Abstract
Aims: MitraClip implantation is evolving as a potential alternative treatment to conventional surgery in high-risk patients with significant mitral regurgitation (MR). However, outcome predictors are under-investigated. The aim of this study was to identify predictors of midterm mortality and heart failure rehospitalisation after percutaneous mitral valve repair with MitraClip.
Methods and results: A total of 150 consecutive patients were followed for a median of 463 days. Survival analyses were performed for baseline characteristics, risk scores and failure of acute procedural success (APS) defined as persisting MR grade 3+ or 4+. Univariate significant risk stratifiers were tested in multivariate analyses using a Cox proportional hazards model. Overall survival was 96% at 30 days, 79.5% at 12 months, and 62% at two years. Multivariate analysis identified APS failure (HR 2.13, p=0.02), NYHA Class IV at baseline (HR 2.11, p=0.01) and STS score ≥12 (HR 2.20, p<0.0001) as significant independent predictors of all-cause mortality, and APS failure (HR 2.31, p=0.01) and NYHA Class IV at baseline (HR 1.89, p=0.03) as significant independent predictors of heart failure rehospitalisation. Furthermore, a post-procedural significant decrease in hospitalisation rate could only be observed after successful interventions (0.89±1.07 per year before vs. 0.54±0.96 after implantation, p=0.01). Patients with severely dilated and overloaded ventricles who did not meet EVEREST II eligibility criteria were at higher risk of APS failure.
Conclusions: The failure of acute procedural success proved to have the most important impact on outcome after MitraClip implantation.
Introduction
Mitral regurgitation (MR) is the second most common form of valvular heart disease requiring treatment in Europe1. Clinical outcome under medical management and after surgery is different in organic and functional disease and varies according to patients’ age, degree of MR, and severity of symptoms2, but in particular patients with functional MR have a poor prognosis, and increasing severity is associated with worse outcome3. Given the high prevalence of MR in the elderly population at high surgical risk due to relevant comorbidities, it is not surprising that surgery is denied in nearly 50% of patients with severe symptomatic MR, with impaired LVEF, older age, and comorbidity as the most striking characteristics of patients who are not operated4.
The percutaneous edge-to-edge mitral valve repair with MitraClip® (Abbott Vascular, Santa Clara, CA, USA) is based on the surgical technique first described by Alfieri5. This novel procedure was initially performed in the EVEREST trials in selected patients amenable to surgery with predominantly degenerative mitral valve disease6,7. However, patients who are currently treated with MitraClip in daily clinical practice are mostly at very high or prohibitive surgical risk with predominantly functional disease due to dilative or ischaemic cardiomyopathy8. Clinical midterm outcome following MitraClip implantation is, as yet, not well documented, and valid outcome predictors for overall survival and heart failure rehospitalisation have not yet been identified. In the present study, we therefore aimed to identify risk factors with prognostic impact on midterm survival and heart failure rehospitalisation in a single-centre cohort of consecutive “real-world” patients treated with MitraClip.
Methods
STUDY DESIGN
We report on the first 150 consecutive patients with moderate to severe MR treated with MitraClip at the University Medical Centre of Göttingen between April 2009 and June 2012. This prospective observational study was reviewed and approved by the ethics committee of the University Medical Centre Göttingen, and written informed consent was obtained from all patients.
PRE-INTERVENTIONAL EVALUATION
The indication and technique for treatment of MR was discussed in an interdisciplinary Heart Team in accordance with current guidelines9,10. All patients underwent intensive pre-interventional screening including transthoracic and transoesophageal echocardiography. Surgical risk was assessed by current scoring systems like the logistic EuroSCORE I and the Society of Thoracic Surgeons (STS) mortality risk calculation. Exclusion criteria for MitraClip implantation were a mitral valve area <2.0 cm2 by planimetry6 and acute endocarditis. The exclusion criteria for the EVEREST II trial7 were evaluated for each patient in order to characterise the patient population, but did not represent a treatment contraindication in the present study.
PROCEDURE
The MitraClip implantation was performed as previously described6 in a hybrid operating room under general anaesthesia and guided by two-dimensional and three-dimensional TEE and fluoroscopy. All procedures were performed by the same interventional cardiologists (W.S. and M.H.) and a team of two experienced echocardiographers (M.P. and K.R.).
ECHOCARDIOGRAPHY
Echocardiographic measurements were performed before the MitraClip implantation, during and directly after the procedure, at discharge, and during follow-up (at six months and 12 months) according to current recommendations. The severity of MR was graded as proposed in the EVEREST I trial6 in accordance with the American Society of Echocardiography11, and post-procedural grading was performed according to Foster et al12. The definition of acute procedural success (APS) was adopted from the EVEREST II trial9 and was defined as a residual MR grade of 2+ or less measured at the time of hospital discharge after MitraClip implantation. To exclude the influence of general anaesthesia, we explicitly did not count the direct postoperative MR grade. Measurement of left ventricular volumes and ejection fraction was performed by using the biplane Simpson’s method.
FOLLOW-UP
At six and 12 months, we performed transthoracic echocardiography, a clinical examination, a six-minute walk test and a structured interview including the Minnesota Living with Heart Failure Questionnaire (MLWHF QoL), and assessed the serum N-terminal pro-brain natriuretic peptide (NT-proBNP) levels. In some patients who refused to undertake a surveillance visit (mainly due to the long distance of our clinic from their places of residence), the follow-up was restricted to a telephone interview, but medical documents and echocardiographic recordings were acquired from resident cardiologists whenever possible. Before completion of the manuscript, all patients were followed by telephone contact again in January and February 2013 to investigate the incidence of major adverse events and of death. Also, the number of heart failure hospitalisations one year before and one year after MitraClip implantation was investigated and all relevant medical documents acquired to examine possible alterations in hospitalisation rates.
STUDY ENDPOINTS
All-cause mortality was defined as the primary clinical endpoint of the study. As the secondary endpoint, heart failure hospitalisation was chosen and was defined as hospitalisation due to clinical signs of worsening congestive heart failure with objective symptoms including pulmonary congestion, worsening oedema, hypoperfusion or documented volume overload leading to administration of diuretic and/ or inotropic therapy, or institution of mechanical support (i.e., assist devices).
STATISTICAL ANALYSIS
Statistical analysis was performed with the Statistical Computing Software R (version 2.15.1; http://www.r-project.org) or with GraphPad Prism (version 4.0). Continuous variables are presented as median and interquartile range, and were compared using the Mann-Whitney U test. Categorical variables are presented as absolute numbers and percentage, and were compared by Pearson’s chi-square test. A value of p<0.05 was considered statistically significant.
Survival analysis was performed on time to event data (i.e., time to first heart failure hospitalisation or time to death of any cause) using the R package survival. Survival data were visualised by Kaplan-Meier plots, and significance was calculated by the log rank test (for two group comparisons). For multivariate models, the Cox proportional hazards model was used. Survival and rehospitalisation analyses were performed for baseline characteristics, risk scores and failure of procedural success. Risk stratifiers that were found to be significant in univariate analyses were tested in multivariate analyses. The influence of different risk scores (logistic EuroSCORE I, STS score) and the presence of EVEREST II exclusion criteria as a risk stratifier were assessed using ANOVA analysis. In this model, STS score and EuroSCORE were used as continuous variables. A multivariate Cox model (coxph) was fitted using forward variable selection and model comparisons in different orders.
Results
BASELINE CHARACTERISTICS
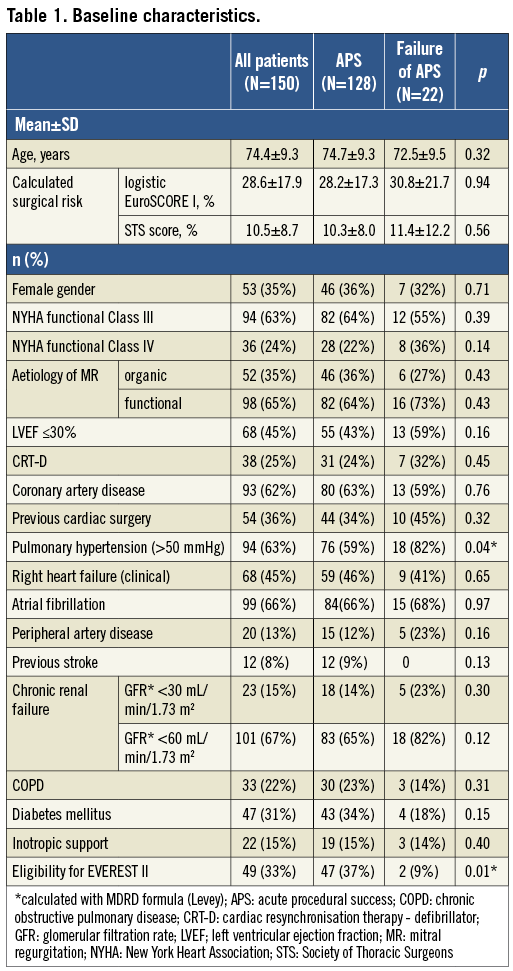
The patient cohort was characterised by advanced age (mean, 74.4±9.3 years) and significant comorbidities leading to a high surgical risk expressed by a high logistic EuroSCORE I (mean, 28.6±17.9%) and a high STS score (mean, 10.5±8.7%) (Table 1). The aetiology of MR was predominantly functional (in 66%), and 45% of the total patient cohort suffered from a severely reduced left ventricular ejection fraction (LVEF) of ≤30%. Of note, only 33% of patients would have met the EVEREST II eligibility criteria7. Furthermore, our patients showed severe symptoms of congestive heart failure, with 87% presenting in New York Heart Association (NYHA) Classes III and IV, 45% suffering from clinical signs of right heart failure, and 15% even being dependent on inotropic support.
PROCEDURAL AND IN-HOSPITAL OUTCOME
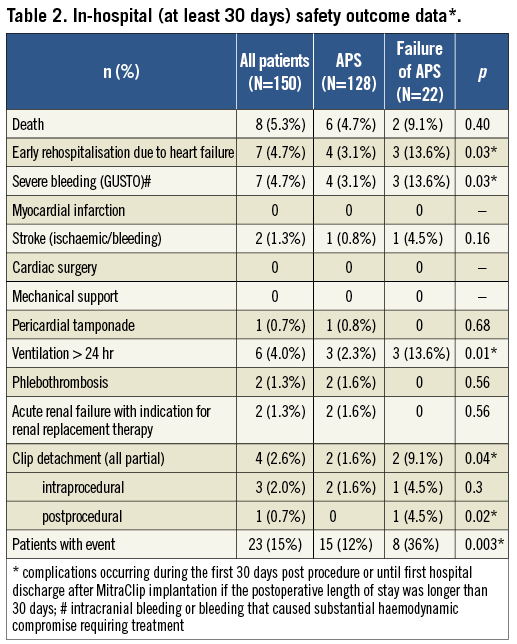
The procedure itself was relatively safe, and no peri-interventional mortality was observed. The in-hospital mortality was 5.3%. Four patients died after a complicated in-hospital course with primary bleeding complications and consecutive further events on days 33, 34, 24 and 12 (see also below). The causes of death in the remaining four cases were therapy-resistant ventricular fibrillation (day 10) in one patient and septic shock (days 6, 11 and 5) in three patients (one of whom had already received MitraClip implantation as last-resort therapy in a very bad clinical condition with acute respiratory distress syndrome and massive dependence on inotropic support).
Complications occurring in the first 30 days post procedure or until first hospital discharge after MitraClip implantation (if the postoperative length of stay was longer than 30 days) are demonstrated in Table 2. Peri-interventional myocardial infarction, need for mechanical assistance or immediate surgery did not occur. However, 23 patients (15%) experienced procedure-related complications. Relevant bleeding complications (classified as GUSTO severe) were observed postoperatively in seven cases (4.7%) and were often associated with the need for therapeutic anticoagulation due to mechanical prosthetic valves (n=2) or atrial fibrillation (n=2) leading to diffuse bleeding (in three patients) or retroperitoneal haematoma (in one patient). The bleeding led to further complications like prolonged ventilation (>24 hr) with development of pneumonia, acute renal failure or development of ischaemic stroke in the latter four patients who died due to septic shock in the end on days 33, 34, 24 and 12. The remaining bleeding complications were groin haematoma with need for transfusion in one patient (who experienced no further complications), pericardial tamponade (which was successfully treated by pericardiocentesis) in one patient who had undergone left atrial appendage occlusion at the same time as MitraClip implantation, and intracranial bleeding on day 8 after the procedure in one patient (who showed a good neurological recovery without relevant residua).
On average, 1.5±0.6 clips per patient were implanted (0 clips in one case, one clip in 79 cases, two clips in 65 cases, three clips in five cases). Acute procedural success was achieved in 128 patients (85%). No complete clip detachment with embolisation occurred. However, four patients (2.6%) experienced a partial clip detachment from one leaflet which was successfully treated with two additional clips during the same procedure in two cases. One further patient underwent operative mitral valve replacement on day 49, and the fourth patient was scheduled for surgery but died from severe pneumonia before the operation. Before hospital discharge, 18 patients had a residual MR grade 3+ and four patients a residual MR grade 4+. These 22 individuals are summarised as “patients with failure of APS” in the following section.
SIX-MONTH OUTCOMES (MATCHED DATA FOR STATISTICAL ANALYSES)
At six months post procedure, 22 patients had already died. All 128 remaining patients could be contacted via telephone, and 112 of them consented to undergo a follow-up visit including clinical examination and transthoracic echocardiography (106 at our clinic, six visiting their resident cardiologists). 79% had an MR grade ≤2, and 65% corresponded to NYHA functional Classes I/II.
Among the 22 patients with failure of APS, four had undergone a second mitral valve intervention (surgical mitral valve replacements, n=3; second MitraClip procedure, n=1) and seven had already died before the time of the six-month follow-up. Concerning the 128 individuals with initial APS, 15 deaths and no reinterventions occurred in the first six months. However, an echocardiographic deterioration of the acute procedural result with recurrence of MR grade 3+ or 4+ was present in 14 patients at six-month follow-up (MR 3+, n=12; MR 4+, n=2) (Figure 1).
TWELVE-MONTH OUTCOMES (MATCHED DATA FOR STATISTICAL ANALYSES)
Of the 131 patients who had completed 12 months post procedure at the time of manuscript preparation, 31 had already died. All 100 remaining patients were contactable by telephone, and 92 of them had already undergone a follow-up visit including echocardiography (83 at our clinic, nine at their resident cardiologist). In the eight patients who refused to undergo a surveillance visit, the follow-up was restricted to a telephone interview. Of the 92 patients with available echocardiographic data, 85% had an MR grade ≤2, and 67% were in NYHA functional Classes I/II.
Regarding the APS failure cohort, one further surgical mitral valve replacement and four further deaths occurred between six and 12-month follow-up. Among patients with initial APS, five further patients died before 12-month follow-up, two underwent a second MitraClip procedure, one received a left ventricular assist device and one underwent heart transplantation (Figure 1).
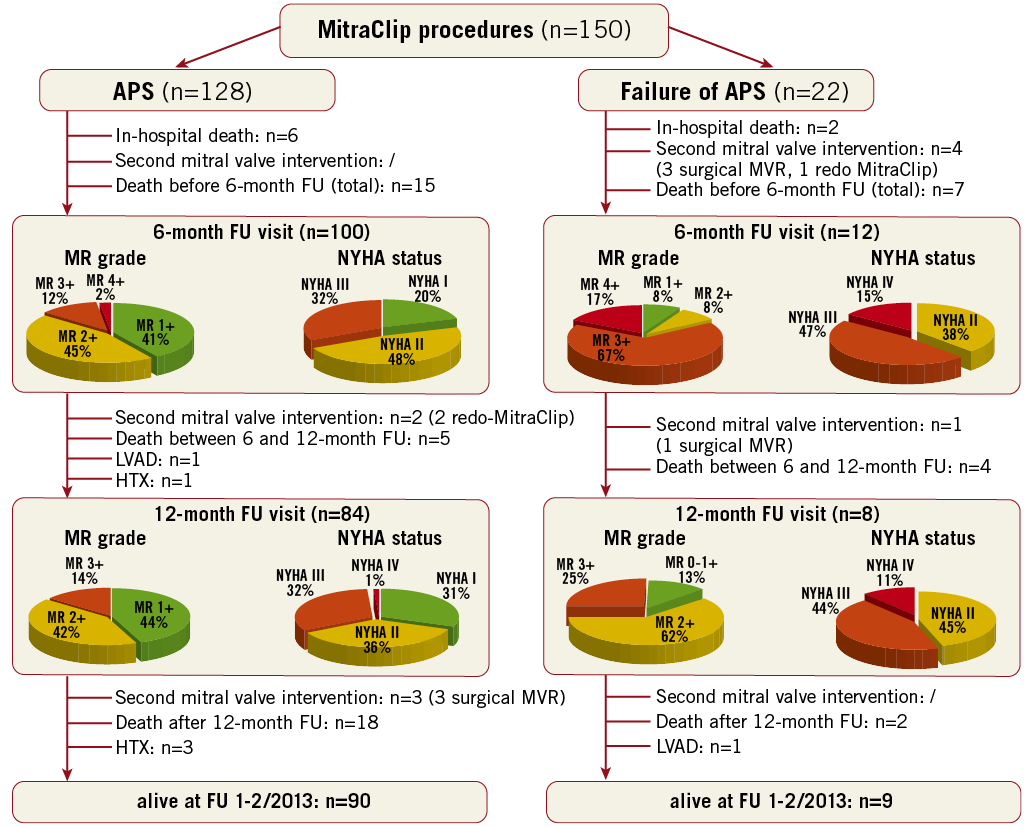
Figure 1. Flow chart demonstrating clinical course and follow-up events of our MitraClip cohort of 150 consecutive patients depending on presence or absence of acute procedural success. APS: acute procedural success; FU: follow-up; HTX: heart transplantation; LVAD: left ventricular assist device; MR: mitral regurgitation
EVENTS OCCURRING BEYOND 12-MONTH FOLLOW-UP
Concerning the APS failure cohort, two further patients died and one received a left ventricular assist device. No further mitral valve interventions were carried out. At the time of the last telephone follow-up in Jan/Feb 2013, nine patients (41%) were still alive in this group (n=1 in NYHA Class I, n=4 in NYHA Class II, n=4 in NYHA Class III).
Regarding the 128 patients with initial acute procedural success, 18 further deaths occurred after the 12-month follow-up, three surgical mitral valve replacements were carried out due to a deterioration of the initial procedural result, and three patients underwent heart transplantation. At the time of follow-up in Jan/Feb 2013, 90 patients (70%) were still alive in this group. Of the survivors, 24% (n=22) pertained to NYHA Class I, 46% (n=41) to NYHA Class II, 27% (n=24) to NYHA Class III and 3% (n=3) to NYHA Class IV.
IMPACT OF RECURRENT HIGH-GRADE MR DURING FOLLOW-UP
As already reported, an echocardiographic deterioration of the acute procedural result with recurrence of MR grade 3+ or 4+ was present in 14 patients after initial successful MitraClip implantation (organic MR, n=3; functional MR, n=11) at six-month follow-up. Among these individuals, four deaths (29%) occurred during follow-up, two patients underwent a second MitraClip procedure, one patient a surgical mitral valve replacement, and two patients underwent heart transplantation.
ANALYSIS OF ALL-CAUSE MORTALITY DURING FOLLOW-UP
Median follow-up was 463 days. Overall survival was 96% at 30 days, 85% at six months, 80% at 12 months, and 62% at two years. Altogether, 51 patients (34%) had died at the time of recent follow-up. The eight in-hospital deaths have already been reported. Causes of death occurring after first discharge (n=43) were the following: heart failure and/or documented ventricular arrhythmia (n=19), sudden death (n=7), death due to perioperative complications after conventional mitral valve surgery (n=2), acute renal failure (n=3), pneumonia (n=3), cancer (n=2), pleural empyoema (n=1), septic shock due to ileus (n=1), intracranial bleeding (n=1), expected death at home (or in nursing home) in bad clinical condition without further classifiable cause (n=3), unknown (n=1).
Furthermore, we performed survival analyses to test the potential of different baseline parameters, risk scores and the presence of failure of acute procedural success to predict mortality. Univariate analyses identified NYHA functional Class IV at baseline, glomerular filtration rate (GFR) <60 mL/min/1.73 m2, logistic EuroSCORE I ≥20%, STS score ≥12%, presence of exclusion criteria for EVEREST II and failure of acute procedural success (APS) as significant predictors of mortality (Table 3A). Importantly, established risk factors for elevated mortality in conventional surgery like age, female gender, previous cardiac surgery, reduced LV ejection fraction, peripheral vascular disease, and chronic lung disease did not predict mortality in our MitraClip patient cohort.
Risk factors that were found to be significant in univariate analyses were tested in multivariate analyses using a Cox proportional hazards model. Heart failure symptoms according to NYHA functional Class IV (p=0.01) and APS failure (p=0.02) proved to be significant also in multivariate analyses, whereas GFR <60 mL/min/1.73 m2 failed to be significant (p=0.08) (Table 3B).
Similarly, the independent predictive value of an STS score ≥12 could be confirmed by ANOVA analysis using forward variable selection, whereas logistic EuroSCORE I and EVEREST II exclusion criteria did not yield any additional significance. In a combined ANOVA analysis of all significant parameters, failure of APS and STS score ≥12 still had significant impact on mortality prediction, whereas NYHA functional Class IV did not, in the presence of the STS score.
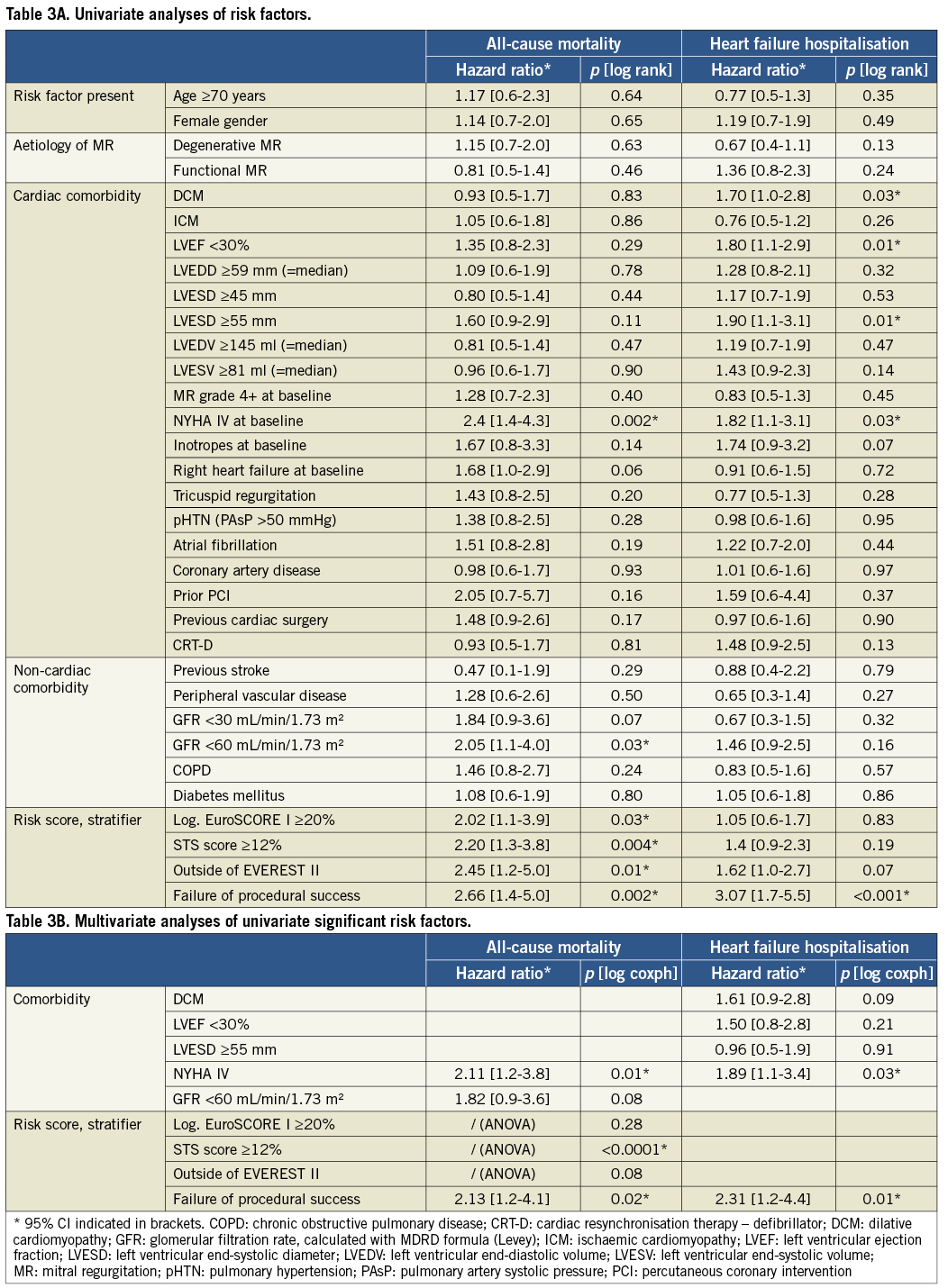
In conclusion, failure of acute procedural success and STS score of ≥12 could be identified as highly significant independent predictors of all-cause mortality by different statistical models. A Kaplan-Meier curve demonstrating the impact of APS failure on survival is demonstrated in Figure 2.
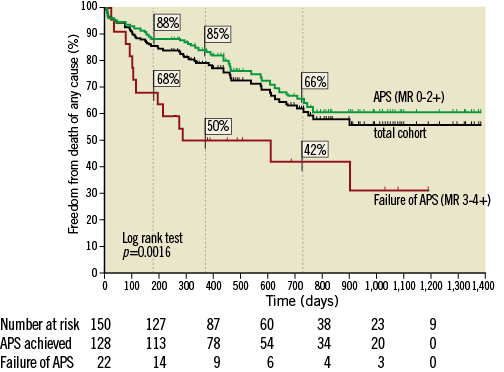
Figure 2. Survival dependent on acute procedural success. Kaplan-Meier curve demonstrating the impact of acute procedural success (APS) on freedom from death of any cause (survival proportions at 6 months, 1 year and 2 years are indicated for patients with and without APS).
ANALYSIS OF HEART FAILURE REHOSPITALISATION DURING FOLLOW-UP
In the whole cohort, freedom from heart failure rehospitalisation was 95% at 30 days, 73% at six months, 65% at 12 months, and 47% at two years.
Univariate analyses were able to confirm a significant impact on heart failure hospitalisation for dilative cardiomyopathy (DCM), left ventricular ejection fraction (LVEF) <30%, left ventricular end-systolic diameter (LVESD) ≥55 mm, NYHA functional Class IV at baseline and failure of acute procedural success (Table 3A). NYHA functional Class IV at baseline and failure of APS proved to be significant also in multivariate analysis (p=0.03 and p=0.01), whereas DCM, LVEF ≤30% and LVESD ≥55 mm were not significant independent predictors (p=0.09, p=0.21 and p=0.91). ANOVA analysis revealed that only APS failure remained a significant predictor of heart failure hospitalisation independent of the order in which the parameters were fitted into the model (Figure 3, corresponding Kaplan-Meier curve).
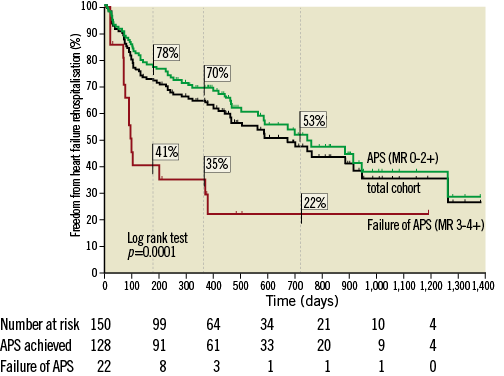
Figure 3. Freedom from heart failure rehospitalisation dependent on acute procedural success. Kaplan-Meier curve demonstrating the impact of acute procedural success (APS) on freedom from heart failure rehospitalisation (survival proportions at 6 months, 1 year and 2 years are indicated for patients with and without APS).
HOSPITALISATION RATE BEFORE AND AFTER MITRACLIP IMPLANTATION
We compared the heart failure hospitalisation rate during one year before and one year after the procedure in all patients who had been discharged alive after MitraClip implantation and who had completed 12 months post procedure (n=129 persons). In patients with successful procedures (n=109), a significant decrease in heart failure hospitalisations was observed (0.89±1.07 pre-procedure vs. 0.54±0.96 post-procedure, p=0.01), whereas no change was present in patients with APS failure (n=20, 1.45±1.79 vs. 1.30±2.43, p=0.76) (Figure 4).
Figure 4. Comparison of heart failure hospitalisation rates before and after MitraClip. Number of heart failure hospitalisations during 12 months before and 12 months after MitraClip implantation depending on presence or absence of acute procedural success. Bars indicate mean and standard error.
PREDICTORS OF ACUTE PROCEDURAL FAILURE
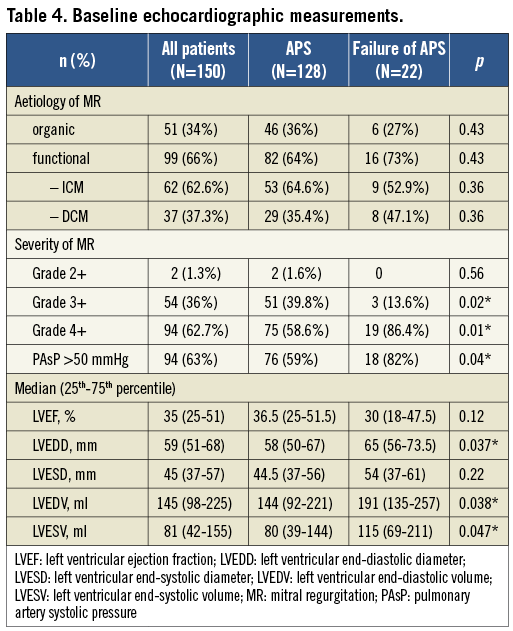
In order to characterise patients with acute procedural failure further, we considered baseline characteristics, echocardiographic parameters and peri-interventional features. Patients with unsuccessful procedures presented more frequently with pulmonary hypertension (defined as pulmonary artery systolic pressure of >50 mmHg, p=0.04) and met the EVEREST II eligibility criteria less frequently (p=0.01) (Table 1). However, remaining baseline characteristics as well as calculated surgical risk expressed by logistic EuroSCORE I and STS score did not differ. Regarding echocardiographic measurements, the patient cohort with APS failure was characterised by significantly higher left ventricular volumes and left ventricular end-diastolic diameters as well as a significantly higher prevalence of MR grade 4+ at baseline (Table 4), whereas the distribution of MR aetiologies was comparable. Furthermore, total procedure time (median, 132 [105-220] vs. 93 [65-127] min, p<0.0001) and radiation time (median, 34.9 [25.3-63] vs. 21 [15-31] min, p<0.0001) were significantly higher in unsuccessful procedures, and patients were more likely to experience procedural complications like severe bleeding (p=0.03), need for prolonged ventilation (p=0.01), and partial clip detachment (p=0.04) (Table 2). The distribution of one-clip, two-clip or three-clip procedures did not differ significantly.
Discussion
First of all, our observational study provides an insight into the use of MitraClip therapy and its midterm outcomes in daily clinical practice, reporting on a cohort characterised by a median LVEF of 35% and a prevalence of functional MR of 66%. Similar patient characteristics are observed, for example, in the German transcatheter mitral valve interventions (TRAMI) registry8 suggesting that MitraClip therapy is evolving as a new therapeutic option for heart failure patients with significant MR, possibly even as a bridge to transplant (four of our patients underwent heart transplantation one to three years after MitraClip implantation; Figure 1).
CLINICAL OUTCOMES
As we were able to demonstrate in our study and as consistently documented in previous studies for follow-up periods of around 12 months, MitraClip therapy is able to reduce MR severity, to reverse ventricular remodelling, and to improve clinical symptoms in a high proportion of patients with significant MR and high or prohibitive risk of conventional surgery13-16. To our knowledge, our study is the first that was able additionally to demonstrate a significant decrease in heart failure hospitalisations after successful MitraClip implantations.
However, all previously published studies report on patients with failure of acute procedural success, commonly defined as MR grade >2+ at discharge. In the EVEREST I trial6 26% of patients met this criterion, in the EVEREST II trial7 23%, in the EVEREST II High Risk Cohort16 20.5%, in the PERMIT-CARE study13 approximately 20%, in the Swiss registry17 15% and in the Hamburg single-centre cohort15 8%. Regarding our own series, 15% of patients experienced APS failure, which is well in line with the previous reports.
PREDICTORS OF ALL-CAUSE MORTALITY AND HEART FAILURE REHOSPITALISATION
In our study, we were able to identify the failure of acute procedural success, an STS score of ≥12 and NYHA Class IV at baseline as independent predictors of all-cause mortality, and APS failure and NYHA Class IV at baseline alone as independent predictors of heart failure hospitalisation during follow-up, emphasising the adverse impact of procedural failure. Further risk predictors that have recently been reported to have significant independent impact on survival and rehospitalisation were a logistic EuroSCORE I of ≥20%18 and the degree of residual MR14. However, in our own patient cohort a logistic EuroSCORE I of ≥20% was a significant predictor of all-cause mortality only in univariate analysis but lost its significant impact in multivariate ANOVA analysis as soon as the STS score was inserted into the model. Concerning the degree of post-interventional MR, only the presence of MR 3+ and 4+ (defined as failure of APS) was associated with significantly increased mortality and morbidity during follow-up, whereas no significant differences could be observed in survival curves of patients with MR 0-1+ and 2+ at discharge (p=0.6). Very recently, Sürder et al17 also identified failure of APS and discharge MR grade as univariate significant predictors of survival among the first 100 consecutive patients in the Swiss registry.
Importantly, the presence of established risk factors for adverse outcome after conventional surgery like age, female gender, previous cardiac surgery, atrial fibrillation, peripheral vascular disease, diabetes and chronic lung disease10 had no impact on outcome in our study as well as in a previous report14, and should therefore not be a reason for rejection of a patient for MitraClip implantation.
PREDICTION OF PROCEDURAL FAILURE
Patients with severely dilated and overloaded ventricles who did not meet EVEREST II eligibility criteria, that is to say patients with worse clinical baseline conditions, were at higher risk of procedural failure. The resulting question whether the procedural failure is the primary cause of worse outcomes or whether it might also be an indicator of worse baseline status cannot be answered on the basis of our observational study.
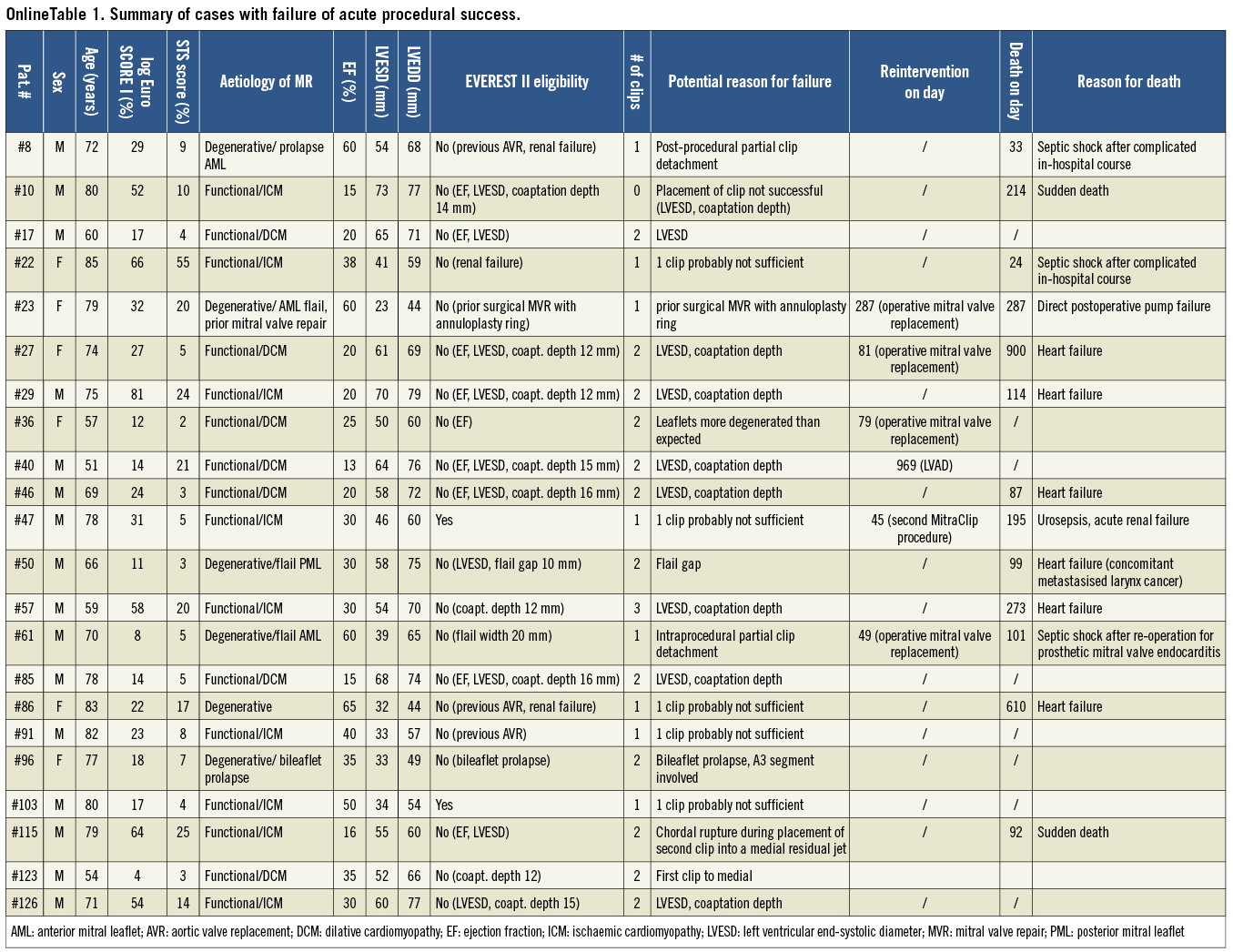
A clinical summary of the 22 cases with APS failure is presented in Online Table 1. The majority of unsuccessful procedures took place in our first 50 MitraClip cases. This has recently been suggested, for the most part, to reflect the impact of the learning curve19. However, APS failure still occurred beyond this initial experience. This indicates that procedural success is influenced by additional factors not related to learning. Many patients with APS failure in our series exhibited anatomical valve morphologies or ventricular dysfunction that met EVEREST II exclusion criteria. The main (coherent) reasons for EVEREST II ineligibility in the APS failure group were a severely reduced left ventricular ejection fraction (≤25%, n=9), severe left ventricular dilation (LVESD >55 mm, n=10), and coaptation depths of >11 mm (n=9)6, resulting in significant volume overload and pulmonary hypertension (patients # 10, 17, 27, 29, 36, 40, 46, 57, 85, 115, 123, 126). These patients with functional MR were predominantly at high surgical risk and per se not good surgical candidates, because current guidelines give no surgical recommendation for patients with secondary MR and LVEF ≤30% without the option for revascularisation10. In patients with degenerative mitral valve disease and APS failure, the EVEREST II ineligibility resulted mainly from anatomical causes like flail gap ≥10 mm (#50), flail width ≥15 mm (#61), bileaflet prolapse (#96), or prior surgical mitral valve repair with an annuloplasty ring (#23). Renal failure would have excluded three further patients (#8, 22, 86) from the EVEREST II trial. Some of our patients with APS failure would in principle have been surgical candidates, but reasons like metastasised cancer (patient #50), repeated previous cardiac operations (#61), or patient’s wish (#96) led to a referral for MitraClip implantation.
IMPACT OF RECURRENT HIGH-GRADE MR DURING FOLLOW-UP
Recurrence of MR grade 3+ or 4+ at six-month follow-up was associated with elevated mortality during further follow-up (29% in this subgroup compared to 19% in patients with MR ≤2). The late procedural failure was speculatively attributed to a progression of the underlying cardiomyopathy with alterations of LV geometry in most cases - which might also have influenced mortality in addition to MR itself worsening. No late clip detachments were observed.
STUDY LIMITATIONS
This analysis represents a single-centre experience, which could be regarded as a limitation of the study. However, the complete analysis of consecutive patients without any exclusion is never realised in registries which additionally often lack a good follow-up which could create an analytical bias. For example, in the German transcatheter mitral valve interventions (TRAMI) registry, a substantial number of patients were enrolled retrospectively which allowed no follow-up beyond discharge after MitraClip implantation. In our study, no patient was completely lost to follow-up. However, a significant proportion of patients had already died at six or 12 months, which may have caused an optimistic selection bias concerning these measurements.
Haemodynamic parameters measured during MitraClip implantation that have recently been shown to predict midterm outcome (mean pulmonary capillary wedge pressure and mean pulmonary artery pressure20) were not included in the present analysis. Implantation success was assessed by Doppler echocardiographic parameters that are known to be potentially challenging and associated with inter-observer variability after MitraClip implantation.
Furthermore, this purely observational study was not designed to address pathophysiological issues (particularly with regard to differences between functional and degenerative MR). This should be done by future multicentre studies.
Conclusions
Treatment of significant MR with MitraClip is efficacious and results in significant clinical improvements in a high proportion of patients after six and 12 months. In our patient cohort, the failure of acute procedural success emerged as the strongest independent predictor of midterm outcome concerning both all-cause mortality and heart failure hospitalisation during follow-up. Therefore, every effort should be undertaken to avoid APS failure. First of all, this includes a thorough discussion of the different treatment options and their risks in the Heart Team. The only randomised trial comparing MitraClip implantation and surgical MVR in patients with predominantly degenerative MR (EVEREST II7) demonstrated a superior efficacy of conventional surgery in terms of MR reduction and freedom of repeat interventions, whereas MitraClip therapy was associated with superior safety. This fact should be taken into account particularly in patients with degenerative mitral valve disease in whom conventional surgery as the gold standard for treatment10 is still an option.
However, the number of elderly patients with severe functional MR and relevant comorbidities who are not good surgical candidates is expected to increase further in the future due to an ageing population and improved medical therapy. In patients who are considered ineligible for surgery, eligibility for MitraClip therapy should be carefully evaluated according to anatomical considerations. As we can confirm after analysis of our own data, the EVEREST II exclusion criteria are a useful guideline for patient selection at the beginning of MitraClip implantations in new centres, whereas patients with difficult anatomical conditions should be postponed until approximately 50 procedures have been performed. Despite this, a majority of patients with successful procedures in our series also did not meet the EVEREST II eligibility criteria, indicating that we need to carry out prospective studies to define new anatomical criteria that allow a better prediction of APS. Nevertheless, the occurrence of procedural failure with the described adverse impact will not be completely avoidable even in highly experienced centres. Patients with unsuccessful procedures require particularly close attention in post-interventional care. Corrective percutaneous or surgical interventions should be considered and discussed early.
Conflict of interest statement
M. Puls, M. Hünlich, K. Rüter, R. Seipelt and W. Schillinger have received travel expenses from Abbott Vascular. W. Schillinger has also received lecture fees from Abbott Vascular. The other authors have no conflicts of interest to declare.
Online data supplement