We recently reported the coronary artery bypass graft (CABG) SYNTAX Score, an objective measure of anatomical complexity and revascularisation post coronary artery bypass graft (CABG) surgery1. At four-year follow-up, a non-significant trend towards more adverse clinical outcomes, including all-cause death, was reported in the higher CABG SYNTAX group (≥22)1. The final five-year outcomes of the SYNTAX trial have recently been reported2,3. We report the five-year outcomes of the CABG SYNTAX Score from the CABG arm of the SYNTAX-LE MANS left main angiographic substudy.
At five years, significantly greater all-cause death was seen in the high CABG SYNTAX Score group (≥22) compared to the low CABG SYNTAX Score group (<22) (14.5% vs. 9.1%, log rank p-value=0.012) (Figure 1). Similarly, significantly greater five-year all-cause death/cerebrovascular accident (CVA)/myocardial infarction (MI) (log rank p-value=0.025) and MACCE (major adverse cardiac and cerebrovascular events) (log rank p-value=0.050) were reported.
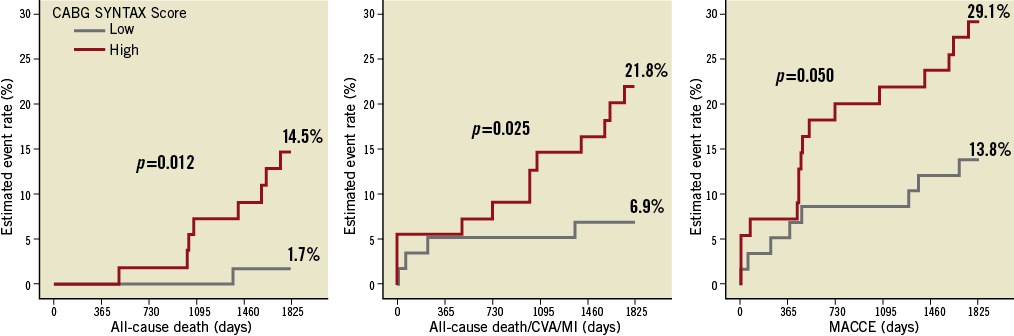
Figure 1. Outcomes (Kaplan-Meier curves) separated by the median of the CABG SYNTAX Score into low (0-21) (n=58) and high (≥22) (n=55) score groups. At 5 years, significantly greater all-cause mortality (left image), significantly greater all-cause death/CVA/MI (middle image) and MACCE (right image) were evident in the high CABG SYNTAX Score group compared to the low CABG SYNTAX Score group. Note the peak in MACCE at approximately 18 months secondary to patients undergoing scheduled coronary angiography, the findings of which triggered repeat revascularisation. Log rank p-values are shown.
Incomplete revascularisation (ICR) has recently been hypothesised and shown to be a surrogate marker of a greater burden and complexity of coronary disease, other vascular disease, and clinical comorbidity, in both CABG and PCI (percutaneous coronary intervention) treated patients4,5. Specifically, in the all-comers CABG and PCI arms of the SYNTAX trial, adverse long-term (four-year) clinical outcomes –including mortality, all-cause revascularisation, and MACCE– were shown to occur more frequently in patients who were incompletely revascularised.
The CABG SYNTAX Score and its PCI equivalent, the residual SYNTAX Score6, both provide objective measures of the complexity of the residual disease and level of revascularisation. These scores may aid in determining a level of “reasonable revascularisation” after undergoing surgical or percutaneous-based revascularisation7, and may have a long-term prognostic role in identifying high-risk patients undergoing CABG or PCI. Validation studies are awaited.
Acknowledgements
The authors would like to thank all the study centres and participants.
Funding
The SYNTAX trial was funded by Boston Scientific.
Conflict of interest statement
K. Dawkins is a full-time employee of, and holds stock in, Boston Scientific. M. Mack has served on the speakers bureau of Boston Scientific, Cordis and Medtronic. T. Feldman has served on the speakers bureau of Boston Scientific; has received grant support from Abbott, Atritech, Boston Scientific Corporation, Edwards, and Evalve; and has worked as a consultant for Abbott, Coherex, Intervalve, Square One, and WL Gore. M-A. Morel’s institution has received a research grant from Boston Scientific. M-A. Morel, H.M. Garcia-Garcia and G.A. van Es are employees of Cardialysis. The other authors have no conflicts of interest to declare.

