
Abstract
Aims: Aortic valve preparation by performing balloon aortic valvuloplasty (BAV) has always been considered mandatory during transcatheter aortic valve implantation (TAVI) procedures. We aimed to investigate the feasibility and safety of performing balloon-expandable TAVI with moderate or with no predilatation (PD).
Methods and results: Overall, 121 patients underwent TAVI with no PD and 392 with moderate PD. TAVI endpoints and adverse events were considered according to the Valve Academic Research Consortium (VARC)-2 definitions. Device success for the entire cohort was 95.1%. Post-dilatation was performed in nine patients in the no PD group (7.4%) and in 40 patients in the moderate PD group (10.2%) (p=0.06). Total fluoroscopy time and the amount of contrast used were lower in the no PD group. All-cause mortality up to 30 days was 3.3% in the no PD group vs. 3.6% in the moderate PD group (p=0.89). VARC-2 defined complication rates at 30 days including cerebrovascular accident (CVA)/transient ischaemic attack (TIA) were similar between groups. Overall, there was no significant difference in survival rates between the two groups (HR 1.33, 95% CI: 0.75-2.35; p=0.34).
Conclusions: Balloon-expandable TAVI with moderate or without balloon PD is feasible and safe. The omission of PD in appropriate cases was associated with reduced fluoroscopy time without affecting procedural success.
Abbreviations
AU: Agatston units
AS: aortic stenosis
BAV: balloon aortic valvuloplasty
CVA: cerebrovascular accident
NYHA: New York Heart Association
PD: predilatation
PVL: perivalvular leak
ROC: receiver operating characteristic
TAVI: transcatheter aortic valve implantation
TIA: transient ischaemic attack
TEE: transoesophageal echocardiography
VARC: Valve Academic Research Consortium
Introduction
Transcatheter aortic valve implantation (TAVI) has emerged as a treatment option for inoperable or high-risk surgical patients with severe aortic stenosis (AS)1,2. Aortic valve preparation by performing balloon aortic valvuloplasty (BAV) has, so far, been considered mandatory, especially when using balloon-expandable valves. Nonetheless, BAV-inherent risks include stroke caused by distal embolisation, atrioventricular conduction disturbances requiring permanent pacing, and severe aortic regurgitation with intraprocedural haemodynamic instability3-5.
The rationale for performing BAV prior to TAVI is that it facilitates device introduction within the diseased aortic valve, enables an optimised expansion by reducing radial counterforces and enables final measurement of the aortic annulus6. Notwithstanding, new device generations with reduced profile sizes, improved navigability and strong radial forces allow a safe valve deployment without the need for aggressive valve preparation. By avoidance of the latter, preservation of the native aortic valvular complex architecture until valve deployment could conceivably facilitate better prosthetic anchoring. Moreover, utilisation of computed tomography (CT) and three-dimensional echocardiography enables better annular and coronary height assessment, further minimising the benefits of BAV during TAVI.
Previous reports of performing TAVI without predilatation (PD) included self-expanding valves7-9, transapical balloon-expandable valves10,11, or small case series of transfemoral balloon-expandable implantations12,13. Moderate PD during TAVI was only examined in one previous report which included only self-expanding valves14. The purpose of the present study was to assess the feasibility and safety of performing balloon-expandable TAVI with moderate or without PD.
Methods
We retrospectively examined consecutive patients with severe symptomatic AS who underwent Edwards SAPIEN or SAPIEN XT (Edwards Lifesciences, Irvine, CA, USA) TAVI during a two and a half year period at our institute. All patients had congestive heart failure with New York Heart Association (NYHA) Class II-IV symptoms. All underwent pre-procedural coronary angiography to assess the need for revascularisation. Aortic valve disease was assessed initially with transthoracic echocardiography followed by an ECG-gated, multislice CT angiography study with a Siemens Somatom Cardiac 64 scanner (Siemens Medical Solutions USA Inc., Malvern, PA, USA). Aortic valve calcification was quantified by a standard Agatston methodology for all available non-contrast CT scans, with a threshold for calcium detection set at 130 Hounsfield units (HU)15. Prosthetic valve size selection was based on CT or immediate pre-procedural three-dimensional (3D) transoesophageal echocardiography (TEE). TAVI was performed under general anaesthesia in all cases. All patients were considered high risk for valve surgery by our institutional Heart Team. The vascular access approach was chosen on the basis of the individual patient’s risk profile. Following valve deployment, TEE was used to assess the severity of perivalvular leak (PVL). Baseline clinical, echocardiographic and procedural details for TAVI were recorded for all patients including one-month clinical and echocardiographic assessments during a follow-up visit. TAVI endpoints, device success and adverse events were considered according to the Valve Academic Research Consortium (VARC)-2 definitions16. The study was approved by the Institutional Review Board at Cedars-Sinai Medical Center.
MODERATE VERSUS NO PREDILATATION
From April 2012 we started performing TAVI with moderate or with no PD approaches. Previously, a “regular” PD approach included utilisation of 20 mm, 23 mm and 25 mm valvuloplasty balloons for the 23 mm, 26 mm and 29 mm valves implanted, respectively. The moderate PD group included patients who underwent BAV during TAVI using small-size balloons. This included the Z-MED II™ balloon (B. Braun Interventional Systems Inc., Bethlehem, PA, USA) with a diameter of ≤18 mm for the 23 mm implanted valves and of ≤22 mm for the 26 or 29 mm implanted valves. In the no PD group, intraprocedural BAV was omitted. The decision to perform the procedure with moderate or without PD as well as the valvuloplasty balloon size to be used was left to the discretion of the operator. Indications for using moderate PD instead of direct TAVI were severe or asymmetric calcification of the aortic valve, small aortic valve area (<0.5 cm2) and bicuspid aortic valve anatomy.
STATISTICAL ANALYSIS
All data were summarised and displayed as mean±standard deviation (SD) for continuous variables, and as number (percentage) of patients in each group for categorical variables. The Student’s t-test and Pearson χ² tests were used to evaluate statistical significance between continuous and categorical variables, respectively. A predictive model for moderate vs. no PD, incorporating variables associated with a significance level of less than 0.1, was designed. This model was used to generate a propensity score for moderate vs. no PD. Receiver operating characteristic (ROC) analysis was used to assess the predictive value of this model for moderate vs. no PD. A Cox proportional hazards model was used to assess overall survival. The propensity score was then added to the Cox proportional hazards model in order to assess the adjusted overall survival and also added to a binary logistic model in order to assess the influence of moderate vs. no PD on the likelihood of PVL ≥mild, post-dilatation, second valve, new permanent pacemaker implantation, 30-day cerebrovascular accident (CVA)/transient ischaemic attack (TIA), 30-day or one-year mortality, fluoroscopy time >15 minutes and contrast utilisation >90 ml. All of the analyses were considered significant at a two-tailed p-value of less than 0.05. The SPSS statistical package, Version 20.0 (IBM Corp., Armonk, NY, USA) was used to perform all statistical evaluation.
Results
Between April 2012 and December 2014, 556 patients underwent Edwards SAPIEN/SAPIEN XT TAVI at our institute. We excluded from analysis the 38 patients who had valve-in-valve implantations, three patients who had previous BAV within 30 days, and two patients who had “regular” PD (20 mm balloon valvuloplasty before a 23 mm valve implantation in both cases). Of the remaining 513 patients, 121 patients underwent TAVI with no PD and 392 underwent TAVI with moderate PD. The mean follow-up period was 14.8±9.9 months. The transfemoral approach was used in 82.5% of the cases, the transapical approach in 6.0%, the transaortic approach in 10.5% and the subclavian approach in 1.0%. The baseline clinical patient characteristics and pre-TAVI imaging details of the study population are shown in Table 1. Several baseline characteristics were not similar between groups: patients in the no PD group were younger (80.6±9.4 vs. 83.0±8.3; p=0.006), the percentage of males was lower (52.1% vs. 64.8%; p=0.012), they had a higher frequency of previous stroke (19.8% vs. 12.2%; p=0.036) and a lower frequency of peripheral artery disease (28.9% vs. 43.1%; p=0.005) compared to patients in the moderate PD group. Echocardiography demonstrated significantly higher mean aortic valve area and smaller aortic valve pressure gradients in the no PD group, and their CT scan analysis demonstrated significantly smaller mean annulus diameter and area (Table 1). Data on aortic valve CT calcification scoring were available for 404 out of 513 patients. The average calcification score was 2,232±1,323 Agatston units (AU) in the no PD group compared to 4,214±2,253 AU in the moderate PD group (p<0.001).
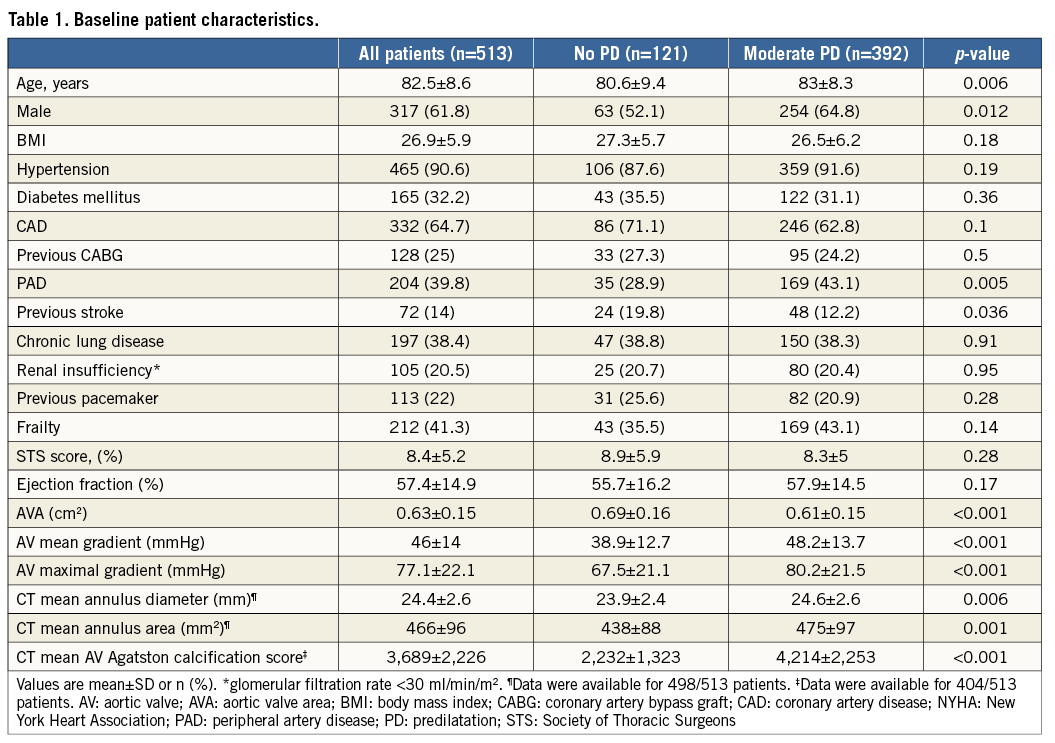
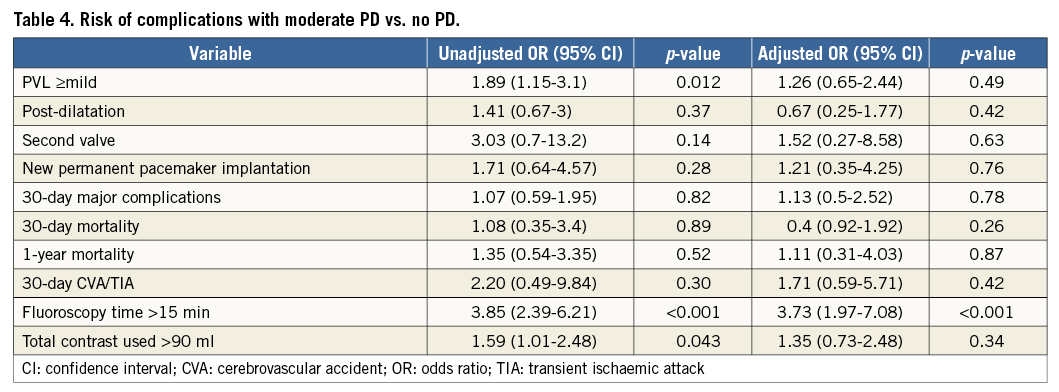
The mean valvuloplasty balloon diameter in the moderate PD group was 16.1±2.5 mm. The mean PD valvuloplasty balloon diameter/CT mean annulus diameter was 0.65:1. In the moderate PD group, balloon size exceeded the minor axis dimension of the aortic annulus in only 6/392 (1.5%) cases. The range of balloon diameters used for each of the three valve sizes implanted is shown in Figure 1. Procedural details are shown in Table 2. There were no significant differences between valve type, size or vascular access site between the groups. The device success rate was 98.3% (119 of 121) for the patients in the no PD group compared with 94.1% (369 of 392) for the patients in the moderate PD group (p=0.06). Post-dilatation was performed in nine patients in the no PD group (7.4%) and in 40 patients in the moderate PD group (10.2%) (p=0.37). Difficulty to cross the native valve with the bioprosthesis mandating utilisation of the buddy balloon technique was encountered in seven cases – one in the no PD group (0.8%) and six in the moderate PD group (1.5%). Total fluoroscopy time was significantly lower in the no PD group (12.3±5.1 minutes vs. 17.1±8.4 minutes; p<0.001). The total amount of contrast material used was significantly lower in the no PD group (78.5±37.1 ml vs. 86.9±41.1 ml; p=0.048). There was less post-procedural PVL in the no PD group: 80.2%, 19.8%, 0%, 0% vs. 67.3%, 26.8%, 5.6%, 0.3% for no/trace, mild, moderate and severe PVL, respectively (p=0.015). The mean Agatston calcification score of patients with no/trace PVL was 3,460±2,100 AU vs. 4,238±2,462 AU for patients with mild or higher PVL (p=0.003).
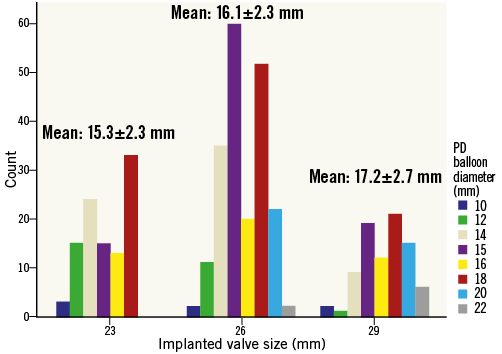
Figure 1. Valvuloplasty balloon sizes used for moderate PD according to implanted valve diameter. The mean PD valvuloplasty balloon diameter used for the moderate PD group (n=392) was 16.1±2.5 mm. Data for each of the three implanted valve sizes are shown. The mean PD valvuloplasty balloon diameter/CT mean annulus diameter was 0.65:1.
All-cause mortality up to 30 days was 3.3% in the no PD group vs. 3.6% in the moderate PD group (p=0.89). Thirty-day complications, according to VARC-2 criteria including CVA/TIA, were similar between groups (Table 3). Six-month mortality was 9.4% in the no PD group vs. 10.7% in the moderate PD group (p=0.71). Overall, there was no significant difference in survival rates between patients who underwent TAVI with moderate PD compared to patients who underwent TAVI without PD (HR 1.33, 95% CI: 0.75-2.35; p=0.34) (Figure 2A).
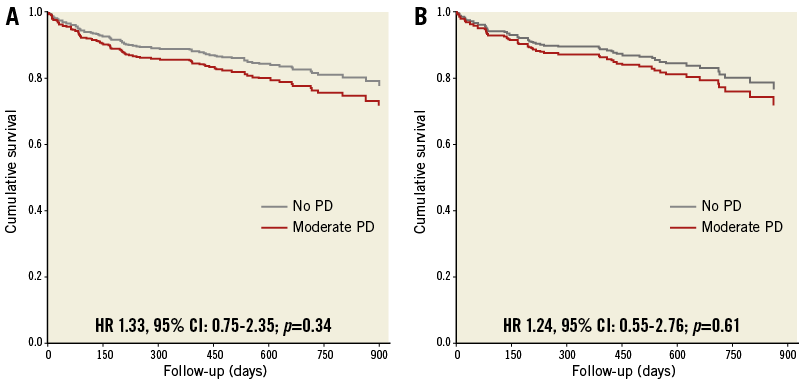
Figure 2. Overall survival during the study follow-up period. A) A Cox proportional hazard analysis for overall survival up to 900 days in patients who underwent TAVI with moderate PD compared to patients who underwent TAVI without PD (HR 1.33, 95% CI: 0.75-2.35; p=0.34). B) A Cox proportional hazard analysis for overall survival up to 900 days in both groups adjusted with the propensity score (HR 1.24, 95% CI: 0.55-2.76; p=0.61).
MULTIVARIATE ANALYSIS
A predictive model for moderate vs. no PD, incorporating variables associated with a significance level of less than 0.1 had a strong predictive value for this parameter (AUC 0.86, 95% CI: 0.81-0.90, p<0.001). These variables included age, gender, coronary artery disease, peripheral artery disease, previous stroke, aortic valve area, aortic valve mean gradient, aortic valve mean annulus diameter and aortic valve calcification score. This model was used to generate a propensity score for moderate vs. no PD. With adjustment using this score, there was no difference in overall survival and in the likelihood of PVL ≥mild, post-dilatation, second valve, new permanent pacemaker implantation, 30-day CVA/TIA, and 30-day or one-year mortality for moderate vs. no PD (Figure 2B, Table 4). With adjustment, there was still a difference in fluoroscopy time >15 minutes but not with contrast utilisation >90 ml.
Discussion
The results of the present study indicate that performing balloon-expandable TAVI with moderate or without PD is feasible and safe. In the study period of approximately two and a half years, we did not use large-size valvuloplasty balloons during TAVI. A total of 121 patients were treated with no PD and 392 with moderate-size valvuloplasty balloons. We found a high device success rate in both groups, and a shorter fluoroscopy time and lower amount of contrast used in the no PD group. We also found similar short-term major complication rates for both groups which are comparable to previous reports.
Since the introduciton of TAVI, BAV has been considered as a mandatory step in the procedure7. The purposes of predilatation are to increase the aortic valve orifice to –in turn– facilitate the introduction of the device within the diseased aortic valve, to reduce radial counterforces, thus optimising prosthesis expansion, and to help in the final assessment of annular dimensions and rule out coronary ostium obstruction just before valve implantation6. Performing BAV with large diameter balloons has been associated with an increase in conduction abnormalities, embolic adverse events and acute haemodynamic instability due to acute severe aortic regurgitation3-5. New-generation devices with reduced profile sizes and increased radial forces, along with advances in the pre-TAVI imaging techniques, have set the ground for the performance of TAVI with moderate PD or without PD7-14,17,18.
A direct TAVI approach was first reported by Grube et al in 20117. This was a pilot study which included 60 consecutive patients treated with a self-expanding device via the transfemoral approach. They demonstrated a high success rate, a low permanent pacemaker implantation rate, similar acute haemodynamic effects and lower rates of PVL compared to their historical cohort in which PD was employed. Omission of BAV was described in 50 patients undergoing transapical balloon-expandable TAVI and was found to be effective and safe10. Direct transfemoral balloon-expandable TAVI was only evaluated in small case series12,13,17, and in a recent series which included both self-expanding and balloon-expandable valves (n=28 vs. 51, respectively)18. Although the number of patients was limited, the procedural success rate and short-term complication rates were acceptable in all of these reports. A moderate predilatation approach with undersized valvuloplasty balloons was found to decrease the pacemaker implantation rate significantly after TAVI using a self-expanding device14. To the best of our knowledge, our study is the first to describe and compare an approach of direct TAVI versus moderate PD. Moreover, in the only previous report of moderate PD, the average PD balloon diameter was 21.3 mm (range: 18-23 mm)14 compared to a lower average balloon diameter used in the present study (16.1 mm; range: 10-22 mm).
There were several differences in baseline characteristics between the moderate and the no PD groups. The patients in the no PD group were four years younger and had a higher proportion of females. Pre-TAVI imaging revealed larger aortic valve areas and lower pre-TAVI gradients, lower mean aortic annulus diameter and area, and lower mean aortic valve calcification scores for the patients in the no PD group. This could be attributed to the fact that the decision to perform TAVI with moderate or without PD was left to the discretion of the operator and was based on the comprehensive pre-TAVI evaluation and tailored for each patient individually. Since the introduction of the direct TAVI approach, concerns have been raised about omitting BAV –especially in cases of bulky calcified aortic valve, valve area <0.4 cm2, eccentric residual valvular orifice and larger bioprosthesis6,12,18,19. Pre-procedural CT and echocardiographic evaluation of the native valve, including calcification severity, homogeneity of the calcification within the valve apparatus, severity of AS and valve anatomy (e.g., symmetric tricuspid valve versus asymmetric bicuspid valve), were the factors that helped in our decision whether to use the direct approach or to predilate the valve moderately.
As in previous series, device success among our patients was high and similar between groups (overall, 95.1%). Not surprisingly, by omitting PD we were able to decrease fluoroscopy time (14.2 vs. 18 minutes; p=0.001) and amount of contrast material used (78.5 ml vs. 86.9 ml; p=0.048) significantly. Omitting PD did not result in an increased need to perform post-dilatation (7.4% vs. 10.2% in the no PD and moderate PD groups, respectively). The post-procedural PVL rates were lower in the no PD group only in the univariate analysis (Table 4). A trend towards lower post-dilatation rates and lower residual PVL following direct TAVI has been demonstrated in the past7,8. Nonetheless, in our cohort the significantly lower aortic valve calcification scores of patients in the no PD group (2,232 AU vs. 4,214 AU; p<0.001) probably led to the lower post-procedural PVL. It has recently been shown by our group as well as by others that the amount of aortic valve calcification correlates highly with the severity of PVL and the frequency of post-dilatation after balloon-expandable TAVI20,21. This is supported by our finding of significantly higher calcification scores in patients who had mild or higher PVL post-procedurally (4,238 AU vs. 3,460 AU; p=0.003).
The safety of omitting large-size valvuloplasty balloons is further reinforced by our short-term mortality and major complication rates. The overall 30-day mortality of 3.5%, CVA/TIA of 3.1% (1.7% in the no PD group; p=0.29) and myocardial infarction of 0.8% are comparable with the previously published randomised trial and registries of balloon-expandable valves22-24 and the previous series examining the direct TAVI approach7,8,10.
We found a similar new permanent pacemaker implantation rate at 30 days in both groups (5.3% vs. 8.7% in the no PD and moderate PD groups, respectively; p=0.28). Lange et al examined 237 patients who underwent TAVI with a self-expanding device and found a new permanent pacemaker implantation rate of 27.1% in patients who underwent BAV during TAVI with a 25 mm balloon compared to a rate of 15.4% in patients treated with smaller balloons (p=0.04)14. To explain their results they suggested a two-hit model, in which the trauma inflicted by a larger valvuloplasty balloon is a first hit, usually insufficient to generate a persistent high degree atrioventricular block, unless a second hit is applied to the conduction system by the valve frame. Our series included only balloon-expandable valves in which the risk of pacemaker implantation is significantly reduced compared to self-expanding valves25. Moreover, the moderate PD approach in the present study included much smaller valvuloplasty balloons compared to the previous series described above (16 mm vs. 21.3 mm), being larger than the minor axis of the aortic annulus in only 1.5% of cases, with the balloon probably not making contact with the aortic annular margins in the remaining 98.5% of cases. We cannot therefore conclude whether omitting BAV in selected cases can minimise the risk of atrioventricular conduction abnormalities.
Study limitations
The present study represents a retrospective, single-centre experience with moderate or without PD approaches. The decision between approaches, as well as predilatation valvuloplasty balloon size selection, was left to the discretion of the operator and was not based on prospective solid inclusion criteria for each of the two approaches. Nonetheless, during the study period, 513 patients underwent balloon-expandable TAVI with moderate or no PD and had a high procedural success rate and low overall complication rates. Randomised trials evaluating the effects of regular, moderate and no PD are needed to provide insights into the differences between different intensities of valve preparation and to determine the criteria for the safe utilisation of each approach. The currently ongoing randomised multicentre EASE-IT trial is examining the effect of performing balloon-expandable transapical TAVI with and without PD and may answer some of these questions26.
Conclusions
Balloon-expandable TAVI with moderate or without balloon PD is feasible and safe. The omission of PD in appropriate cases was associated with reduced fluoroscopy time without affecting procedural success.
| Impact on daily practice Aortic valve preparation by performing BAV was previously considered mandatory during TAVI procedures. BAV-inherent risks including stroke and conduction abnormalities as well as reduced device profile size established the rationale for safe valve deployment without the need for aggressive valve preparation. We used an approach of performing TAVI with moderate or with no predilatation in 513 consecutive patients and found overall high device success and similar short-term major complication and mortality rates for both groups, which are comparable to previous reports. Balloon-expandable TAVI can be performed safely without the need for large valvuloplasty balloons. |
Conflict of interest statement
R.R. Makkar has received grant support from Edwards Lifesciences and St. Jude Medical, is a consultant for Abbott Vascular, Cordis, and Medtronic, and holds equity in Entourage Medical. H. Jilaihawi is a consultant for Edwards Lifesciences, St. Jude Medical, and Venus MedTech. The other authors have no conflicts of interest to declare.

