
Abstract
Aims: The APPOSITION III registry evaluated the feasibility and performance of the STENTYS self-apposing stent in an ST-segment elevation myocardial infarction (STEMI) population. This novel self-apposing stent device lowers stent strut malapposition rates and therefore carries the potential to prevent stent undersizing during primary percutaneous coronary intervention (PCI) in STEMI patients. To date, no long-term data are available using this device in the setting of STEMI. We aimed to evaluate the long-term clinical outcomes of the APPOSITION III registry.
Methods and results: This was an international, prospective, multicentre post-marketing registry. The study population consisted of 965 STEMI patients. The primary endpoint, major adverse cardiac events (MACE), was defined as the composite of cardiac death, recurrent target vessel myocardial infarction (TV-MI), and clinically driven target lesion revascularisation (CD-TLR). At two years, MACE occurred in 11.2%, cardiac death occurred in 2.3%, TV-MI occurred in 2.3% and CD-TLR in 9.2% of patients. The two-year definite stent thrombosis (ST) rate was 3.3%. Incremental event rates between one- and two-year follow-up were 1.0% for TV-MI, 1.8% for CD-TLR, and 0.5% for definite ST. Post-dilation resulted in significantly reduced CD-TLR and ST rates at 30-day landmark analyses. Results were equivalent between the BMS and PES STENTYS subgroups.
Conclusions: This registry revealed low rates of adverse events at two-year follow-up, with an incremental ST rate as low as 0.5% in the second year, demonstrating that the self-apposing technique is feasible in STEMI patients on long-term follow-up while using post-dilatation.
Abbreviations
BMS: bare metal stent
ISA: incomplete strut apposition
MACE: major adverse cardiac events
MI: myocardial infarction
OCT: optical coherence tomography
PCI: percutaneous coronary intervention
PES: paclitaxel-eluting stent
STEMI: ST-segment elevation myocardial infarction
TIMI: Thrombolysis In Myocardial Infarction
TLR: target lesion revascularisation
TV-MI: target vessel myocardial infarction
TVR: target vessel revascularisation
Introduction
In the treatment of patients presenting with an ST-segment elevation myocardial infarction (STEMI), primary percutaneous coronary intervention (PCI) with balloon dilatation and stent placement has become the standard treatment strategy in current clinical practice1. Multiple factors contribute to improved clinical results: one of them is optimal stent selection and stent deployment during PCI, which is often more challenging in STEMI patients. Many advances have been made to improve coronary stent designs, including stent platform technologies. With the introduction of the nitinol STENTYS self-apposing stent (STENTYS S.A., Paris, France), several advantages are proposed which make this stent technology especially appealing for STEMI patients undergoing primary PCI. The nitinol platform has the ability to appose itself to the arterial wall in time, even after stent deployment. This accommodates vessel diameter changes due to the positive remodelling and vasodilatation of the vessel after the acute event. To evaluate this self-expanding stent technology in STEMI patients, the APPOSITION clinical trial programme is being conducted. The expansive property of the stent platform was observed on intravascular ultrasound after stent deployment, in APPOSITION I2. Compared to a conventional balloon-expandable stent, the STENTYS stent has shown superiority in strut malapposition on optical coherence tomography (OCT) in APPOSITION II3. Three days after primary PCI, OCT images revealed tissue resorption and vasorelaxation in all STEMI patients. These biological factors were associated with new appearance of incomplete strut apposition (ISA). Less ISA and an increased stent area were seen in the STENTYS group, demonstrating that this stent is able to adjust to the dynamic changes of the arterial wall in the first days after the acute event. These observations were extended in a STEMI trial with longer OCT follow-up, demonstrating that the early stent apposition in the STENTYS group is maintained up to four and nine months4. However, it is unknown whether safety and efficacy concerns regarding this device may be present beyond one year.
We therefore present the two-year follow-up data of the international, multicentre APPOSITION III registry (A Post-Market registry to assess the STENTYS self-exPanding COronary Stent In AcuTe MyocardIal InfarctiON) to investigate whether the use of the self-apposing stent technology translates into long-term favourable outcomes in STEMI patients.
Methods
STUDY DESIGN AND PATIENTS
The APPOSITION III registry’s design, methods and one-year results have been previously reported5. Briefly, this was a prospective, multicentre, international post-market registry in STEMI patients. A total of 965 patients were enrolled, who all met the inclusion criteria and in whom a STENTYS stent had been placed successfully. After an interim analysis of the first 400 included patients, the steering committee recommended that stent post-dilatation was mandatory, due to the higher event rate in the group without post-dilatation. Patients were contacted at 30 days, one year and two years post procedure by telephone for follow-up.
OUTCOME DEFINITIONS
The primary endpoint of the APPOSITION III study was the occurrence of major adverse cardiac events (MACE), which was defined as the composite of cardiac death, recurrent target vessel myocardial infarction (TV-MI), and clinically driven target lesion revascularisation (TLR). Secondary endpoints included death from any cause, any myocardial infarction (MI), any target vessel revascularisation (TVR), definite stent thrombosis (ST), probable ST, and the combined endpoints of cardiac death/TV-MI, death from any cause/any MI, and definite/probable ST. All endpoint definitions were according to the Academic Research Consortium (ARC) consensus6. All endpoints were adjudicated by an independent clinical events committee.
STATISTICAL ANALYSIS
Continuous variables were presented as mean±standard deviation or median (interquartile range), where appropriate, and categorical variables as frequencies (percentage). Cumulative event rates were estimated using the Kaplan-Meier method and compared with the log-rank test in different subgroups. Follow-up was censored at two years or at the last known date of follow-up, whichever came first. SPSS software package, Version 23 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses.
Results
PATIENT AND PROCEDURAL CHARACTERISTICS
Between April 2010 and January 2012, a total of 1,002 patients who underwent primary PCI were enrolled in this registry. Twenty-four patients did not meet the pre-specified criteria for inclusion in the registry, and in another 10 patients the STENTYS stent could not be placed: in seven patients it was unable to cross the lesion and in three patients the stent could not be deployed. Three patients were lost to follow-up. These patients were excluded from the analysis, resulting in a final study population consisting of 965 patients. The baseline clinical, angiographic and procedural characteristics are reported by Koch et al5.
TWO-YEAR CLINICAL OUTCOMES
The two-year clinical outcomes are listed in Table 1; follow-up was complete in 93% of patients. The primary endpoint of MACE occurred in 105 patients (11.2%) within the two-year follow-up period. Twenty-two patients died from a cardiac cause (2.3%). Recurrent TV-MI occurred in 21 patients (2.3%) (Figure 1). Definite/probable ST occurred in 37 patients (3.9%). Of these ST, 31 (3.3%) were definite ST according to the ARC criteria. Landmark analyses at 30 days and at one year showed that there was only a limited number of incremental events beyond one year.
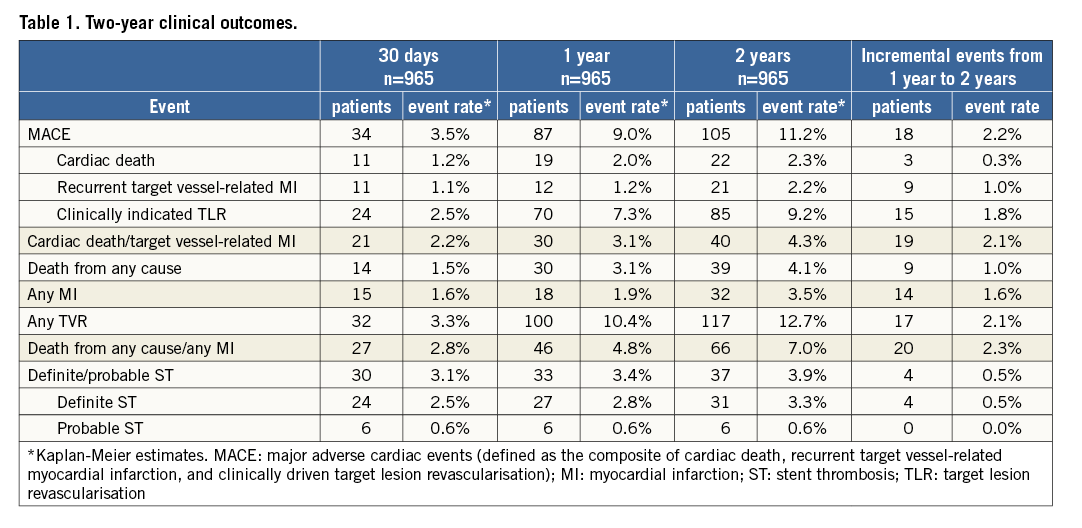
Figure 1. Two-year Kaplan-Meier curves of MACE, TV-MI and cardiac death.
CLINICAL OUTCOME BY POST-DILATATION
In total, balloon post-dilatation was performed in 71% of patients. At two-year follow-up, both probable/definite ST rates (2.8% vs. 6.4%, p=0.009) as well as definite ST rates (2.2% vs. 5.7%, respectively, p=0.006) were significantly lower in the post-dilatation group (Table 2). Landmark analyses showed that MACE rates at 30-day follow-up were significantly lower in the post-dilatation group (2.7% vs. 5.6%, p=0.02) (Figure 2). Beyond 30-day follow-up, from one-year to two-year follow-up there was no difference between the groups. For CD-TLR and probable/definite ST, the landmark analysis showed the same trend in the early significant reduction of events in the post-dilatation group, and no difference beyond 30 days.
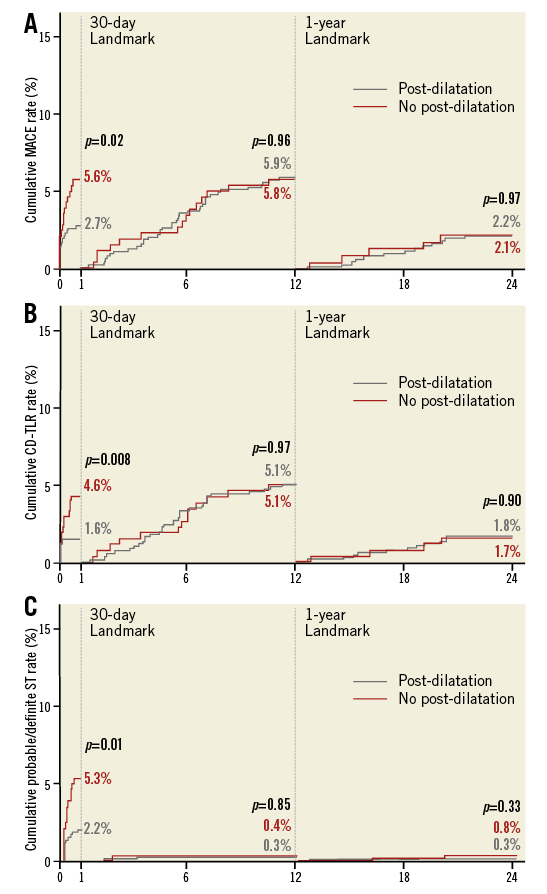
Figure 2. Two-year Kaplan-Meier curves with 30-day and one-year landmark analyses of MACE, clinically driven TLR and ST. A) Cumulative MACE rates. B) Cumulative CD-TLR rates. C) Cumulative probable/definite ST rates.
CLINICAL OUTCOME BY STENTYS TYPE
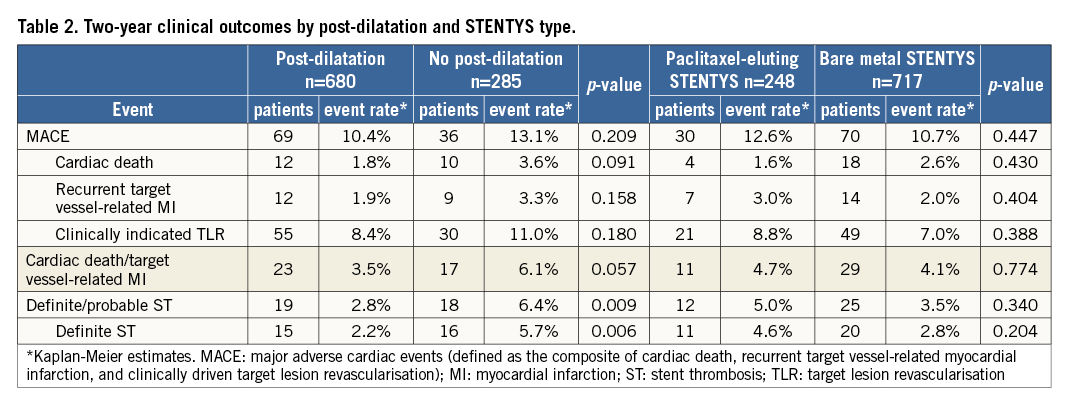
A total of 248 patients (25.7%) were treated with a PES STENTYS and 717 patients (74.3%) with a BMS STENTYS. As previously reported5, the PES group was significantly more at risk as compared to the BMS group. Clinical event rates at two-year follow-up seemed equivalent when comparing the STENTYS PES and BMS subgroups (Table 2). Two-year CD-TLR rates were 8.8% in the PES group and 7% in the BMS group (p=0.388) (Figure 3).
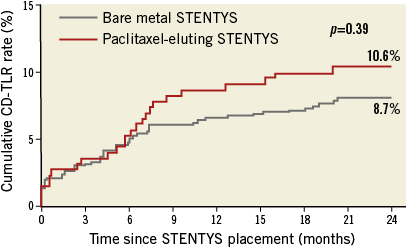
Figure 3: Two-year clinically driven target lesion revascularisation (CD-TLR) rates for bare metal and paclitaxel-eluting STENTYS stent subgroups.
Discussion
The principal findings of this study are the following. 1) The use of the STENTYS self-apposing stent technique is feasible in STEMI patients considering that long-term clinical event rates are low. 2) Post-dilatation gives a significant reduction of MACE rates in the first 30 days after primary PCI. 3) Beyond 30 days and up to two-year clinical follow-up there was no significant difference in MACE rates between patients who received post-dilatation versus patients with no post-dilatation, suggesting that the benefit of post-dilatation was achieved early within the first 30 days after the procedure.
THE STENTYS STENT TECHNIQUE IN THE SETTING OF STEMI
Although superiority has been found in stent strut apposition on intravascular imaging, clinical data are still lacking to prove that this self-apposing technique will lead to superior clinical results compared to contemporary DES. Theoretically, the significantly lower rates of strut malapposition seen on OCT should translate into a lower rate of ST, since malapposition is one of the major causes of early and late ST. If we compare our findings with trials investigating balloon-delivered stent systems, the HORIZONS-AMI trial reported a definite ST rate of 3.7% in the PES group at two-year follow-up7. Other contemporary trials evaluating newer-generation DES in STEMI revealed lower event rates. The biolimus-eluting stent with a biodegradable polymer, evaluated in the COMFORTABLE AMI trial, demonstrated a two-year definite ST rate of 1.4%8. The everolimus-eluting stent, investigated in the EXAMINATION trial, even revealed a two-year definite ST rate of 1%9. Incremental ST rates between one and two years were 0.6% in the BMS group versus 1.1% in the PES group in HORIZONS-AMI, and 0.6% in the biolimus-eluting stent group in the COMFORTABLE trial. We found a similar incremental definite ST rate between one- and two-year follow-up of 0.5%, which corroborates that most ST occurred in the first 30 days. However, the fact remains that contemporary everolimus-eluting stents have proved to be the most safe and efficacious DES to date in STEMI10.
POST-DILATATION OF THE SELF-EXPANDING STENT
Controversy remains with regard to the performance of a balloon post-dilatation in a self-expanding stent. With the landmark analyses at 30 days post procedure, we found a significant reduction of adverse events beyond 30 days in the patients who received balloon post-dilatation. Up to two-year follow-up, there were no differences between the group with and the group without post-dilatation. The definite and probable ST rate in the post-dilatation group was 2.2% at 30 days and rose by 0.6% from 30-day to two-year follow-up. This demonstrates that post-dilatation is required with the use of this device in order to prevent early problems related to the PCI. It is proposed that the outward force of the nitinol platform of the STENTYS stent is not sufficient to obtain a well deployed stent in a stenotic coronary artery by itself5. We have learned from this registry that, despite the reduced ISA using this device, post-dilatation helps to prevent non-optimal stent deployment and underexpansion, both known factors for impaired clinical outcome11.
Study limitations
This study is limited by its observational, non-randomised design. The use of both PES and BMS challenges the interpretation of the clinical results. During the inclusion period of the study, the recommendations for post-dilatation changed after an interim analysis of the first 400 patients included. However, post-dilatation did not become mandatory for the operators. This might have influenced the selection of patients.
Conclusions
The current study provides insights into the long-term clinical results of the use of the STENTYS self-apposing stent technique in STEMI patients. The self-apposing stent technique is feasible in STEMI patients if post-dilatation is performed. Concerning the good performance of present contemporary DES, we need to investigate whether the STENTYS stent could be more beneficial in a subgroup of patients.
| Impact on daily practice In STEMI patients undergoing primary PCI, stent sizing could be more challenging than in elective PCI for treatment of stable coronary artery disease due to the differences in pathophysiology. In STEMI, local thrombus formation, impaired microcirculation and vasoconstriction could all contribute to an underestimation of the true vessel size and therefore stent size. The STENTYS self-apposing stent, which theoretically could solve the problem of stent undersizing, is feasible in STEMI patients and has favourable two-year outcomes considering the stent types used in this study. Future trials should include a limus version of the STENTYS stent in order to compare with contemporary DES outcomes. In the current study, we have seen that, in a STEMI population, balloon post-dilatation should be performed to ensure adequate stent and vessel expansion, resulting in improved clinical outcomes. Furthermore, in those patients presenting in certain anatomic subsets with an increased risk of vessel and stent-size mismatch, such as ectatic target vessels, aneurysms in the target vessel segment or a large difference between the proximal and distal vessel diameters, the STENTYS stent could optimise strut apposition and clinical outcome. |
Appendix. Study organisation and management
The following individuals and organisations contributed to the APPOSITION III study.
Steering Committee: G. Amoroso, OLVG, Amsterdam, the Netherlands; G. De Luca, Azienda Ospedaliera “Maggiore della Carità”, Novara, Italy; U. Limbruno, Ospedale della Misericordia, Grosseto, Italy; M. Wiemer, Herz- und Diabeteszentrum NRW, Bad Oeynhausen, Germany
Clinical Events Committee: P.F. Agostoni, UMC Utrecht, Utrecht, the Netherlands; G.J. Laarman, Tweesteden Ziekenhuis, Tilburg, the Netherlands
Site Management and Data Monitoring: Genae Associates, Antwerp, Belgium
Data Analysis: M.J. Grundeken & J.G.P. Tijssen, AMC, Amsterdam, the Netherlands
Funding
The APPOSITION III study and associated data analysis were funded by STENTYS S.A., Paris, France. Conflict of interest statement
The participating institutions have received institutional research grants from STENTYS. R-J. van Geuns and R. Wessely are members of the scientific advisory board of STENTYS. R. Spaargaren is an employee of STENTYS. The other authors have no conflicts of interest to declare.

