Abstract
Aims: Accurate positioning of aorto-ostial coronary stents is challenging. Coronary CT angiography (CCTA) allows detailed imaging of the coronary sinuses and implanted stents. We utilised CCTA to evaluate the accuracy of aorto-ostial stenting and to assess the efficacy of conventional angiography for guiding these procedures.
Methods and results: We analysed 256-row CCTA scans in 23 patients who had undergone aorto-ostial stenting. Optimal stent positioning was defined as presence of the entire circumference of the proximal stent edge within an aorto-ostial landing zone (AOLZ), located within 1 mm of the aorto-ostial plane. Geographic miss was diagnosed when at least a section of the proximal stent edge was located proximal or distal to the AOLZ. CCTA findings were compared with review of the conventional two-dimensional implantation angiogram (two experienced operators). By CCTA, the entire circumference of the proximal stent edge was located within the AOLZ in only three (13%) cases, with geographic miss in the remainder. Conversely, conventional coronary angiography suggested complete lesion coverage to be present in 95% of cases and optimal location of the proximal stent edge within the AOLZ in 76%.
Conclusions: Geographic miss was common in aorto-ostial stenting and was underestimated by conventional two-dimensional angiography.
Introduction
Precise positioning of stents for treatment of aorto-ostial coronary lesions is difficult using conventional angiographic landmarks1-3. This is due to limitations of two-dimensional angiographic imaging in delineating and profiling the three-dimensional aorto-ostial anatomy, continuous movement of the guiding catheter relative to the ostium and the need to disengage the catheter from the ostium during stent deployment4.
The optimal site for positioning the proximal stent edge has been suggested to be inside an aorto-ostial landing zone (AOLZ) located within 1 mm of the aorto-ostial plane3. Implantation distal to the AOLZ leads to incomplete lesion coverage, inadequate treatment of dissections and possibly restenosis. Proximal stent placement within the aorta may impede stent endothelialisation and may prevent future re-engagement of the coronary artery with a guiding catheter. Implantation of both bare metal and drug-eluting stents in aorto-ostial coronary lesions is associated with a worse outcome compared to stenting of non-aorto-ostial lesions5,6. This may be due to undetected stent misplacement and geographic miss.
A recent retrospective angiographic analysis of cases of aorto-ostial stenting reported a high incidence of inaccurate stent placement3. It therefore seems that conventional two-dimensional angiographic assessment at the time of implantation is an imperfect tool for achieving accurate stent placement. Coronary computed tomography angiography (CCTA) enables three-dimensional analysis of the aorto-ostial plane which is often funnel-shaped7-12 and allows superior identification of the metallic stent struts13,14. The purpose of this study was to assess the positioning of previously implanted aorto-coronary stents in relation to the AOLZ in patients who subsequently underwent 256-slice CCTA in order to assess the need for a more accurate technique for placement of aorto-ostial stents.
Methods
PATIENT POPULATION
The CCTA database at the Lady Davis Carmel Medical Center was analysed, and patients who had previously undergone aorto-ostial coronary stenting were identified. Demographic, clinical and procedural patient data were collected and the stenting procedures were reviewed. The study was approved by the institutional review board with waiver of informed consent.
STUDY DEFINITIONS
The AOLZ was defined as the area along the axis of the coronary artery located within 1 mm proximal and distal to the aorto-ostial plane (Figure 1). Optimal stent positioning was defined as presence of the entire circumference of the proximal stent edge within the AOLZ (Figure 2). Geographic miss was diagnosed by either conventional angiography or CCTA when any part of the circumference of the proximal stent edge was located proximal or distal to the AOLZ (Figure 2). Geographic miss was classified as partial if any part of the proximal stent edge was located within the AOLZ. Otherwise, geographic miss was classified as complete. When at least part of the proximal stent edge was located within the AOLZ or when the proximal stent edge extended both proximal and distal to the AOLZ, geographic miss was attributed to the angulated vessel take-off from the aorta preventing precise localisation of the cylindrical stent structure within the constraints of the AOLZ and was therefore defined as anatomy-dependent. Geographic miss in which all aspects of the proximal stent edge extended beyond the AOLZ either proximally or distally was attributed to incorrect implantation of the stent and was defined as procedure-dependent.
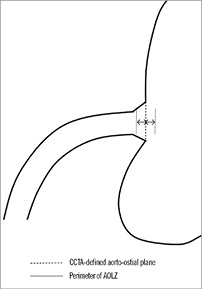
Figure 1. The aorto-ostial landing zone (AOLZ) was defined as the segment 1 mm proximal and distal to the CCTA-defined aorto-ostial plane.
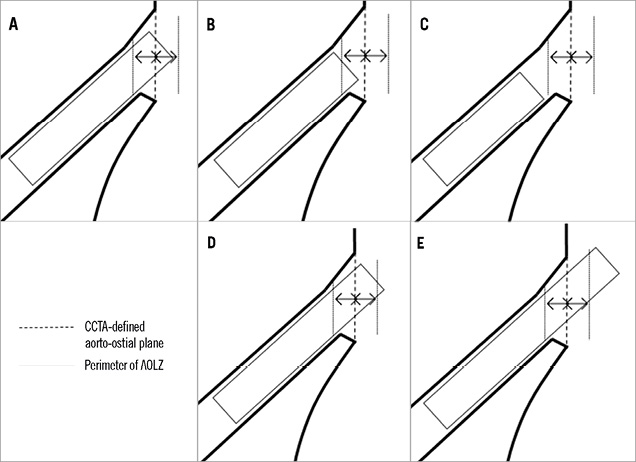
Figure 2. Location of the proximal stent edge in relation to the aorto-ostial plane and aorto-ostial landing zone (AOLZ). A) Optimal stent location; B) partial distal geographic miss; C) total distal geographic miss; D) partial proximal geographic miss; E) total proximal geographic miss.
STUDY ENDPOINTS
The primary study endpoint was determination by CCTA of optimal aorto-ostial stent implantation within the AOLZ. The secondary endpoint was assessment of the efficacy of conventional coronary angiography for assessment of optimal stent implantation, using CCTA as the gold standard.
REVIEW OF INVASIVE CORONARY ANGIOGRAMS
Aorto-ostial lesions were defined as lesions located within 3 mm of the aorto-ostial plane6. The index coronary angiograms were reviewed by two experienced interventional cardiologists (DAH and BSL) blinded to the CCTA findings who independently assessed the presence of complete lesion coverage and optimal stent implantation within the AOLZ.
CCTA ACQUISITION
CCTA was performed using a 256-row scanner (Brilliance iCT; Philips Healthcare, Cleveland, OH, USA), which has a longitudinal coverage of 8 cm, rotation times of 0.27-0.33 sec and a 120 kW generator. CCTA was performed in the prospectively triggered “step-and-shoot” mode (SAS) or with helical retrospective electrocardiographic (ECG) gating. ECG-based tube current modulation was used when possible with retrospective gating. Oral and/or intravenous beta-blockers were used to lower heart rate when >70 beats per minute (BPM). Sublingual nitroglycerine (0.4 mg) was given prior to CCTA to all patients with systolic blood pressure of ≥110 mmHg and no clinical contraindications (such as aortic stenosis). A contrast-enhanced scan was performed using a bolus of 50-80 ml iohexol (OMNIPAQUE™ 350 mg I/ml; GE Healthcare, Princeton, NJ, USA) injected into an antecubital vein at a flow rate of 5-6 ml/s, followed by a mixed 50% contrast/saline injection and then a 20-30 ml saline chaser bolus. Scans were performed at 120 kV with a slice collimation of 128×0.625 mm, with dual z-focal spot positions (which leads to doubling of the number of simultaneous imaged slices per gantry rotation, therefore 256-row acquisition). The helical scans (retrospective ECG gating) were performed with an effective tube current (rotation time product normalised by the pitch) in the range of 900-1500 mAs (effective) depending on BMI and body habitus, and a pitch of 0.14-0.18. The SAS scans were performed in patients with stable heart rhythm and heart rate <65 beats per minute with a tube current–x-ray ON time product of 160-300 mAs. Reconstruction was performed using a window centred at 75% of the R-R interval as default. For heart rates above 70 BPM an earlier reconstruction phase (usually 45% or less) was frequently used with retrospective gating.
CCTA ANALYSIS
CCTA scans were read by one author (RR) blinded to the invasive angiographic results. CCTA images were constructed using a cross-sectional technique (“double oblique”) to define the angle between the left main coronary artery (LMCA) or the proximal right coronary artery (RCA) and the longitudinal axis of the ascending aorta at the level of the corresponding coronary sinus (perpendicular to the aortic valve plane). Two-dimensional reformations of each coronary ostium at axial and sagittal orientations were reconstructed using original slice thickness (0.625 mm). The aortic-coronary junction was defined by outlining the borders of the aorta adjacent to the origin of the coronary artery and the borders of the coronary artery until the point where the coronary arterial walls became parallel to each other. The take-off angles of the coronary artery from the aorta were measured on each image11. The AOLZ was defined as the segment 1 mm proximal and distal to the CCTA-defined aorto-ostial plane. For each stent the location of the most proximal strut in relation to the aorto-ostial plane was examined in various views. The two views with greatest divergence in proximal strut location in relation to the aorto-ostial plane were identified.
Results
STUDY POPULATION
We identified 23 patients with previous aorto-ostial stenting who had undergone CCTA for clinical indications detailed below. Demographic and clinical patient characteristics are detailed in Table 1.
Procedural characteristics are detailed in Table 2. All stenting procedures had been performed electively or semi-electively in haemodynamically stable patients with stable angina or non-ST-segment elevation acute coronary syndromes. In 14/23 (61%) of the cases the stent was implanted within the left main coronary artery and in the remainder within the right coronary artery. An accessory floating wire was positioned within the coronary sinus in 4/23 (17%) of the cases for demarcation of the coronary ostium during percutaneous coronary intervention (PCI). In all other cases stent deployment was based on unaided visual estimation of the location of the aorto-ostial plane from an optimal angiographic plane.
CCTA was performed in five patients (22%) with severe aortic stenosis as part of an evaluation protocol prior to transcatheter aortic valve implantation and in the remainder for evaluation of stent patency due to recurrent chest pain or for follow-up imaging after unprotected left main coronary stenting.
CCTA FINDINGS
CCTA scan characteristics and findings are detailed in Table 3. Retrospective ECG gating was used in 20 (87%) patients. In all cases there was divergence in the distance between different points on the circumference of the proximal stent edge and the aorto-ostial plane (Figure 3) which averaged 2.0±1.3 mm (range: 0.3-5.0 mm). The most distally located point on the circumference of the proximal stent edge was located on average 0.3±2.1 mm proximal to the CCTA-defined aorto-ostial plane (range: –3.0 to 4.4 mm, a negative number implying incomplete coverage of the ostium). The most proximally located point on the circumference of the proximal stent edge was located 2.3±1.8 mm proximal to the CCTA-defined aorto-ostial plane (range: –1.1 to 5.9 mm). Complete lesion coverage was achieved in 18/23 (78%) of the cases. The proximal stent edge was located completely distal to the AOLZ in one (4%) artery, partially distal and partially within the plane in two (9%), both distal and proximal to the plane in two (9%), and completely proximal to the plane in another seven (30%) (Figure 4). In only three (13%) cases was the proximal stent edge completely contained within the AOLZ. These data represent partial geographic miss of the AOLZ by the proximal stent edge in 10/23 (44%) and total geographic miss in 10/23 (44%) of the cases. Sixty percent of the cases of geographic miss were classified as anatomy-dependent and 40% as procedure-dependent (Figure 5). Geographic miss cases had a greater coronary artery take-off angle in relation to the longitudinal axis of the ascending aorta (more downsloping origin) than optimally placed cases; however, this trend did not reach statistical significance (Figure 6).
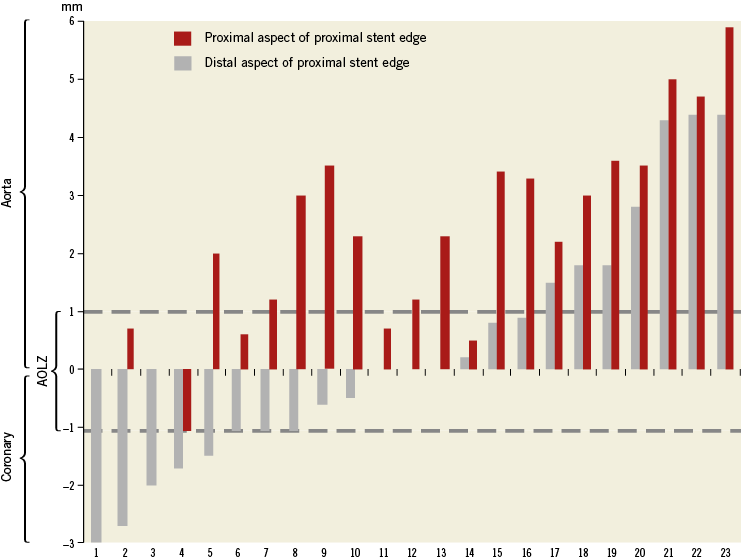
Figure 3. Location of most proximal and most distal struts at the proximal edge of individual stents in relation to the aorto-ostial plane.
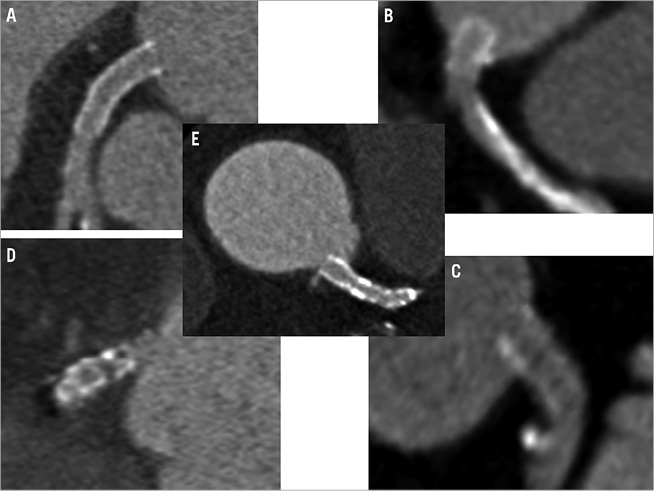
Figure 4. Representative CCTA images of aorto-ostial stent positioning in relation to the aorto-ostial plane. A) Optimal stent location; B) total proximal geographic miss; C) partial proximal geographic miss; D) total distal geographic miss; E) combined proximal and distal geographic miss.
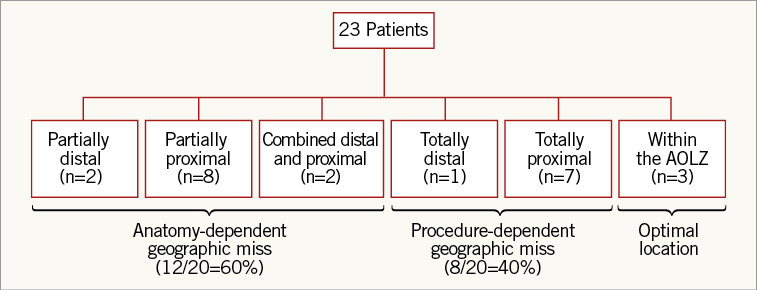
Figure 5. Classification of geographic miss as anatomy-dependent or procedure-dependent.
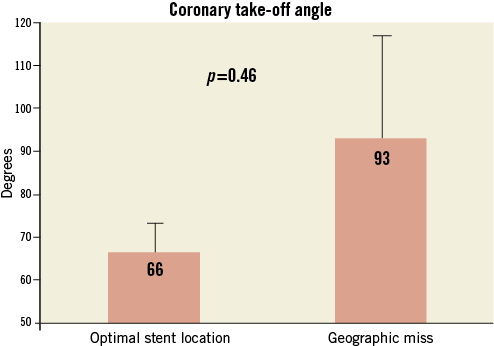
Figure 6. Relation between coronary artery take-off angle from the aorta and geographic miss.
REVISION OF PROCEDURAL ANGIOGRAMS
Twenty-one (91%) coronary angiograms were available for full review. Reviewers diagnosed complete lesion coverage in 20/21 (95%) and optimal location of the proximal stent edge within the AOLZ in 16/21 (76%) of the reviewed cases.
FOLLOW-UP
During follow-up out to six years following the index procedure one patient died, due to complications of transcatheter aortic valve implantation. Three patients (13%) underwent repeat coronary angiography within 266±124 days due to unstable angina and were diagnosed with in-stent restenosis. All cases of restenosis occurred following implantation of drug-eluting stents within the ostium of the right coronary artery. An accessory floating wire within the coronary sinus had been used in one of these patients during the index procedure.
Discussion
To our knowledge this is the first study to analyse the accuracy of aorto-ostial stent positioning using CCTA as the gold standard. We documented frequent geographic miss, leading to stents being implanted too distally or proximally relative to the aorto-ostial plane, which was not evident angiographically in most cases.
Reasons for stent misplacement were twofold. In 50% of cases of geographic miss the proximal stent edge was at least partially within the AOLZ. In another 10% of missed cases the proximal stent edge extended both proximal and distal relative to the AOLZ. These cases are likely attributable to the funnel-shaped aorto-ostial anatomy with angulated take-off from the aorta11 which cannot be fully appreciated by two-dimensional imaging, precluding optimal positioning of the cylindrical proximal stent edge within an asymmetric AOLZ. In only 40% of cases of geographic miss were all aspects of the proximal stent edge located distal or proximal to the AOLZ. These cases may be attributed to operator-dependent factors and to inadequacy of currently available imaging and angiographic techniques to delineate accurately the aorto-ostial plane. Indeed, three-dimensional angiographic analysis of the left main coronary artery has demonstrated significant anatomic variability which was not apparent on conventional two-dimensional angiography15.
Implications for optimising aorto-ostial stenting
Our findings suggest that achieving optimal stent implantation in aorto-ostial lesions requires two complementary approaches: improved techniques for identifying the aorto-ostial plane and development of specially designed stents compatible with the complex aorto-ostial anatomy. Several strategies have been proposed to improve fluoroscopic detection of the aorto-ostial plane. Optimal utilisation of these approaches depends on an angiographic view that is orthogonal to the aorto-ostial plane. Insertion of an accessory free-floating guidewire from the guide catheter into the coronary sinus in order to mark the aorto-ostial plane has been proposed; however, the efficacy of this manoeuvre is unknown16. Insertion of a tail anchor guidewire within the proximal stent cell (Szabo technique) to prevent deep insertion beyond the aorto-ostial plane has been suggested to improve accuracy of stent implantation1,17,18; however, the efficacy and safety of this approach are controversial19. The Ostial PRO® device (Merit Medical Systems, Inc., South Jordan, UT, USA) has been reported to facilitate accurate aorto-ostial stenting in a small cohort of patients20. However, further research is needed to evaluate the long-term outcome of Ostial PRO-guided PCI in a large patient cohort. Funnel-shaped stents such as the Cappella Sideguard® self-expanding stent (Cappella Medical Devices Ltd, Galway, Ireland) may potentially achieve more complete coverage of the aorto-ostial lesion without protruding into the aorta21. Importantly, the efficacy of these techniques has not been evaluated by CCTA which allows accurate (three-dimensional) assessment of the aorto-coronary anatomy. It remains to be seen whether new imaging techniques such as three-dimensional rotational angiography will allow superior and real-time “CT-like” image reconstruction and more accurate stent positioning.
Limitations
This retrospective single-centre study included a small number of patients, therefore limiting the statistical power of the findings. The small number of patients precluded analysis of predictors of geographic miss and subsequent target lesion failure. However, the purpose of this hypothesis-generating study was to analyse acute procedural stent positioning. Since studied patients had undergone CCTA for clinical indications, we cannot rule out selection bias with inclusion of patients who had adverse outcome of stenting and complex aorto-ostial anatomy. Specialised techniques for fluoroscopic identification of the aorto-ostial plane were employed in a minority of the patients; however, the utility of these methods is unproven.
Conclusions
Conventional two-dimensional angiographic assessment at the time of implantation is an imperfect tool for achieving accurate stent placement. Moreover, current stent designs are often unable to offer optimal lesion coverage within the AOLZ. These limitations underlie the high rate of geographic miss observed in our study. CCTA seems to be a useful tool which provides new insights into the assessment and follow-up of patients undergoing aorto-ostial stent implantation.
| Impact on daily practice Aorto-ostial stenting is technically challenging and is associated with worse outcome than non-ostial coronary interventions. Our CCTA analysis of stented aorto-ostial lesions revealed a high prevalence of stent geographic miss which was undetected by conventional angiography. Our findings highlight the clinical need for improved intraprocedural imaging, advanced techniques to guide these procedures and dedicated stent designs for optimal coverage of the unique aorto-ostial anatomy. CCTA should be used as the benchmark for assessing the efficacy of novel approaches for aorto-ostial stenting. |
Conflict of interest statement
R. Jaffe has submitted a patent application for a novel aorto-ostial stenting device. The other authors have no conflicts of interest to declare.

