Abstract
The clinical value of fractional flow reserve and non-hyperaemic pressure ratios are well established in determining an indication for percutaneous coronary intervention (PCI) in patients with coronary artery disease (CAD). In addition, over the last 5 years we have witnessed a shift towards the use of physiology to enhance procedural planning, assess post-PCI functional results, and guide PCI optimisation. In this regard, clinical studies have reported compelling data supporting the use of longitudinal vessel analysis, obtained with pressure guidewire pullbacks, to better understand how obstructive CAD contributes to myocardial ischaemia, to establish the likelihood of functionally successful PCI, to identify the presence and location of residual flow-limiting stenoses and to predict long-term outcomes. The introduction of new functional coronary angiography tools, which merge angiographic information with fluid dynamic equations to deliver information equivalent to intracoronary pressure measurements, are now available and potentially also applicable to these endeavours. Furthermore, the ability of longitudinal vessel analysis to predict the functional results of stenting has played an integral role in the evolving field of simulated PCI. Nevertheless, it is important to have an awareness of the value and challenges of physiology-guided PCI in specific clinical and anatomical contexts. The main aim of this European Association of Percutaneous Cardiovascular Interventions clinical consensus statement is to offer up-to-date evidence and expert opinion on the use of applied coronary physiology for procedural PCI planning, disease pattern recognition and post-PCI optimisation.
Introduction
Intracoronary physiological assessment is acknowledged as a valuable strategy to identify the presence of flow-limiting epicardial stenoses in patients with chronic coronary syndromes (CCS) and to determine an indication for percutaneous coronary interventions (PCI)1. Yet, its role in procedural planning and in assessing the functional results of PCI is less clear.
In practice, it had been previously assumed that once the presence of flow-limiting disease was confirmed with a single pressure guidewire measurement, PCI guided by angiography should lead to effective restoration of vessel conductance. However, studies with physiological post-PCI evaluation based on fractional flow reserve (FFR) and non-hyperaemic pressure ratios (NHPR) demonstrate that this supposition is not correct and that relying on angiographic guidance alone can be associated with suboptimal functional results post-PCI in many cases234.
This document addresses the use of coronary physiology to plan and guide PCI using physiological tools, providing evidence from the literature, expert opinion from operators, and revisiting available tools. The document provides an expert consensus on how to use commercially available physiology tools for these purposes. It has been reviewed and approved by the European Society of Cardiology (ESC) Clinical Guidelines Committee to ensure that no conflict exists with available guidelines. Many of the topics discussed herein pertain to the use of intracoronary pressure guidewires, which are the most widely used devices for physiological assessment in the cardiac catheterisation laboratory. Functional coronary angiography (FCA) tools, which merge angiographic information with fluid dynamic equations to deliver information equivalent to intracoronary pressure measurements and which constitute a valuable alternative to invasive tools in planning coronary revascularisation, are also discussed. A detailed and updated revision of the use of NHPR and FFR in different clinical scenarios is provided in Supplementary Appendix 1.
From confirming the indication to planning and guiding PCI
The limitations of coronary angiography in characterising the flow-limiting effect of coronary stenoses can be overcome by non-invasive and/or adjunctive invasive physiological assessments of coronary artery disease (CAD), the value of which are supported by evidence-based guideline recommendations5. When prior evidence of myocardial ischaemia is not available, FFR or instantaneous wave-free ratio (iFR) are recommended by the guidelines to assess the haemodynamic relevance of intermediate-grade coronary stenoses. FFR can also be considered in patients with multivessel disease (MVD) undergoing PCI5.
Over the last decade, criticism concerning the use of angiography alone to guide revascularisation decisions has been extended to the decision of when an optimal functional result of the intervention has been achieved. Such criticism is supported by suboptimal functional results identified in vessels despite a satisfactory angiographic PCI result23. Prior and recent observations have shown that such functionally suboptimal PCI results carry prognostic relevance (Supplementary Table 1)4678910111213141516171819202122232425. Physiological guidance could contribute to improved PCI results in at least 3 ways: 1) improving preprocedural planning and simulation, 2) improving intraprocedural precision of PCI in addressing flow-limiting disease, and 3) guiding procedural optimisation of suboptimal PCI results (Central illustration).
Despite the limited number of available studies in this field, there are now rational grounds to consider using physiological tools to plan effective PCI strategies to ideally remove all flow-limiting stenoses, to verify that functionally successful PCI has been achieved, and to contribute to procedural optimisation. Table 1 provides key information on the physiological guidance of PCI.
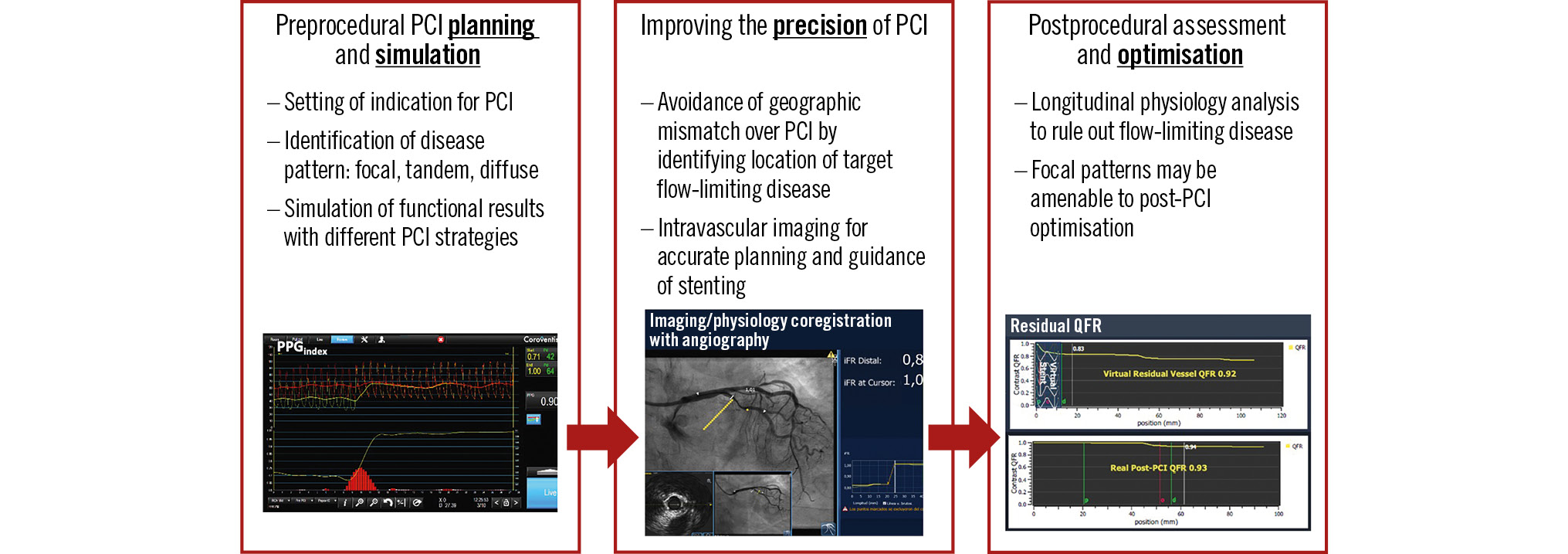
Central illustration. Applications of physiology in planning and guiding PCI procedures. iFR: instantaneous wave-free ratio; PCI: percutaneous coronary intervention; PPG: pullback pressure gradient; QFR: quantitative flow ratio
Table 1. Objectives of physiological planning and guidance of PCI
| Pre-PCI physiology assessment | ||
|---|---|---|
| Objective | Technique | Benefit |
| Determining the indication for PCI | Pressure wire or FCA to identify the presence of flow-limiting disease | – Management of significant stenoses may confer prognostic or symptom benefit– Deferral of non-flow-limiting disease avoids unnecessary PCI |
| Identifying patterns of flow-limiting disease | Longitudinal physiological vessel analysis to identify focal, tandem, and diffuse patterns of flow-limiting disease | – Assists in gauging effectiveness of PCI– Aids with planning the length of stent and/or number of stents– Reconsider PCI when a suboptimal result is anticipated |
| Simulate impact of stenting in specific locations | NHPR and imaging-based functional assessments to simulate relief of stenosis virtually | – Plan effectiveness of PCI before stent deployment– Allows for several simulations prior to PCI |
| Facilitating precision stent deployment | Correlating physiology and angiography using either coregistration technologies or visual assessment, use of concomitant intracoronary imaging | – Avoidance of geographical miss during stenting, missing flow-limiting lesions– Accurate sizing of balloons and stents |
| Post-PCI physiology assessment | ||
| Objective | Technique | Benefit |
| Ensuring an optimal functional result of PCI | Physiological measurements in PCI target vessel after satisfactory angiographic result, jailed side-branch interrogation | – Early identification of residual flow-limiting disease in the PCI target vessel after the intervention |
| Identifying potential targets of functional optimisation of PCI | Post-PCI longitudinal vessel analysis | – Establishing the cause of suboptimal functional PCI results– Establishing the feasibility and mode of PCI optimisation |
| Assessing the impact of PCI optimisation | Repeat physiological measurements after physiology-based optimisation | – Residual disease not amenable to PCI may identify need for directed medical therapy or surgical revascularisation |
| FCA: functional coronary angiography; NHPR: non-hyperaemic pressure ratio; PCI: percutaneous coronary intervention | ||
Technological developments in applied coronary physiology in PCI
HYPERAEMIC AND NON-HYPERAEMIC PRESSURE INDICES
FFR is the most widely used intracoronary tool to define flow-limiting epicardial stenoses, with randomised clinical trials and observational studies supporting deferral262728 or performance2629 of revascularisation based on FFR values, predominately in patients with CCS. Contrast-based FFR, which relies upon submaximal myocardial hyperaemia induced by iodinated contrast administration, has been proposed as an adenosine-free alternative to FFR30. NHPR is increasingly utilised in clinical practice owing to a more favourable side-effect profile, reduced cost and procedure time, and ease of use compared with pharmacological hyperaemia. This facilitates multiple intraprocedural measurements either in different vessels or at different stages of the PCI procedure. Guidance by iFR, a widely utilised NHPR, was non-inferior to FFR guidance of revascularisation for major adverse cardiac events (MACE) at 1-year3132 and 5-year follow-ups33. Other diastolic NHPR correlate closely with the iFR, with a similar ischaemic cut-off of ≤0.8934. All NHPR have demonstrated significant association with the risk of 2-year vessel-oriented composite endpoints35.
In the extended scenario of planning and guiding PCI, the use of pressure guidewires can be subject to specific caveats related to physiological assessment in complex clinical and anatomical scenarios (Table 2). Additional information detailing potential pitfalls in physiological assessment and considerations regarding the use of guide catheters, guide catheter extensions and microcatheters is provided in Supplementary Appendix 1.
Table 2. Challenges and pitfalls related to physiological assessment
| Patient factors | ||
|---|---|---|
| Challenges and pitfalls | Comment | Solution |
| Presence of significant ostial CAD | Identified with ventricularisation or “damping” pressure waveform on engagement, not assessable with some FCA modalities | Reposition guide catheter to a more proximal position, ensure coaxiality, consider downsizing guide catheter, use an additional guidewire or septal wire to control catheter tip position |
| Haemodynamic crosstalk in presence of tandem lesions | Sequential lesions along a vessel invariably physiologically influence the other lesions in the vessel | Consider simulated planning prior to undertaking PCI on 1 or 2 stenoses based on longitudinal vessel analysis obtained with functional coronary angiography, NHPR, or FFR |
| Procedural factors | ||
| Challenges and pitfalls | Comment | Solution |
| Pressure wire drift | Offset of pressure reading during the procedure | Repeat procedure, use drift correction/re-normalise, or use image-based technique |
| Pseudostenosis caused by intracoronary wires | Straightening of tortuous vessels by intra-coronary wire may generate introsusceptions with haemodynamic relevance | Consider FCA as an alternative to intracoronary pressure guidewires |
| Ventricularisation/damping of aortic pressure | Guiding catheter engaged abutting the vascular lumen or in significant ostial lesions | Reposition guide catheter, ensure coaxiality, consider downsizing guide catheter, equalisation of pressure guidewire is advised |
| Use of guide catheter with side holes | False estimate of Pa at the tip of the guiding catheter | Advise not to use catheter with side holes for intracoronary assessment. If needed for clinical reasons, ensure catheter disengagement and use of IV adenosine. |
| Use of guide catheter extensions | May cause ventricularisation of Pa, if introduced after pressure equalisation, may modify Pa and affect measurements | Perform pressure equalisation with extension within guide catheter, withdraw guide extension at the time of physiological assessment |
| Use of coronary microcatheters | If used after pressure equalisation, may modify Pa and affect measurements | Perform pressure equalisation with microcatheter within guide catheter before measurements |
| Wire whipping | Pressure wire signal degrading through contact with vascular lumen | Repositioning of the pressure wire often resolves this issue |
| Transient microvascular dysfunction associated with the procedure | Rotational atherectomy and PCI tools generating microparticles may cause transient microvascular dysfunction, affecting pressure indices | Consider FCA as an alternative to intracoronary assessment if transient microvascular dysfunction is assumed or suspected |
| FFR measurement in region without stable hyperaemia | Whilst the software reports the lowest physiological reading, these might not be in stable hyperaemia and therefore overestimate lesion severity | Measure FFR in stable hyperaemia, and if needed adjust measurement points manually |
| CAD: coronary artery disease; IV: intravenous; FCA: functional coronary angiography; FFR: fractional flow reserve; NHPR: non-hyperaemic pressure ratio; Pa: aortic pressure; PCI: percutaneous coronary intervention | ||
FUNCTIONAL CORONARY ANGIOGRAPHY
The success of FFR and NHPR as indices to estimate functional stenosis severity has led to the development of new FCA technologies. These can derive similar information from an invasive coronary angiogram or non-invasive computed tomography coronary angiography (CTCA), with both approaches demonstrating good correlation with wire-based FFR. Computed tomography-derived FFR (FFRCT) is obtained by merging a three-dimensional (3D) reconstruction of vessels from CTCA with computational fluid dynamics36, thereby providing a reliable estimate of invasive FFR37. Similarly, several FFR-equivalent measurements can also be derived from invasive coronary angiograms38394041; these are supported by prospective validation studies. Quantitative flow ratio (QFR) is the only angiography-based physiological index that has been prospectively validated and has already been demonstrated to be associated with improved clinical outcomes when used to decide upon coronary revascularisation, compared with conventional angiography42. A distinct advantage of all FCA modalities is that they provide longitudinal vessel analysis, allowing accurate length measurements and localisation of flow-limiting disease to vessel anatomy. Detailed information on potential limitations of imaging-based functional coronary analysis is provided in Supplementary Table 2.
LONGITUDINAL PHYSIOLOGICAL VESSEL ANALYSIS
Customarily, indices of coronary physiology have been reported as a single value in the distal segment of the interrogated coronary artery, reflecting cumulative pressure losses along the entire length of the epicardial vessel. Longitudinal assessment, utilising a pressure wire pullback, adds an additional dimension to the physiological analysis. Pullback manoeuvres can be performed manually43 with either FFR or NHPR4445. Accurate length measurements to the order of millimetres can be obtained by using either motorised pullback46 or dedicated software that tracks the radiopaque wire tip during pullback47. Dedicated software is available to provide stable FFR, iFR, and resting full-cycle ratio (RFR) pullback curves, free of fluctuations due to the Venturi effect, where a drop in intracoronary pressure is seen as the sensor crosses the stenosis (Supplementary Figure 1).
Prior to PCI, longitudinal vessel assessment informs on the distribution of pressure losses along the epicardial vessel and can differentiate focal and/or diffuse patterns of flow-limiting disease (Figure 1). Longitudinal vessel analysis allows for the identification of focal and diffuse patterns of flow-limiting disease, either subjectively by visual inspection of the pullback curve or objectively by using the pressure pullback gradient index (PPGindex)45, which can be performed manually with high inter- and intraoperator reproducibility48. Differentiation of diffuse from focal disease is also feasible with an automated algorithm that analyses the instantaneous FFR gradient per time unit, the dFFR(t)/dt index49. Both the PPGindex and dFFR(t)/dt can be derived from QFR, and their prognostic relevance have been documented with post-PCI clinical outcomes4950. The criteria used to define focal, tandem, and diffuse patterns, as well as supporting studies, can be found in Table 3 and Supplementary Table 3, respectively2345505152535455565758596061. The current definitions of focal and diffuse disease are largely qualitative, and research to formally define these lesion characteristics is awaited. The ability to discriminate between patterns of CAD carries immediate and relevant clinical implications; a focal pattern is associated with an optimal physiological result after PCI, with consequent good prognosis and relief of angina. On the contrary, a diffuse pattern of disease is associated with suboptimal post-PCI results and prognosis and more residual anginal symptoms62.
After PCI, longitudinal physiological vessel interrogation may identify residual focal pressure gradients inside or outside the stent which might be amenable to additional stent post-dilatation or PCI2. Alternatively, a diffuse pattern of residual disease after PCI may discourage operators from further vessel instrumentation.
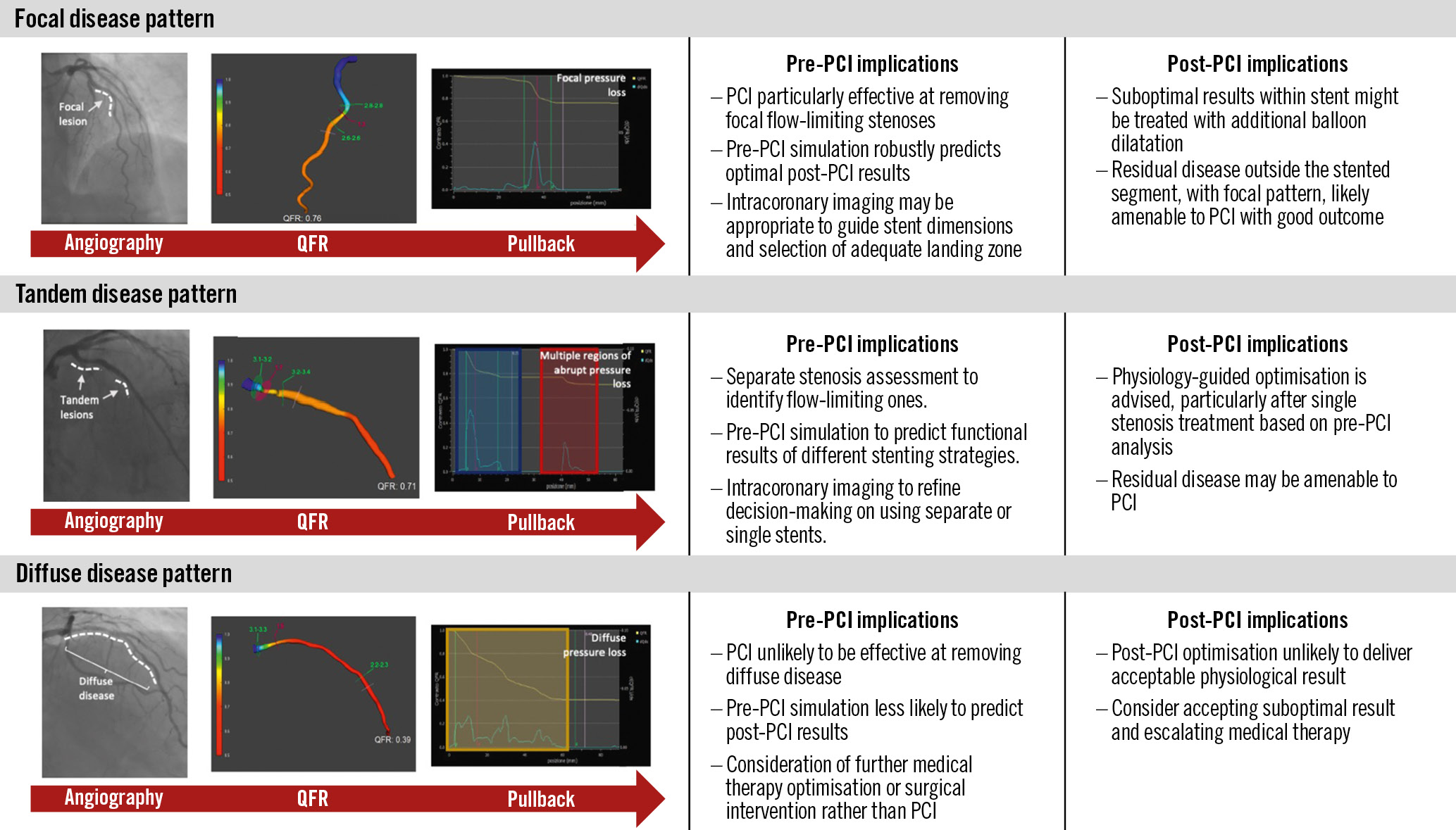
Figure 1. Patterns of flow-limiting disease identified in longitudinal vessel analysis before and after PCI. PCI: percutaneous coronary intervention; QFR: quantitative flow ratio
Table 3. Criteria used to define focal, tandem and diffuse patterns of obstructive coronary disease
| Pre-PCI physiology assessments | |||
|---|---|---|---|
| Pattern | FFR | NHPR | FCA |
| Focal | FFR ≤0.80 at distal segment, abrupt single point of pressure loss. | iFR/RFR/DFR ≤0.89 at distal segment, with abrupt single point of ≥0.03 index units over ≤15 mm segment. | QFR/vFFR/FFRCT ≤0.80 at distal segment, with single region of pressure loss of >0.05 in <10 mm segment. |
| Tandem | FFR ≤0.80 at distal segment, presence of ≥2 abrupt change of FFR values. | iFR/RFR/DFR ≤0.89, at distal segment, with presence of ≥2 abrupt changes of index values. | QFR/vFFR/FFRCT ≤0.80 at distal vessel with presence of ≥2 abrupt changes of pressure loss. |
| Diffuse* | FFR ≤0.80 at distal segment with progressive and linear loss in FFR values over length of vessel. | iFR/RFR/DFR ≤0.89 at distal segment, with progressive and linear loss in pressure over length of vessel during iFR pullback. | Progressive and linear loss in QFR/vFFR/FFRCT values over length of vessel during longitudinal analysis. |
| Post-PCI physiology assessments | |||
| Pattern | FFR | NHPR | FCA |
| Focal | FFR with abrupt pressure loss at the stented site or elsewhere within the treated vessel. | Abrupt drop of iFR/RFR/DFR values at the stented site or elsewhere within the treated vessel. | Abrupt drop of QFR/vFFR values at the stented site or elsewhere within the treated vessel. |
| Tandem | Abrupt drop of FFR values at the level of an untreated tandem stenosis. | Abrupt drop of iFR/RFR/DFR values at the level of an untreated tandem stenosis. | Abrupt drop of QFR/vFFR values at the level of an untreated tandem stenosis. |
| Diffuse* | Progressive and linear loss in FFR values over length of stented vessel during pullback. | Progressive and linear loss in iFR/RFR/DFR values over length of stented vessel during pullback. | Progressive and linear loss in QFR/vFFR values over length of stented vessel. |
| *These criteria are clinical consensus statements and further validation is warranted. DFR: diastolic hyperaemia-free ratio; FCA: functional coronary angiography; FFR: fractional flow reserve; FFRCT: computed tomography-derived FFR; iFR: instantaneous wave-free ratio; NHPR: non-hyperaemic pressure ratio; PCI: percutaneous coronary intervention; QFR: quantitative flow ratio; RFR: resting full-cycle ratio; vFFR: virtual FFR | |||
COREGISTRATION WITH ANGIOGRAPHY
Merging longitudinal vessel physiology with the coronary angiogram allows for accurate localisation of flow-limiting atherosclerotic disease and facilitates procedural planning47. The coregistered map provides signposts with a clear distribution of regions of pressure loss, enabling optimal localisation of specific target lesions that might benefit from PCI, and the implementation of length measurements (Supplementary Figure 2). This technology can be used in conjunction with iFR and is particularly useful when forming strategies for intervention on tandem lesions and interrogating areas of diffuse disease.
SIMULATION OF FUNCTIONAL PCI RESULTS
Despite consistent evidence showing that suboptimal values of physiological indices after PCI are associated with poorer outcomes, there is a relative paucity of evidence regarding whether they can be used to guide PCI optimisation and, ultimately, to improve patient outcomes. Since the aim of PCI is the elimination of ischaemia-generating lesions, predicting the haemodynamic results of a given strategy before embarking on stenting appears to be a rational approach to avoid suboptimal results of the intervention. The concept of in silico simulation of PCI to predict functional results of an intervention is appealing, as it allows for both procedural planning and modelling of post-PCI physiology prior to undertaking the procedure.
There are different approaches to simulate functional PCI results from baseline longitudinal vessel analysis (Figure 2). A simple mathematical approach to estimate the impact of PCI based on pullback curves can be followed: predicted NHPR (NHPRpred)=pre-PCI NHPR (lowest value) + âintention-to-treat NHPR gradient(s). Pioneering studies with iFR pullback have demonstrated its ability to predict post-PCI results63. Subsequently, this has also been confirmed for other NHPR such as the RFR and diastolic pressure ratio61. Subtraction of the flow-limiting effect of one or more coronary stenoses can be readily performed with current software versions of iFR coregistration with angiography31. From the perspective of FCA, a dedicated virtual stenting tool has been developed for FFRCT64. The FFRCT Planner (HeartFlow) has been shown to be accurate and precise at predicting post-PCI FFR65. Analysis with QFR and virtual FFR (vFFR) provides an estimate of post-PCI values after treatment of a given stenosis (residual QFR and vFFR), which has been shown to predict post-PCI FFR256667. Figure 3 shows a pullback curve analysis performed with iFR and coregistration, including simulated stenting at a specific location.
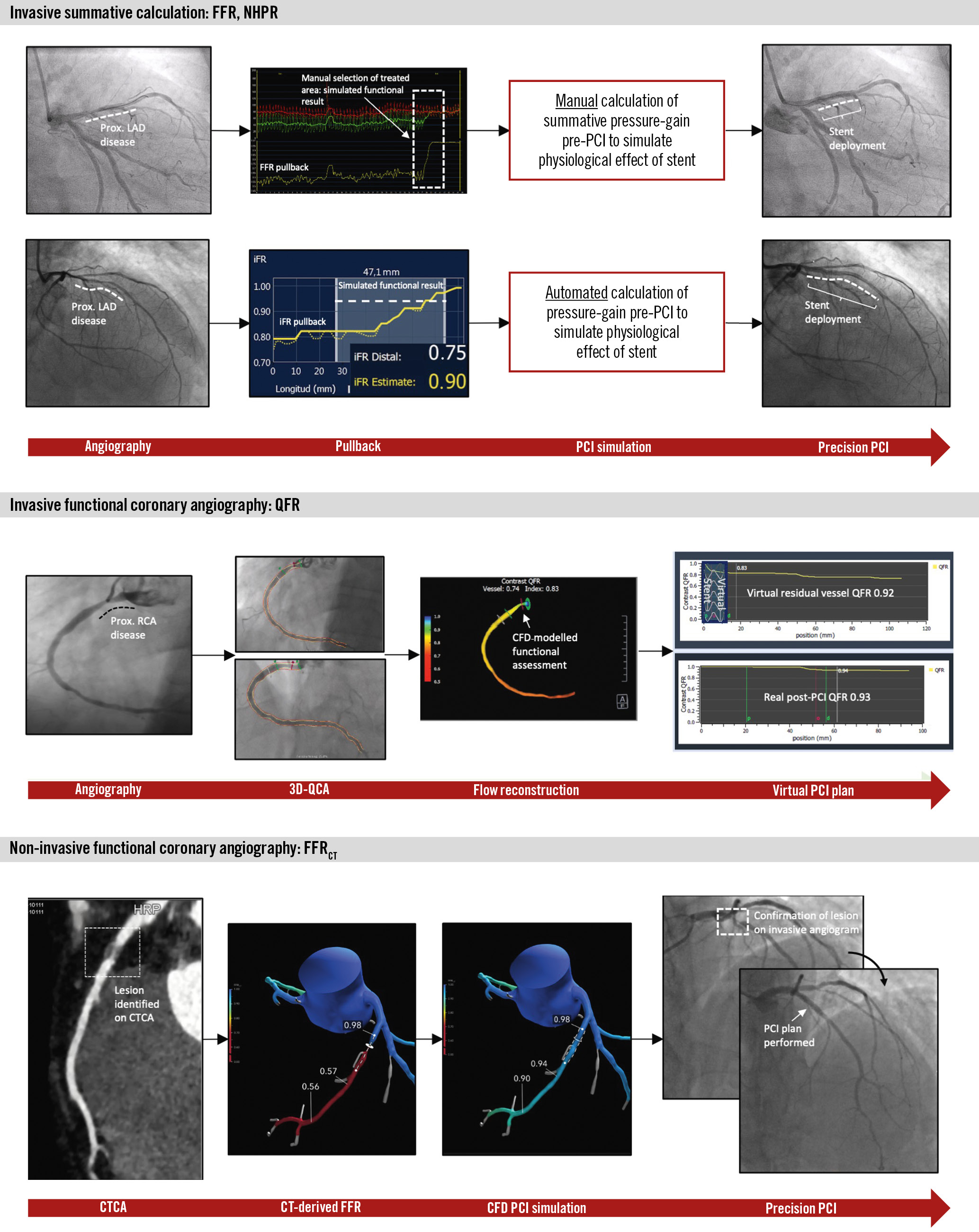
Figure 2. Approaches for simulating functional PCI results based on longitudinal vessel analysis. 3D-QCA: three-dimensional quantitative coronary angiography; CFD: computational fluid dynamics; CTCA: computed tomography coronary angiography; FFR: fractional flow reserve; FFRCT: computed tomography-derived FFR; iFR: instantaneous wave-free ratio; LAD: left anterior descending artery; NHPR: non-hyperaemic pressure ratio; PCI: percutaneous coronary intervention; QFR: quantitative flow ratio; RCA: right coronary artery
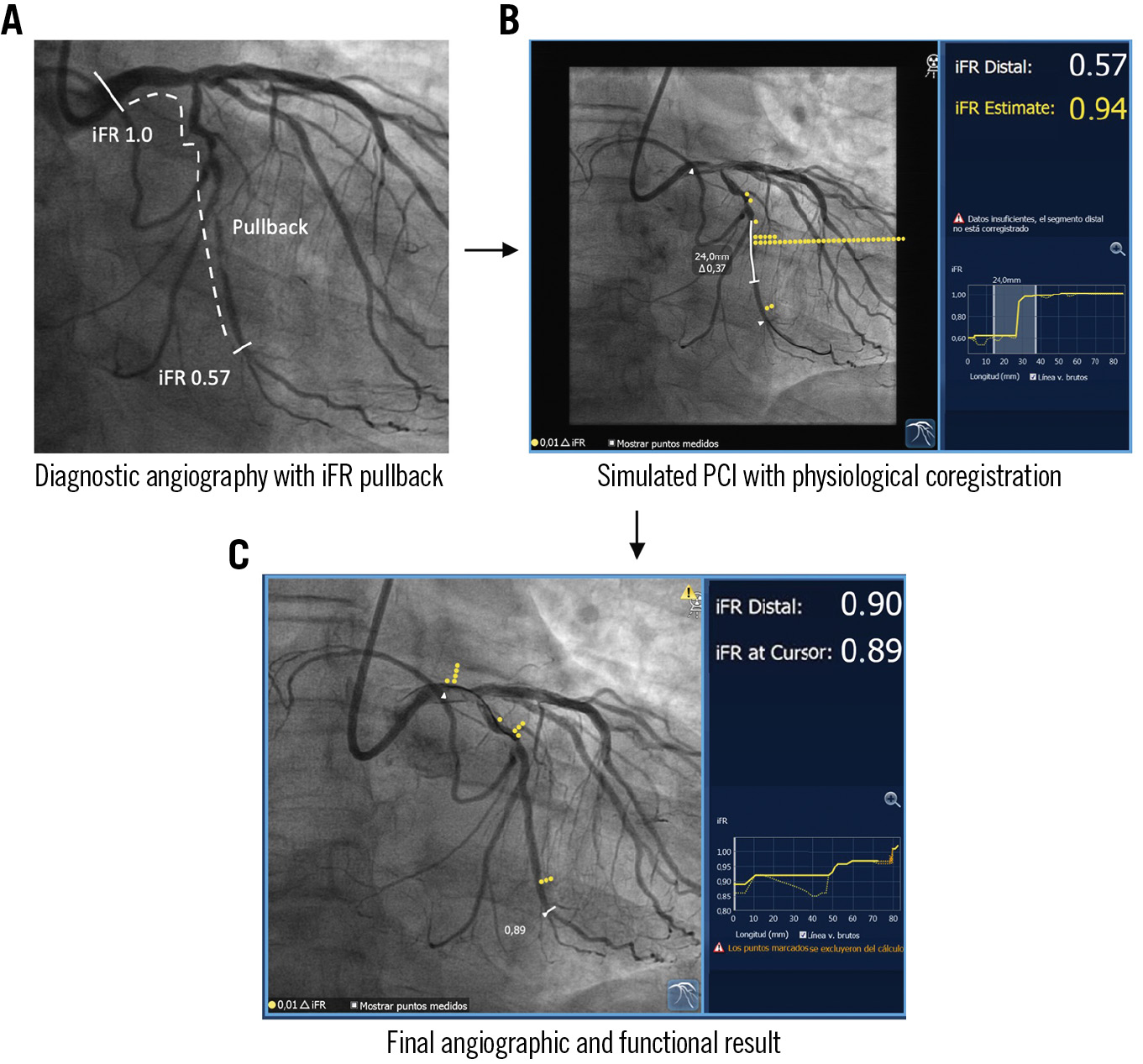
Figure 3. Simulation of functional PCI results using coregistration of physiology and the coronary angiogram. A) Angiography of the left circumflex artery interrogated with longitudinal physiological assessment. B) The presence of flow-limiting disease in the vessel is demonstrated by a distal iFR of 0.57. Longitudinal vessel analysis revealed a focal disease phenotype. iFR coregistration with angiography allows accurate localisation of flow-limiting disease in the coronary anatomy. The PCI simulation suggests adequate functional results of the intervention, with a predicted post-PCI iFR of 0.94. C) Final co-registered angiographic-physiology result shown, with final iFR of 0.90. iFR: instantaneous wave-free ratio; PCI: percutaneous coronary intervention
CORONARY PRESSURE WIRES AND MEASURING TOOLS
Continued improvement in pressure guidewire technology has facilitated the accurate interrogation and measurement of complex coronary anatomies. Contemporary guidewire-based pressure indices are fitted with sensors that use either electrical or fibre-optic signal transmission. Advances in manufacturing processes have resulted in an improved accuracy and stability of the sensor, thus, minimising the drift phenomenon with workhorse guidewire-like characteristics, resulting in optimised torque control and manoeuvrability. The latter is critical for the safe wiring of the target vessel prior to the measurement of pressure loss and PCI. Microcatheter-based pressure measurement technology requires a 0.014” guidewire, according to anatomy or operator preference, but requires a 0.020” shaft profile for the microcatheter, which may interfere with measurements, especially in small vessels or severe stenoses, and can be difficult to navigate around tight angulations. The currently available and pending devices for invasive functional assessments are shown in Supplementary Table 4.
Physiological assessment of post-PCI results
Post-PCI intracoronary pressure measurements can identify residual flow-limiting disease, differentiate residual focal lesions from diffuse disease and provide prognostic information. Whilst a higher post-PCI FFR is associated with a lower incidence of adverse clinical outcomes, the cut-off point for an optimal PCI result is still debated68. A post-PCI FFR ≥0.90 has been associated with a significantly lower risk of repeat PCI and MACE in a systematic review of 7,470 patients69. The most robust recent data, obtained in a patient-level meta-analysis of 5,869 vessels treated with modern drug-eluting stents, reported optimal post-PCI FFR cut-off values of 0.86 for target vessel failure and 0.80 for the composite of cardiac death or target vessel myocardial infarction70. For NHPR, a post-PCI iFR ≥0.95 was associated with improved patient outcomes in the DEFINE PCI study3. An optimal cut-off for post-PCI distal coronary pressure/aortic pressure (Pd/Pa) ratio of>0.96 has also been proposed23.
Contemporary reports indicate that post-PCI physiology results might remain below the clinical revascularisation thresholds of ≤0.80 for FFR and <0.90 for NHPR in approximately 24-36% of cases2371. Potential mechanisms of suboptimal post-PCI physiology results are outlined in Figure 4. Additional interventions guided by post-PCI coronary physiology assessments can improve the final result21071. Caution should be exerted to avoid overtreatment, for example when the identified cause of suboptimal functional results entails treatment of long coronary segments or aggressive lesion manipulation. Intracoronary imaging may contribute to a better understanding of the cause of suboptimal functional results and to safer decision-making. Furthermore, it has to be kept in mind that cardiovascular risk may differ between patients despite having achieved an optimal functional result; this is largely dictated by the extension and characteristics of underlying atherosclerosis.
While functionally suboptimal PCI results are associated with adverse clinical outcomes and can be related to residual treatable disease, they may more frequently be an epiphenomenon of diffuse atherosclerosis that cannot be adequately addressed with revascularisation. This may explain why larger relative increases in FFR after stenting (typically achieved through treatment of focal, physiologically severe lesions) are associated with lower rates of target vessel failure, reduced incidence of angina and improved quality of life142172. As not all residual flow-limiting disease is amenable to treatment with PCI, achieving optimal post-PCI FFR or NHPR target values can be challenging, or frequently impossible, in many patients73. Beyond the pattern of coronary artery disease, there are additional anatomical factors that may contribute to lower pressure-based indices. Evolving evidence suggests that post-PCI FFR values are consistently lower in left anterior descending (LAD) versus non-LAD vessels and might require vessel-specific optimal thresholds, adding a further level of complexity222. The lower LAD values likely occur because of the interplay of several factors, including the higher prevalence of intramuscular coronary tracts observed in the LAD (Supplementary Figure 3), the impact of hydrostatic effects relating to coronary anatomy and the resultant height of the pressure wire sensor above or below the aortic pressure transducer (typically causing a hydrostatic offset of around 3.6 mmHg)74, and higher coronary flow rates due to the larger myocardial mass subtended by the LAD (high flow across long segments of mild residual diffuse atheroma can generate an appreciable pressure gradient). Based on this, normal FFR values for the LAD have recently been shown to be 0.92, compared to 0.96 in non-LAD vessels75. Finally, in all anatomical locations, when NHPR are used, residual reactive hyperaemia induced by intervention-related vessel manipulation may render false positive measurements if performed immediately after stent implantation or contrast/saline injections.
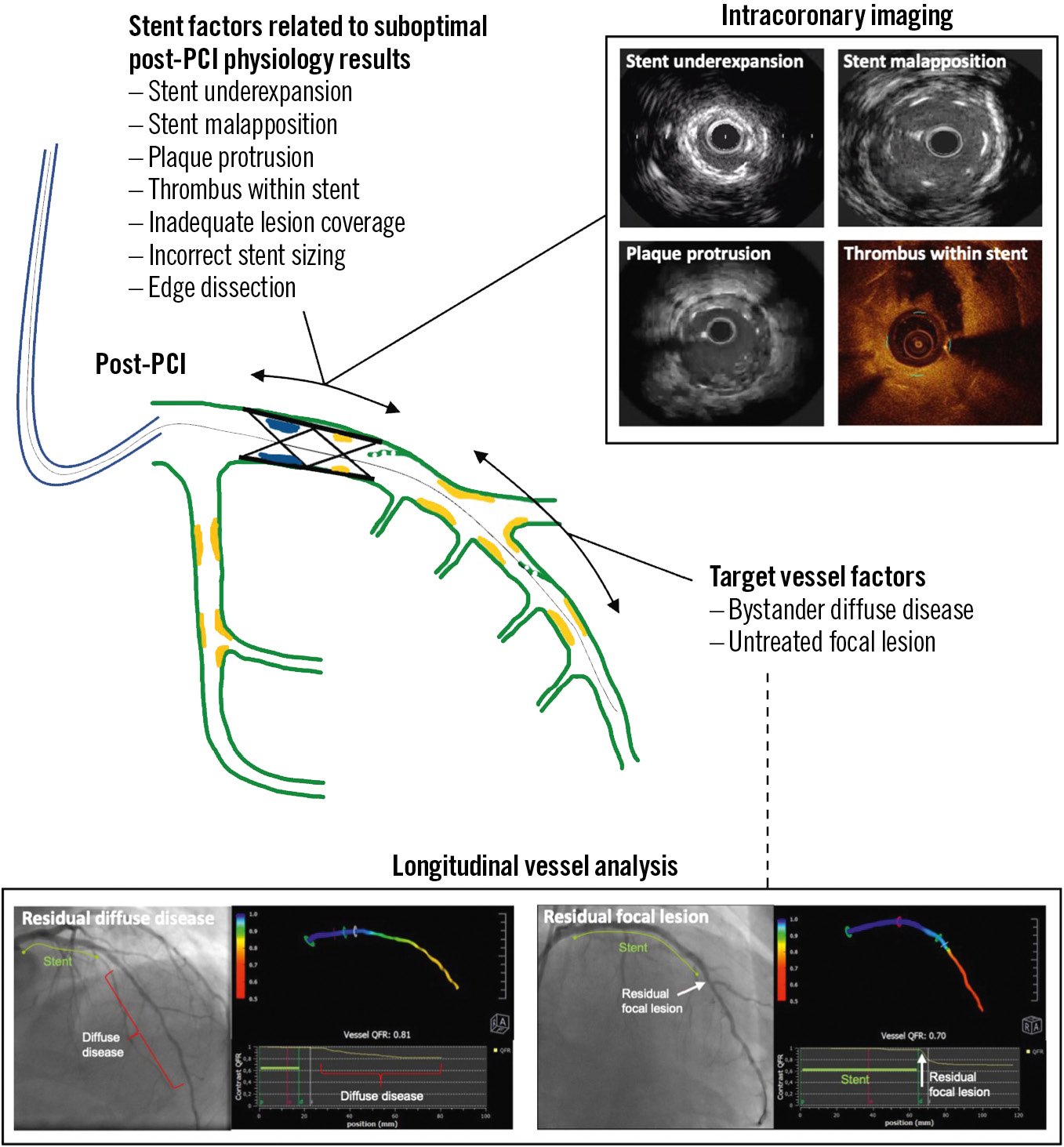
Figure 4. Mechanisms of suboptimal post-PCI physiological results. PCI: percutaneous coronary intervention; QFR: quantitative flow ratio
Complementary role of combined imaging and physiology in PCI planning and guidance
Whilst the objective of PCI is the removal of flow-limiting disease, long-term results of interventions are strongly influenced by procedural factors that are better addressed with intracoronary imaging techniques, such as intravascular ultrasound (IVUS) and optical coherence tomography (OCT) − both of which provide detail on CAD patterns and guidance for PCI. A detailed description of these techniques and on their use in planning, guiding and optimising PCI is available elsewhere76. The results of comprehensive vessel imaging post-PCI compared with FFR and its effect on long-term outcomes have been reported77. In addition, there is growing interest in whether the presence of vulnerable atheroma in non-flow-limiting lesions might be associated with a higher risk of future events78.
Merging information based on the presence and location of flow-limiting stenoses provided by physiology, with information on plaque characteristics and distribution, and vessel dimensions derived from imaging might aid in the selection of PCI devices and strategies in terms of lesion preparation, plaque-free landing zone for PCI, and the selection of stent diameter and length, all important aspects which ultimately improve long-term procedural results. Triregistration, with physiology and intracoronary imaging in conjunction with angiography, is currently available for IVUS and iFR. Coregistration with angiography is also feasible with OCT. Intracoronary imaging-derived physiology, such as OCT-derived FFR, currently lacks validation and is not available for clinical use.
In addition, there is extensive evidence suggesting that PCI optimisation with intravascular imaging improves long-term procedural outcomes276798081828384. This benefit is independent of the modest increase in FFR values noted in image-based PCI optimisation studies798586. The synergistic use of intracoronary imaging and post-PCI longitudinal vessel pullback can be used to investigate causes of focal pressure loss after PCI and assist with decisions on how they could be rectified86. Imaging may also highlight the existence of high-risk morphological features of coronary disease − both at baseline and post-PCI − with prognostic implications, even when these are not ischaemia-generating87, such as in instances of malapposition or significant edge dissection.
Integrating available tools and knowledge into algorithms for physiology-based planning and optimisation
The points discussed thus far can be tentatively integrated into algorithms for PCI planning and post-PCI evaluation. It is suggested that a comprehensive virtual analysis of the target vessel be performed prior to PCI, to support the indication for PCI and to assist in procedural planning and strategising, with the support of simulation if available. Following PCI, the result should be interrogated using a combination of physiology and/or imaging techniques, with post-PCI optimisation carried out when residual focal or tandem pressure loss remains. Suggested algorithms for comprehensive lesion assessment using pre- and post-PCI physiology are shown in Figure 5 and Figure 6, respectively.
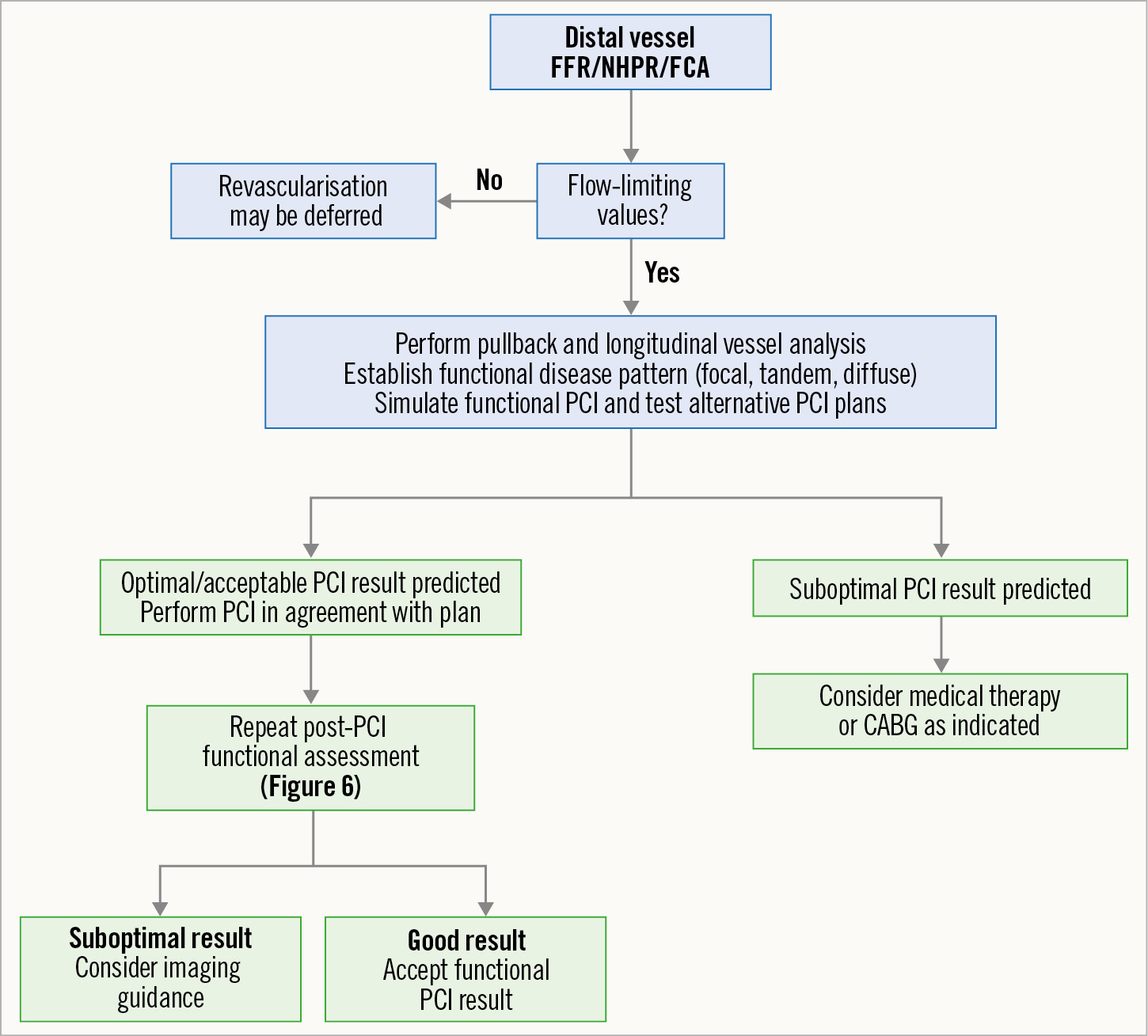
Figure 5. Proposed algorithm for pre-PCI physiology assessment and planning of PCI based on physiological interrogation. CABG: coronary artery bypass graft; FCA: functional coronary angiography; FFR: fractional flow reserve; NHPR: non-hyperaemic pressure ratio; PCI: percutaneous coronary intervention
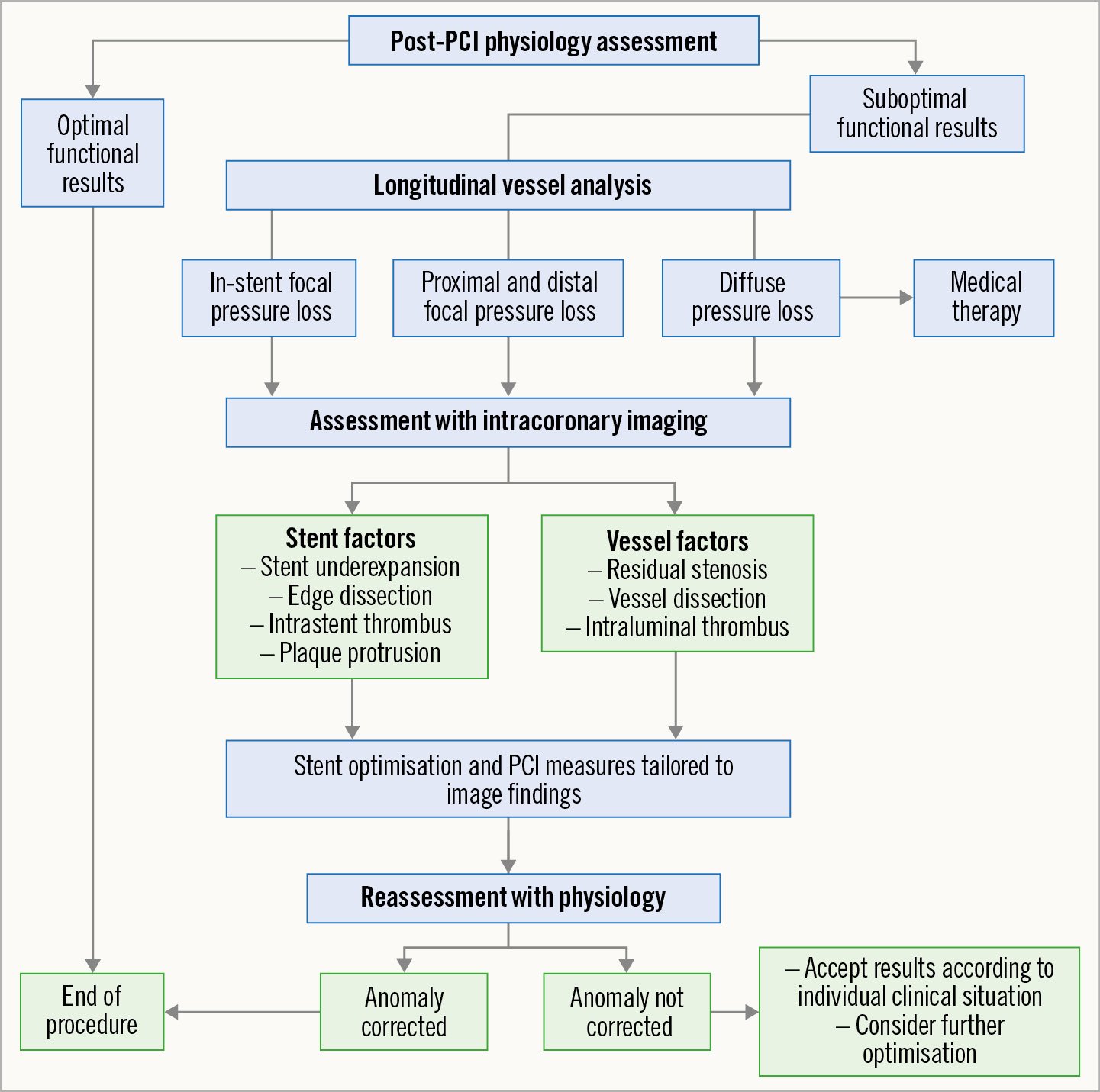
Figure 6. Proposed algorithm for postprocedural assessment and optimisation of functional PCI results based on physiological interrogation. PCI: percutaneous coronary intervention
Applied coronary physiology in specific PCI scenarios
In addition to the general principles discussed above, the use of physiology to plan and guide PCI in specific scenarios is deserving of a separate discussion. Supplementary Appendix 2 outlines applied coronary physiology for the following contexts: multivessel coronary artery disease, vessels with tandem lesions or diffusely diseased vessels, lesions in vessels providing collaterals to chronically occluded arteries, patients with acute coronary syndromes, bifurcation lesions and jailed side branches, left main stem stenosis, coronary lesions in patients with aortic stenosis, native vessel or surgical graft stenosis in patients with prior coronary artery bypass graft, and in vessels with myocardial bridge − all in greater detail.
Future directions and outlook
The use of physiology in procedural planning and simulation of PCI is an area of growing interest, though, as yet, supported only by a relatively small evidence base. Upcoming trials in the field will provide valuable evidence on the use of invasive physiology and FCA for this purpose. Whilst it is generally accepted that PCI is an appropriate therapy for patients with focal CAD, the optimal treatment strategy for patients with haemodynamically significant lesions in the presence of diffuse disease is a subject of ongoing investigation.
Prospective studies are also being conducted to assess the use of coregistration technology on stent deployment and patient outcomes. The DEFINE GPS (Distal Evaluation of Functional Performance With Intravascular Sensors to Assess the Narrowing Effect: Guided Physiologic Stenting; ClinicalTrials.gov: NCT04451044) and iLARDI (Usefulness of the Use of Co-registration Strategy With iFR in Long and/or Diffuse Coronary Lesions; ClincalTrials.gov: NCT04283734) investigators aim to assess the impact of iFR coregistration (SyncVision; Philips) on guiding PCI and influencing the numbers and lengths of implanted stents, and in DEFINE GPS, whether this might influence the rate of MACE. The PPG Global Registry involves a prospective evaluation of the impact of the PPG index on clinical decision-making for PCI (or not) and related outcomes (ClinicalTrials.gov: NCT04789317). Future studies should better define the role of microvascular dysfunction in relation to PCI outcomes. Collectively, these studies will add significant value in objectively assessing the utility of pre-PCI simulation and planning using physiological indices.
Conclusions
The raison d’être of PCI is to abolish flow-limiting stenoses and ischaemia in order to improve patient symptoms and/or prognosis. In conjunction with angiographic findings, physiology determined by FFR, NHPR and FCA assists in weighing important decisions in procedural planning. Emerging studies hint at the application of in silico PCI simulation prior to intervening on a patient. This offers a novel perspective on planning the effectiveness of an intervention and could enable the adoption of revascularisation strategies associated with the highest physiological gain. Furthermore, identifying patients with little physiological gain after PCI, despite optimisation, is highly important, as they are at high risk of premature stent failure.
Physiology in the post-PCI setting facilitates procedural optimisation through descriptive longitudinal vessel analyses of stented segments, offering clarity on the presence of residual flow-limiting disease. These technologies, in combination with the ability to coregister anatomy with physiology and intravascular imaging, which allows for the identification of different disease phenotypes, have revolutionised PCI in the modern era. Clinical trials assessing the effectiveness of these tools on clinical outcomes are eagerly awaited.
Acknowledgements
We are indebted to Silvio Capuano for their technical support in preparing the supplementary tables for this document.
Conflict of interest statement
J. Escaned reports speaker or advisory board member fees from Abbott, Boston Scientific, Medis, and Philips. C. Berry is employed by the University of Glasgow, which holds consultancy and research agreements for his work with Abbott Vascular, AstraZeneca, Boehringer Ingelheim, Causeway Therapeutics, Coroventis, Genentech, GSK, HeartFlow, Menarini, Neovasc, Novartis, Siemens Healthcare, and Valo Health. B. De Bruyne reports receiving consultancy fees from Boston Scientific and Abbott Vascular; research grants from Coroventis Research, Pie Medical Imaging, CathWorks, Boston Scientific, Siemens, HeartFlow, and Abbott Vascular; and owning equity in Siemens, GE HealthCare, Philips, HeartFlow, Edwards Lifesciences, Bayer, Sanofi, and Celyad. C. Collet reports receiving research grants from Biosensor, Coroventis Research, Medis Medical Imaging, Pie Medical Imaging, CathWorks, Boston Scientific, Siemens, HeartFlow, and Abbott Vascular; and consultancy fees from HeartFlow, OpSens, Abbott Vascular, and Philips/Volcano. J.M. Lee received institutional research grants from Abbott Vascular, Boston Scientific, Terumo Corporation, Philips/Volcano, Medis Medical Imaging, and Zoll Medical. Y Appelman received speaker fees from Abbott Vascular. Si. Biscaglia received unrestricted research grants and speaker's fees from SMT, Medis, Abbott, and Insight Lifetech. P.P. Buszman is employed by the American Heart of Poland, which holds research agreements with Meril Life, and received speakers’ honoraria from Novartis. G. Campo received institutional research grants from Medis, GE HealthCare, Siemens Healthcare, Abbott Vascular, Sahajanand Medical Technologies, and Insight Lifetech. D. Collison has received consultancy and speaker fees from Abbott. A. Jeremias reports consultancy fees from Philips/Volcano, Abbott Vascular, Boston Scientific, and ACIST Medical Systems. Z. Piróth has received speakers’ fees from Abbott, Boston Scientific, and Opsens. L. Raposo has received honoraria and research grants from Philips/Volcano, St. Jude Medical (now Abbott Vascular) and HeartFlow, as well as consultancy fees from Boston Scientific. T. Rudolph received speaker honoraria from Abbott Vascular, Philips, Neovasc, AstraZeneca, Bayer, Pfizer, Philips/Volcano, Zoll Medical, and RainMed. G. Sarno has received a research grant from Boston Scientific (to the institution); and personal fees from Abbott Vascular, Boston Scientific, and Pfizer/BMS. D. Dudek reports participation in company-sponsored speaker’s bureaus for Abbott, Boston Scientific, Bracco, Philips, and Siemens Healthcare; and unrestricted grants from Abbott, Boston Scientific, Bracco, Philips, and Siemens Healthcare. The other authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.

