaflet thrombosis is associated with increased mortality, and only a single study has found subclinical leaflet thrombosis to be associated with transient ischaemic attacks (Table 2)46212223. There is some evidence of an association between subclinical leaflet thrombosis and structural valve deterioration46. The long-term risk of adverse events in patients with subclinical leaflet thrombosis was evaluated prospectively in a single-centre study of consecutive non-anticoagulated TAVI patients, half of whom underwent CT scans within a median of 5 days post-implantation (n=804)4. With comparable baseline characteristics, subclinical leaflet thrombosis was not associated with higher 3-year mortality or cerebrovascular events. However, bioprosthetic valve failure at 3 years was higher in patients with early subclinical leaflet thrombosis (8.7% vs 2.0%). Multivariate analysis confirmed a significantly higher rate of bioprosthetic valve failure associated with subclinical leaflet thrombosis (hazard ratio [HR] 6.10, 95% confidence interval [CI]: 2.59-14.29). In low-risk TAVI patients, subclinical leaflet thrombosis at 30 days was not associated with structural valve deterioration after 4 years24.
Imaging modalities
Four-dimensional CT (4D-CT) has become the gold standard for assessing leaflet morphology and mobility. Contrast-enhanced, electrocardiogram (ECG)-gated cardiac CT covering the full cardiac cycle, with retrospective gating and ultrathin (<1 mm) slices, provides high temporal and spatial resolution to display leaflet motion. TOE can also detect subclinical leaflet thrombosis and is particularly useful when 4D-CT is not immediately available. In general, echocardiography (TOE with higher sensitivity) complements CT by evaluating the haemodynamic effects on the valve. This makes it possible to identify the red flags mentioned in the Central illustration. Key echocardiographic measurements include continuous-wave Doppler peak velocity and mean gradient, Doppler velocity index, and effective orifice area. The orifice area is important to determine patient-prosthesis mismatch as a differential diagnosis of gradient alterations. In addition, colour flow Doppler is essential to reveal prosthetic and periprosthetic regurgitation. Cardiac magnetic resonance imaging (MRI) can potentially display leaflet alterations but is more susceptible to metallic artefacts caused by the TAVI prosthesis. Thus, cardiac MRI has played a subordinate role in the referenced studies (Table 2). Positron emission tomography (PET) using 18F-sodium fluoride (18F-NaF) can image bioprosthetic microcalcification activity25 and is currently being investigated to differentiate the role of subclinical leaflet thrombosis on valve degeneration in a clinical trial (POPular PET TAVI; ClinicalTrials.gov: NCT05758662).
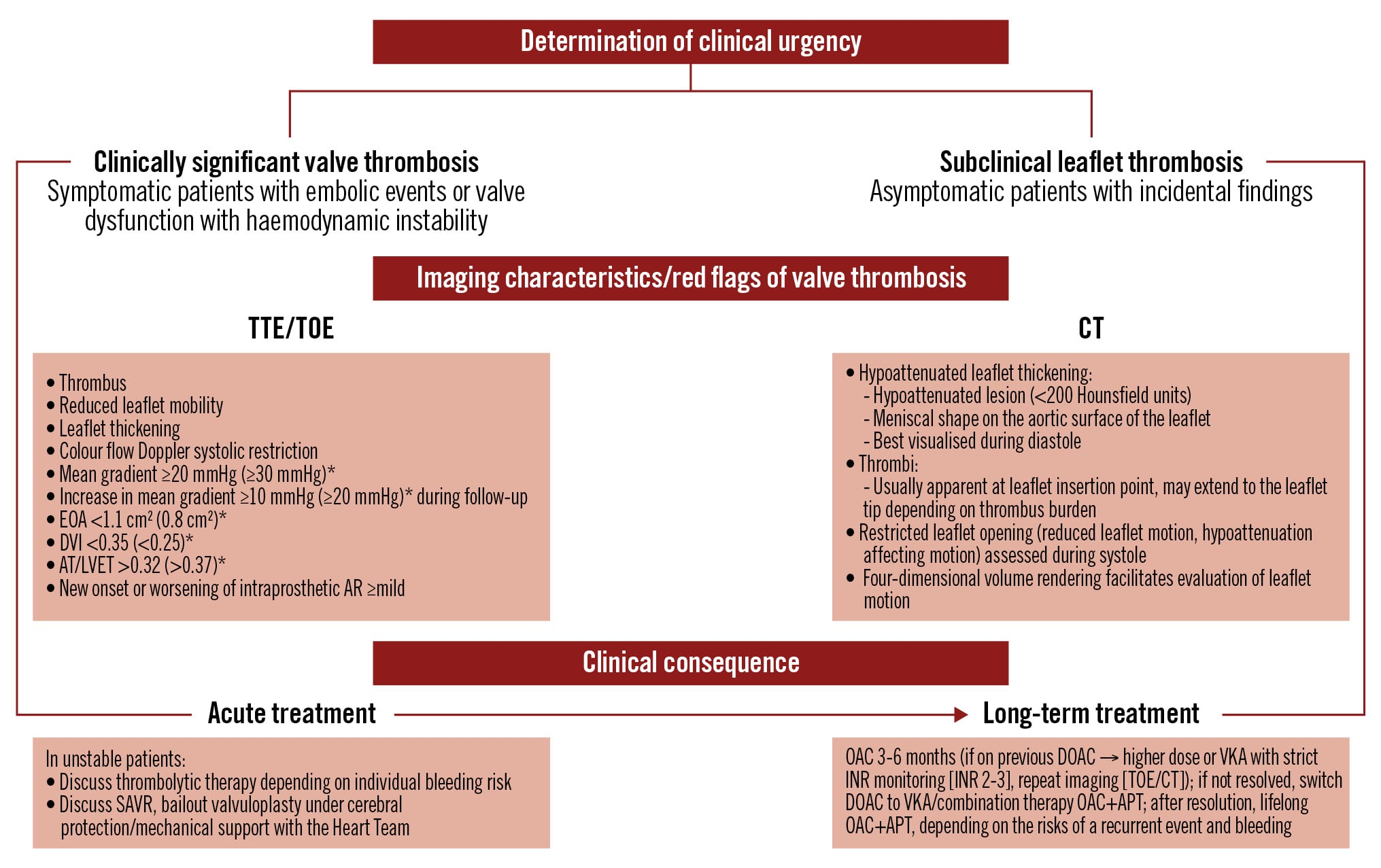
Central illustration. Diagnosis and management of TAVI thrombosis. Adapted with permission from1462. *Red flags with higher level of suspicion of TAVI thrombosis. APT: antiplatelet therapy; AR: aortic regurgitation; AT: acceleration time; CT: computed tomography; DOAC: direct oral anticoagulant; DVI: Doppler velocity index; EOA: effective orifice area; INR: international normalised ratio; LVET: left ventricular ejection time; OAC: oral anticoagulant; SAVR: surgical aortic valve replacement; TAVI: transcatheter aortic valve implantation; TOE: transoesophageal echocardiography; TTE: transthoracic echocardiography; VKA: vitamin K antagonist
Mechanisms
The mechanisms of thrombus formation on implanted valves are related to clinical and procedural risk factors, biomarkers and antithrombotic treatment and are dependent on the time elapsed since implantation2627. Valve thrombosis is likely due to complex interactions between plasma proteins, blood cells, and valve tissue or damaged valve material, though the exact mechanisms remain unclear28.
Whether these mechanisms are pathological or a natural adaptation to achieve valve endothelisation remains uncertain29. A modified Virchow’s triad explains valve thrombosis in TAVI patients, highlighting three factors: stasis of blood flow, hypercoagulability, and endothelial injury (Figure 2)30. Other predictors of subclinical leaflet thrombosis identified in observational studies include male sex, high body mass index, absence of anticoagulants, chronic renal disease, prosthesis underexpansion and deformation, and severe patient-prosthesis mismatch9101721232631.
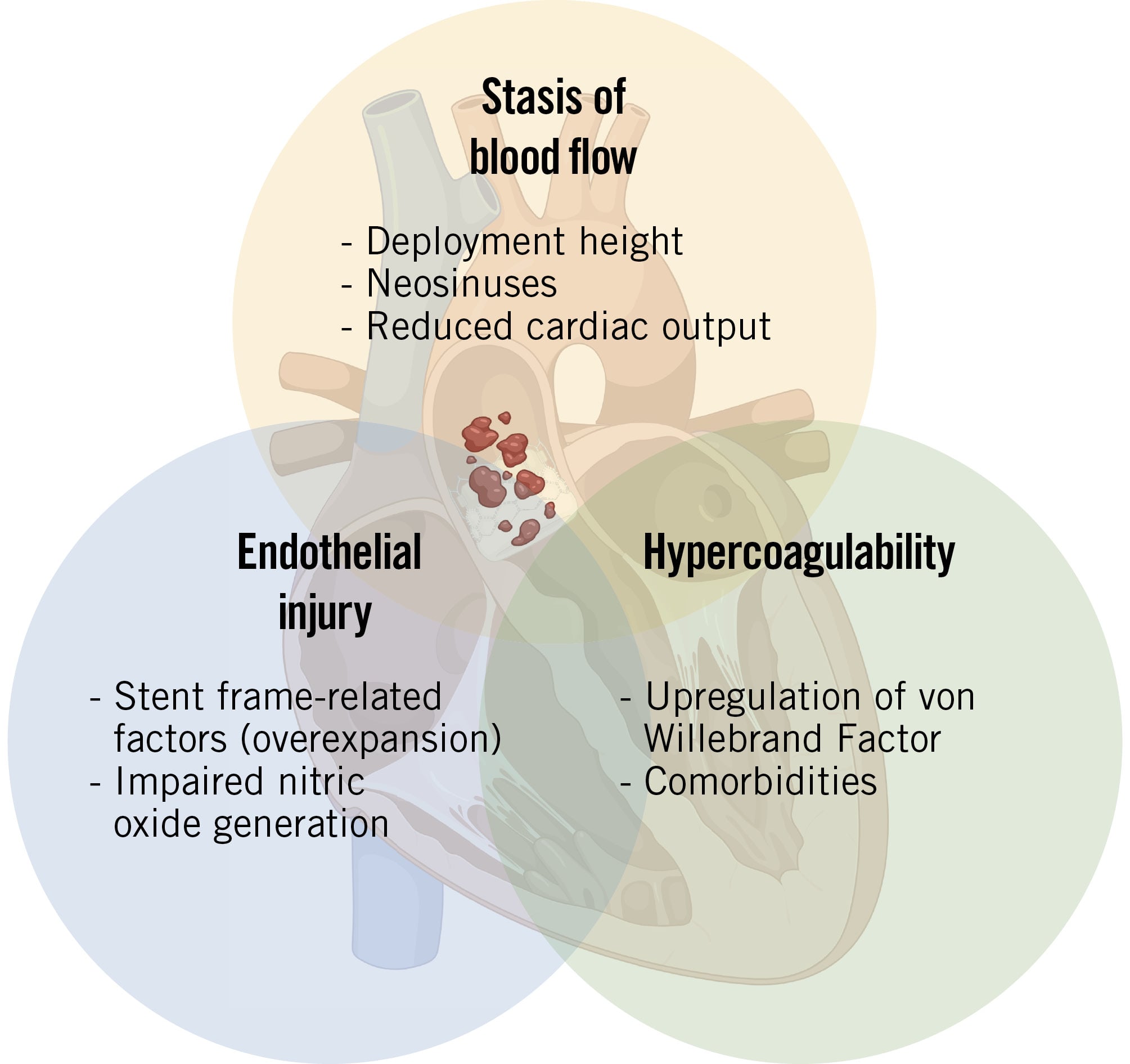
Figure 2. Virchow’s triad in TAVI patients. A modified Virchow’s triad explaining the mechanisms of valve thrombosis formation in patients after TAVI. Three factors are highlighted: stasis of blood flow, hypercoagulability, and endothelial injury. TAVI: transcatheter aortic valve implantation
Hypercoagulable state
In a prospective study of 565 TAVI patients, independent predictors of 30-day stroke and transient ischaemic attacks were major and life-threatening bleeding events, as well as reduced concentrations of high-molecular-weight von Willebrand Factor (VWF) multimers32. The glycoprotein VWF, present in the subendothelium, plasma, platelets, and endothelial cells, plays a key role in platelet adhesion, aggregation, and collagen binding. High-molecular-weight VWF multimers are particularly important for haemostasis under shear stress, such as in severe aortic stenosis33. Acquired VWF abnormalities affect 42% to 92% of patients with severe aortic stenosis, with higher mean transvalvular gradients associated with lower levels of high-molecular-weight VWF multimers, potentially increasing bleeding risk3435. Surgical and transcatheter aortic valve replacement correct these VWF abnormalities3536, including Heyde’s syndrome, a rare condition characterised by angiodysplasia and VWF deficiency, which causes severe gastrointestinal bleeding37. Research by Yamashita et al showed that high-molecular-weight VWF multimers normalise rapidly after surgical valve replacement3839. In fact, these patients may be exposed to a higher thromboembolic risk in the early phase due to a “VWF-predominant state”, in which the balance shifts in favour of VWF over its regulating enzyme ADAMTS13. Although less extensively studied, conditions causing thrombophilia (e.g., Factor V Leiden) may be associated with increased thrombus formation. A recent study of 52 TAVI patients found that 8 patients had thrombophilia (mostly Factor V Leiden). These patients had a numerically higher incidence of subclinical leaflet thrombosis and a significantly greater thrombus volume40.
Stasis of blood flow
Thrombosis forms in the neosinuses due to turbulence and flow stagnation at the base of the leaflets. In an in vitro study utilising a left heart simulator model, Midha et al found a 7-fold increase in stagnant flow area (velocity <0.1 m/s) with low valve deployment and a 4-fold increase with low cardiac output41. The same study also included 72 anticoagulant-naïve patients and reported that deeper valve deployment increased the neosinus volume (Figure 3), which correlated with a greater thrombus volume and lower deployment height in self-expanding CoreValve/Evolut R valves (both Medtronic)41.
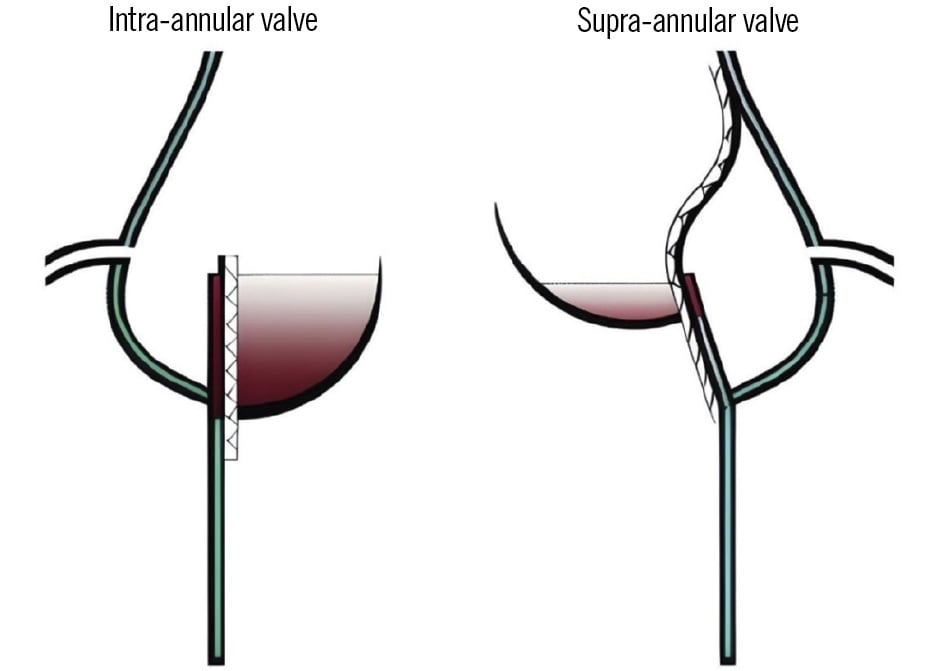
Figure 3. Comparison of neosinus volumes after TAVI. The neosinus is larger with an intra-annular than with a supra-annular prosthesis. The transcatheter aortic valve divides the aortic sinus into the neosinus (red area; from the valve prosthesis to the valve leaflet) and the anatomical sinus (sinus of Valsalva wall to the native aortic leaflet/valve prosthesis). The intra-annular valve creates a larger neosinus than the supra-annular valve. TAVI: transcatheter aortic valve implantation
Endothelial injury
Endothelial injury may occur on native endothelial surfaces, such as the aortic wall or sinuses of Valsalva, particularly in the context of overexpansion or severe commissural misalignment. These procedural factors may cause trauma to the endothelium and possibly contribute to thrombus formation. Delayed or incomplete endothelisation of bioprosthetic valve leaflets may also expose patients to prolonged and increased risks of thrombosis.
For the balloon-expandable SAPIEN 3 valve (Edwards Lifesciences), Midha et al observed a 10% larger valve expansion in patients with subclinical leaflet thrombosis, suggesting that overexpansion may cause endothelial injury, which may be the substrate for thrombosis41. Although this mechanism could potentially increase the risk of thrombosis, a recent cohort study by Sanz SÔ¡nchez et al found no significant differences in mortality or structural valve deterioration between patients who underwent post-dilatation (n=417) and those who did not (n=1,418)42.
Severe commissural misalignment of the native and prosthetic valves may also cause endothelial damage and delayed prosthesis endothelialisation, which was found to be associated with subclinical leaflet thrombosis43. As endothelial injury and a subsequent inflammatory response may play a role in thrombus formation, anti-inflammatory agents may prevent leaflet thrombosis. The recently presented Co-STAR trial investigated the effect of colchicine initiated 1 day before TAVI until day 12 after TAVI. This placebo-controlled, double-blind trial was prematurely stopped after inclusion of 120 patients because of a higher stroke rate in the placebo group. The primary outcome rate of atrial fibrillation occurred less frequently in the colchicine group, but the difference was non-significant. Leaflet thickening was significantly lower in the colchicine group compared to the placebo group (27.1% vs 54.2%)44. These findings suggest that inflammation plays a role in thrombus formation, and that anti-inflammatory agents may have a preventative role.
Specific cohorts
Valve-in-valve
Failure of bioprosthetic aortic valves necessitates valve-in-valve TAVI or repeat surgical aortic valve replacement. Simulations indicate that blood remains in contact with the valve for longer periods in valve-in-valve TAVI than in surgical replacement, potentially increasing the risk of valve thrombosis45. Observational studies have also identified valve-in-valve procedures as a predictor of both clinically significant valve thrombosis and subclinical leaflet thrombosis923. As the existing valve constricts the expansion of the new valve, the effective orifice area becomes smaller, resulting in the risk of patient-prosthesis mismatch. In a meta-analysis of 6 observational studies, valve-in-valve TAVI patients were older, had a higher comorbidity burden and surgical risk, and fewer patients had small valve sizes of ≤21 mm than patients with repeat surgical aortic valve replacement46. While no differences were observed in mortality, ischaemic events, or mean gradients at 30 days, valve-in-valve TAVI was associated with higher rates of patient-prosthesis mismatch compared with repeat surgical aortic valve replacement (13.5% vs 3.3%). Patient-prosthesis mismatch has been associated with an increased risk of clinically significant valve thrombosis in surgical bioprosthetic valve patients treated with valve-in-valve TAVI, with an incidence of 7.6% for clinically significant valve thrombosis within a median of 3 months47. In contrast, 129 patients with valve-in-valve TAVI were propensity score-matched with the same number of first-time TAVI patients in a recent study, and there was no difference in subclinical leaflet thrombosis on CT images at 30 days48. Given the small sample sizes of these studies, larger studies with long-term follow-up in valve-in-valve TAVI patients are warranted.
Bicuspid aortic valves
The elliptical geometry of bicuspid aortic valves challenges prosthesis expansion, potentially increasing the risk of valve thrombosis and affecting valve durability in these often young patients. In the NOTION-2 trial, death or disabling stroke were numerically higher for bicuspid than tricuspid valves after TAVI (6.1% vs 2.2%). However, clinically significant valve thrombosis was not detected in any patient with a bicuspid valve at 1 year49. Among 150 patients with severely stenotic bicuspid aortic valves treated with self-expanding valves, the 30-day incidence of valve thrombosis was 0.7%50. Recent observational data also found the 30-day rates of HALT to be higher in bicuspid aortic valve patients with suboptimal expansion of the balloon-expandable valve51. However, self-expanding valves might perform better than balloon-expandable valves due to more optimal expansion and circularity of the stent frame at the leaflet level in patients with bicuspid valves52. Data on long-term prevalence of subclinical thrombosis are lacking.
Self-expanding versus balloon-expandable valves
Self-expanding valves gradually expand to fit above the native annulus (supra-annular position), whereas balloon-expandable valves deploy with high radial force from a balloon and are generally aligned within the plane of the native annulus (intra-annular position). Each valve type has distinct tradeoffs53545556.
Clinical valve thrombosis rates have not been widely reported, but the REPRISE III trial noted higher rates of valve thrombosis (1.5% vs 0%) with the mechanically expandable LOTUS valve (Boston Scientific) compared with the self-expanding CoreValve53. A systematic review found that subclinical leaflet thrombosis was less common for supra-annular prostheses (7.0%, including CoreValve/Evolut R and ACURATE neo [Boston Scientific]) compared to intra-annular prostheses (12.1%, including SAPIEN I, XT, and 3 [Edwards Lifesciences]; LOTUS; CENTARA [Edwards Lifesciences]; and Direct Flow [Direct Flow Medical])57. The intra-annular self-expanding Portico valve (Abbott) showed the highest prevalence of subclinical leaflet thrombosis of 35.2%. Observational studies have yielded conflicting results with regard to valve type being a predictor of subclinical leaflet thrombosis or clinically significant valve thrombosis958. A recent meta-analysis reported subclinical leaflet thrombosis in 3.2% of patients receiving self-expanding valves and 5.7% with balloon-expandable valves, observed within a median of 98 days post-TAVI59. Bogyi et al have summarised the incidence of subclinical leaflet thrombosis for different valve types at different timepoints, but these rates need to be evaluated with caution due to the heterogeneity and size of the patient cohorts reported23.
The recently presented and not yet published ACURATE IDE trial failed to demonstrate non-inferiority of the self-expanding ACURATE neo2 valve (Boston Scientific) for the 1-year composite of all-cause death, stroke, or rehospitalisation compared with a mixed control group of self-expanding and balloon-expandable valves (Evolut and SAPIEN valves, respectively)60. The investigators are currently searching for a plausible explanation and have already noted that underexpansion was a problem in roughly 20% of the cases, in line with previous findings that underexpansion is an important predictor of HALT in TAVI patients1721. The hypothesis is that severe underexpansion of the ACURATE neo2 valve may have caused increased turbulent flow in the ascending aorta, leading to impaired washout within the sinuses of Valsalva. This disruption could contribute to the local formation of microthrombi originating from the aortic sinuses. Underexpansion might also cause creases in the leaflets, which could serve as sites for microthrombi development.
Another randomised controlled trial, the SMART Trial, evaluated 737 patients, mainly females, with severe aortic stenosis and a small aortic valve annulus (area of ≤430 mm2)61. Participants were randomised to receive either a supra-annular self-expanding valve or a balloon-expandable valve. The trial demonstrated non-inferiority for the 1-year primary composite outcome of death, disabling stroke, or heart failure rehospitalisation (9.4% and 10.6%). The self-expanding valve was superior for the 1-year primary outcome of bioprosthetic valve dysfunction (9.4% and 41.6%). Clinically significant valve thrombosis rates were found to be identical at 0.3% for both groups, but these event rates were obtained using TTE rather than CT and are therefore unlikely to be representative of the true occurrence of subclinical leaflet thrombosis.
Screening for valve thrombosis
Echocardiography, including transprosthetic gradient measurement, is recommended within 30 days after valve implantation (i.e., baseline), at 1 year, and annually thereafter following transcatheter and surgical bioprosthetic heart valve implantation. TOE should be considered if the TTE quality is poor or when prosthetic dysfunction or endocarditis is suspected1213. While cardiac CT scanning (4D-CT) is not routinely recommended, it can provide valuable insights when valve thrombus is suspected as a cause of impaired valve function. A key indicator for subclinical leaflet thrombosis or significant valve deterioration is an increase in the mean pressure gradient of 10 mmHg or more. A recent state-of-the-art paper includes detailed recommendations for Doppler echocardiographic measurements to assess aortic prosthesis dysfunction and defines red flags requiring further evaluation62. Whether patients in high-risk groups for subclinical leaflet thrombosis should undergo intensive screening at initial follow-up remains a topic of debate. Scoring systems including biomarkers, clinical risk factors, anatomy on CT, and procedural findings have been proposed to discriminate risk groups for subclinical leaflet thrombosis27, but this approach requires external prospective validation in an interventional study63]. A flowchart of the diagnosis and management of TAVI thrombosis has been proposed in the Central illustration.
Standard antithrombotic treatment
Optimal antithrombotic therapy for TAVI patients remains uncertain, but most randomised trials support a “less-is-more” approach. A recent state-of-the-art review summarises the knowledge from randomised controlled trials and observational studies, and we here provide a brief overview of standard antithrombotic treatment in TAVI patients64
Preprocedural
In patients without an indication for OAC, acetylsalicylic acid (ASA) is often administered preprocedurally to reduce the risk of periprocedural ischaemic events, but it remains unknown whether this comes at the cost of periprocedural bleeding64. In contrast, the recent POPular PAUSE TAVI trial (n=869), which included patients with an indication for OAC, demonstrated that continuation of OAC was not non-inferior to interruption – supporting the advice to discontinue OAC prior to the procedure65. The 30-day primary outcome of cardiovascular death, all strokes, myocardial infarction, major vascular complications, or major bleeding occurred in 16.5% in the continuation group versus 14.8% in the discontinuation group (difference of 1.7 percentage points [95% CI: −3.1 to 6.6]; p=0.18 for non-inferiority). The numerical difference was mainly due to fewer minor vascular complications; thromboembolic events did not increase in patients who interrupted the OAC treatment. There was no difference in outcome between the patients on a direct OAC (DOAC) (81.8%) or a vitamin K antagonist (VKA).
Periprocedural
Unfractionated heparin (UFH) is administered repeatedly during the procedure because of its short half-life, aiming to prevent catheter thrombosis and to reduce the risk of periprocedural thromboembolic events. At the time of vascular access, UFH is administered at a dose of 50-70 IU/kg66. The activated clotting time (ACT) is measured after UFH administration to confirm adequate anticoagulation with a target range of 250-300 seconds. The ACT should be reassessed every 20-30 minutes during the implantation. Protamine sulphate reverses UFH and is often administered towards the end of the procedure to minimise access site bleeding, or, rarely, in case of heparin-induced thrombocytopaenia. The beneficial effects of routine compared with selective protamine use was demonstrated in the ACE-PROTAVI trial (n=410 patients). Haemostasis within 20 minutes was significantly higher with routine (97.9%) rather than selective (91.6%) protamine use, and the time to haemostasis was significantly reduced (median of 181 [interquartile range IQR 120-420] seconds for routine vs 279 [IQR 122-260] seconds for selective protamine use; p=0.002)67.
The BRAVO 3 trial (n=802) compared UFH and bivalirudin, demonstrating non-inferiority for the 30-day composite outcome of net adverse clinical events68. However, due to its higher cost and no antidote, bivalirudin is unlikely to replace UFH in routine clinical practice but may be considered for patients with heparin intolerance.
Postprocedural
Current guidelines recommend lifelong single antiplatelet therapy after TAVI for patients without an indication for OAC. Dual antiplatelet therapy is often continued after TAVI only in patients with recent percutaneous coronary intervention (PCI; within 1-3 months), given the risk of stent thrombosis. When an indication for OAC is present, the guidelines advise OAC monotherapy. However, after a recent PCI, dual therapy with OAC plus a single antiplatelet agent is advised126970. OAC should not be initiated after TAVI in patients without a pre-existing indication, as two randomised trials showed harm compared to usual antiplatelet therapy2071. Although subclinical leaflet thrombosis was less often seen in patients on OAC, this came with a bleeding and an all-cause or cardiovascular mortality trade-off15207172.
Choice of oral anticoagulant
When oral anticoagulation is required after TAVI, the ENVISAGE-TAVI AF trial demonstrated that edoxaban was non-inferior to a VKA for the composite primary outcome, including all-cause mortality, myocardial infarction, ischaemic stroke, systemic thromboembolism, valve thrombosis, and major bleeding73. However, edoxaban was associated with a higher risk of major bleeding, primarily driven by increased gastrointestinal bleeding. Additionally, the ATLANTIS stratum 1 results indicated no significant differences for all outcomes between apixaban and VKA7172. A recent propensity-matched cohort study found that, over a 5-year period, patients on DOACs had lower mortality but higher stroke rates, with a similar incidence of major bleeding compared to VKAs74. Although robust long-term data on the safety and efficacy of DOACs in this setting are still lacking, their favourable outcomes in pivotal atrial fibrillation trials and ease of use may be decisive factors in clinical decision-making.
Treatment of valve thrombosis
Treatment decisions for the spectrum of valve thrombosis conditions, from subclinical leaflet thrombosis to clinically significant valve thrombosis, should carefully balance the risk of bleeding against the risks of thromboembolism and structural valve deterioration. Options include active surveillance, OAC with a DOAC or VKA, low-molecular-weight heparin or UFH, thrombolysis including ultraslow low-dose thrombolytic therapy, or surgical intervention.
Recently, a treatment algorithm for valve thrombosis in TAVI was proposed (Figure 4)75. Importantly, there is a major lack of knowledge about optimal treatment of subclinical leaflet thrombosis. The phenomenon can progress, regress, and resolve spontaneously, but anticoagulation can also resolve subclinical leaflet thrombosis immediately610. According to guidelines, it is reasonable to consider OAC if the gradients are elevated because of leaflet thickening (Class IIa, Level of Evidence B). Clinically significant valve thrombosis has a wide spectrum of clinical presentations, ranging from worsening of dyspnoea to cardiogenic shock or thromboembolic events. Guidelines state that use of VKAs and/or UFH should be considered before reintervention (Class I, Level of Evidence C)12. Ultraslow low-dose thrombolytic therapy, e.g., 25 mg alteplase infusion over 25 hours, is an additional treatment option, which primarily has been studied in patients with mechanical heart valve thrombosis7677. However, it also has shown effectiveness in a case report treating TAVI thrombosis after failure of oral anticoagulation78. In general, the treatment strategy should be tailored to the individual, carefully balancing the possible effects and side effects75.
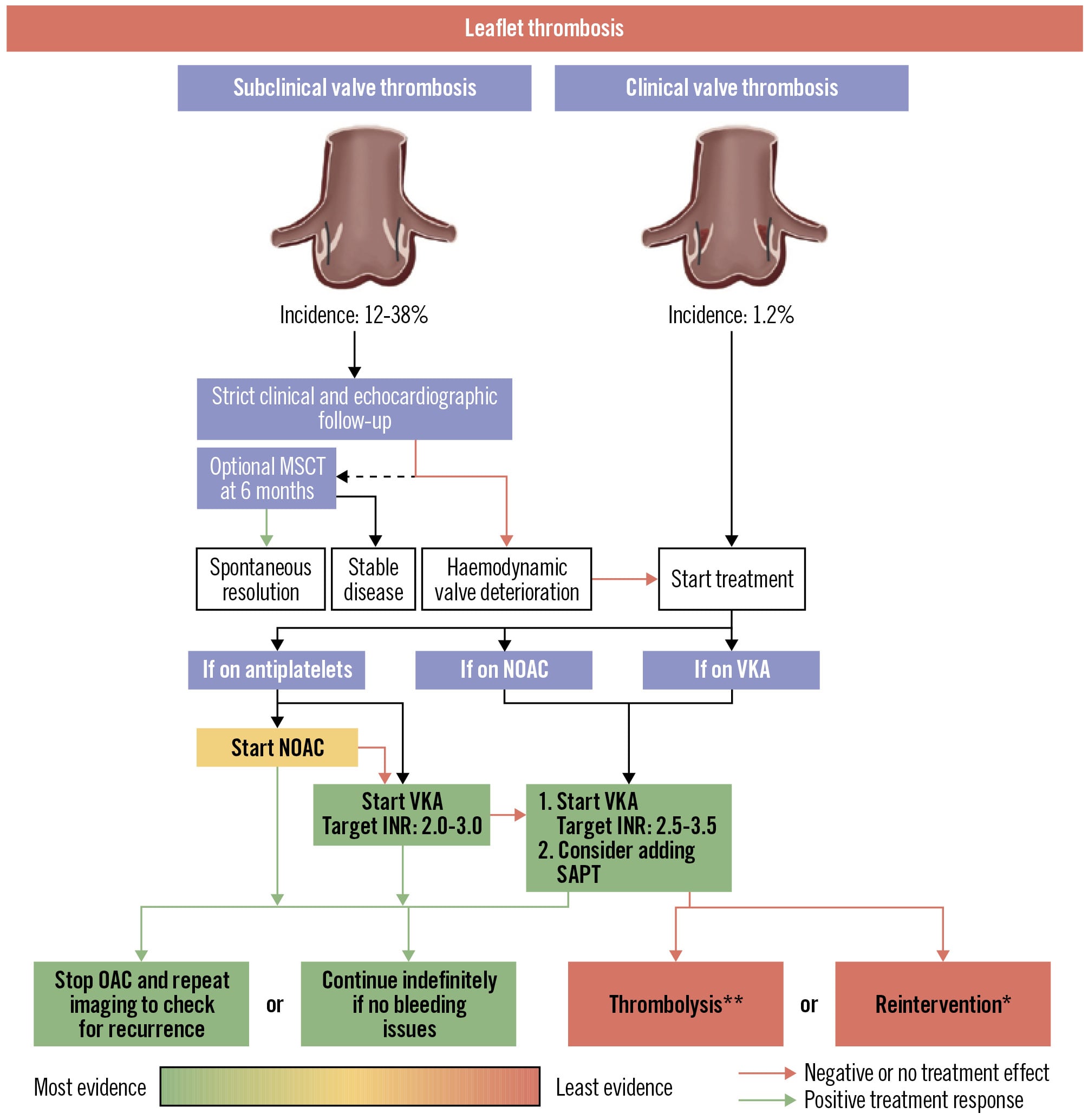
Figure 4. Treatment algorithm for valve thrombosis in TAVI. Reproduced with permission from75. Suggested course of action for patients with confirmed subclinical valve thrombosis or clinical valve thrombosis (CVT) based on clinical condition and current antithrombotic therapy. *To be repeated until CVT resolution. **Preference for ultraslow low-dose alteplase infusion over reintervention unless there is a high bleeding risk or failed previous thrombolysis. Preference for redo-TAVI over TAVI explant due to the high surgical risk associated with TAVI explant. INR: international normalised ratio; MSCT: multislice computed tomography; NOAC: non-vitamin K oral anticoagulant; OAC: oral anticoagulation; SAPT: single antiplatelet therapy; TAVI: transcatheter aortic valve implantation; VKA: vitamin K antagonist
Future developments
Novel drug therapies
The contact activation pathway of the coagulation system is activated in patients with aortic stenosis in relation to the severity of their condition79. On cardiac devices, the activation of factor XI (FXI) and factor XII contributes to thrombosis and inflammation, playing a key role in prothrombotic and degenerative processes2880. Reducing FXI levels, particularly in patients undergoing aortic valve replacement, was associated with a lower incidence of thrombotic events81. While data on contact activation pathway in TAVI patients are limited81, ongoing trials may include TAVI patients, although they do not systematically screen for subclinical leaflet thrombosis. To date, no dedicated trial on FXI inhibition after TAVI is ongoing or planned.
Developments in prosthesis design
The coating of prostheses and the use of polymeric prostheses could help reduce prosthesis thrombosis in the future. In silico models suggest that photo-triggered coatings of prosthetic valve leaflets could provide both antibacterial and thrombolytic effects. Coatings using the photosensitiser chlorin e6 (Ce6) grafted onto polyethyleneimine (PEI) can release reactive oxygen species (ROS) when exposed to near-infrared irradiation, destroying bacterial walls and breaking down fibrin thrombi in vitro and in vivo82.
Polymeric bioactive coatings, such as cross-linked polymeric nanogels loaded with releasable small molecules, offer antiplatelet, anticoagulant, and antimicrobial properties. One example is the ticagrelor-loaded bioactive coating (Triafluogel), which demonstrated antibacterial and antithrombotic effects without altering valve haemodynamics83.
Advances in polymeric materials like silicon-containing polycarbonate urethanes (SiPUU) show promise in improving prosthetic heart valve function and durability while reducing thrombogenic and degenerative risks. The TRIA polymeric valve (Foldax) has shown a favourable safety profile in surgical mitral and aortic valve replacement84. According to the company statement, a preclinical study is intended to support a first-in-human study in the TAVI setting. These polymeric valves could minimise the need for anticoagulation and reduce leaflet damage during the crimping process. Although promising, these concepts need to be tested in clinical trials of appropriate size and follow-up.
Computational/flow modelling
Computational modelling holds promise for analysing the biomechanical interactions between a TAVI prosthesis and patient anatomy and blood flow, helping to identify risk. Computational fluid dynamics (CFD) simulations of blood flow have identified wall shear stress separation points as predictors of valve thrombosis, particularly in valve-in-valve procedures. These simulations suggest that intra-annular valves are associated with a higher risk of thrombosis85. The International Organization for Standardization (ISO) working group proposed a CFD workflow to assess the risk of valve thrombosis in TAVI86. An in silico study showed how patient-specific anatomical and haemodynamic factors influence the volume of thrombosis and neosinus flow patterns87. A semiempirical, mathematical model linking patient-specific anatomical, valve, and flow parameters, using vorticity flux or circulation as a metric to quantify neosinus washout, was proposed to predict the likelihood of valve thrombosis88. By integration of in situ pressure sensors into the TAVI frame, it was possible to characterise clusters of flow and haemodynamic differences. Computational modelling using machine learning was able to detect leaflet dysfunction with >90% accuracy89.
Ongoing studies
Biomarkers of valve thrombosis
Given the potential harm of subclinical leaflet thrombosis and the proven harm of OACs in all TAVI patients (in the absence of OAC indications), the ATOM Study (n=750) aims to identify patients at increased risk of developing valvular thrombosis. This ongoing observational study investigates biomarkers assessing thrombosis and thrombolysis (ClinicalTrials.gov: NCT04234841). The ATOM Study is aiming for study completion at the beginning of 2028. Before that, we expect the RISTRATAVI study (n=100) to be published, in which thromboelastography is used to predict valve thrombosis at 0, 3, and 6 months (NCT03649594).
Antithrombotic therapy
The ACLO-TAVR Trial (n=230) will look at subclinical leaflet thrombosis at 3 months after TAVI when patients are randomised to clopidogrel versus ASA. The primary outcome is the incidence of valve thrombosis at 3 months; total follow-up will be 6 months (NCT05493657). The REAC-TAVI2 trial (n=1,206) is investigating ticagrelor versus ASA, also using valve thrombosis on CT as the primary outcome, but in this study at 3 and 12 months (NCT05283356).
The NOTION-4 Trial (n=324) is investigating lifelong ASA versus 3-month DOAC followed by lifelong ASA in patients without a known OAC indication, and lifelong DOAC versus left atrial appendage closure within 3 months followed by lifelong ASA in patients with atrial fibrillation. The primary outcome is subclinical leaflet thrombosis 1 year after TAVI, as well as thromboembolic and bleeding outcomes up to 5 years. Enrolment is complete, with 5-year follow-up expected in early 2029 (NCT06449469).
The CREATE trial (n=3,380) will investigate stopping antiplatelet therapy at 1 year after TAVI versus lifelong single antiplatelet therapy. The primary outcome is a composite of cardiac death, myocardial infarction, and ischaemic stroke at 1-year follow-up. Enrolment will be at 20 centres in China, with completion expected at the end of 2027 (ChiCTR2400087454).
Imaging
The POPular ATLANTIS study (n=2,500), a joint effort between the POPular TAVI and ATLANTIS study groups, is an ongoing randomised controlled trial on personalised CT-guided care, based on the appearance of subclinical leaflet thrombosis. Patients are randomised after 3 months to either standard of care (lifelong antiplatelet therapy) or a CT-guided approach. In the CT-guided arm, patients without subclinical leaflet thrombosis on CT stop antiplatelet therapy (if no other indication is present). In patients with subclinical leaflet thrombosis, antiplatelet therapy is replaced by an OAC until redo-CT 1 year after TAVI. Study completion is expected by the end of 2028 and will provide insight on a tailored approach using CT (NCT06168370). POPular PET TAVI (n=180) is an observational study focusing on the impact of subclinical leaflet thrombosis on valve degeneration 5 years after TAVI. In this study, the relation between valve thrombosis and valve degeneration is being investigated using PET-CT and the 18F-NaF marker for microcalcification. The end of enrolment is expected by early 2026 (NCT05758662). The Scottish SALTIRE Programme is aiming to provide insights with both biomarkers and PET-CT annually after TAVI for 4 years. The expected enrolment is 300 patients, and study completion is anticipated by mid-2028 (NCT06047561).
Conclusions
Subclinical leaflet thrombosis is common after TAVI, but its impact on clinical outcomes remains unclear. Associations with thromboembolic events and valve deterioration have been suggested, and these eventualities are particularly relevant as TAVI is increasingly used in younger, lower-risk patients. The mechanisms behind thrombosis formation on TAVI prostheses require further investigation but can broadly be explained by Virchow’s triad: blood flow stasis, endothelial injury, and hypercoagulability. Given the potential risks of antithrombotic therapy, specific treatment should currently be limited to patients with clinically significant valve thrombosis, i.e., increasing transvalvular gradients and symptoms of valve stenosis. Ongoing studies will help to further clarify the risk factors, clinical implications, and optimal management strategies for leaflet thrombosis in TAVI patients.
Conflict of interest statement
D. Trenk reports fees for consulting from Daiichi Sankyo and Eleva. F. Andreotti reports speaker or consultancy fees from Amgen, AstraZeneca, Bayer, Menarini, Novo Nordisk, and Servier, unrelated to this work. R.F. Storey reports institutional research grants from AstraZeneca and Cytosorbents; and personal fees from Abbott, Afortiori Development/Thrombolytic Science, AstraZeneca, Boehringer Ingelheim/Lilly, Bristol-Myers Squibb/Johnson & Johnson, Chiesi, Cytosorbents, Idorsia/Viatris, Novo Nordisk, PhaseBio, and Tabuk. R. Sørensen reports institutional research grant from Novo Nordisk foundation (unrelated to this work); and personal fees from Novo Nordisk for data monitoring committee work (unrelated to this work). S.D. Kristensen reports lecture fees from Chiesi (cangrelor); and a National Coordinating Investigator SOS-AMI departmental grant from Idorsia. J.M. ten Berg reports institutional research grants from ZonMw and Daiichi Sankyo. T. Geisler reports personal and institutional fees by Boston Scientific, Edwards Lifesciences, Medtronic, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Ferrer/Chiesi, and Pfizer, unrelated to this work. O. De Backer received institutional research grants and consulting fees from Abbott, Boston Scientific, Medtronic, and Microport. The other authors have no conflicts of interest to declare.

