Intravascular ultrasound (IVUS) and optical coherence tomography (OCT) have been established as important diagnostic and guidance tools for percutaneous coronary intervention (PCI) procedures1. Sequential use of the two modalities might overcome the inherent limitations of each technology; however, the potential risks of complications and cost are hard to ignore. Hybrid IVUS-OCT systems can overcome the limitations associated with each technology and integrate the advantages of both modalities. In 2018, a hybrid IVUS-OCT system for clinical application was reported for the first time2. Recently, a novel hybrid imaging system was approved for clinical use (PANOVISON; Panovision Co., Ltd), allowing synchronous acquisition of the two imaging modalities and an immediate, coregistered image review. We conducted this first-in-human study to test each modality of the hybrid imaging system, representing this novel device’s first large-scale human application.
The study was a prospective, multicentre, randomised, open-label, self-controlled, non-inferiority trial. The protocol was approved by the institutional review boards and ethics committees in all participating centres. All patients provided written informed consent. Specific inclusion and exclusion criteria are described at ClinicalTrials.gov: NCT05596279. We used a web-based randomisation service (sealed envelope; Sealed Envelope Ltd) to randomly assign participants (1:1) to receive either control IVUS (IVUS arm) or control OCT (OCT arm) and all the participants underwent hybrid imaging before or after the control imaging. This study was mainly focused on the identification of stents; therefore the hybrid images were all acquired after stent implantation, which reduced any potential risk of device-related complications and contrast-related renal injury due to additional preoperative imaging. The primary endpoint was to demonstrate the non-inferiority of each imaging modality of the hybrid imaging system versus control IVUS or control OCT regarding the clear stent capture rate (CSCR)3. The secondary endpoints were the proportion of patients with a clear image length larger than 24 mm34, the detection rate of complications after stenting5, and the device success rate. The safety endpoint was the incidence of procedure-related adverse cardiovascular events (i.e., coronary spasm, acute vessel occlusion, coronary dissection, or thrombosis). The non-inferiority margin for this study was set as −9.54%, in accordance with a previous study3. For evaluating the feasibility and safety of the equipment thoroughly, the sample size was extended to 100 by trial leadership.
A total of 99 patients were included in the final analysis (one patient dropped out for failed dual-modality acquisition in the OCT arm due to operational reasons). The median age was 60 years old, and the gender ratio was close to 1:1. In the two arms, the absolute difference of CSCR between stand-alone imaging and control imaging is shown in Central illustration. Since the margin of error of the 95% confidence interval did not cross the predefined value of −9.54% (p<0.001), the primary endpoint was met. Coregistered dual-modality images were successfully acquired in all participants. Each imaging modality of the hybrid IVUS-OCT system achieved a clear imaging length greater than 24 mm. The detection of complications after PCI is shown in Central illustration B. Surprisingly, the detection rate of tissue prolapse was significantly higher by stand-alone IVUS compared with control IVUS. There were no procedural complications observed during the procedure.
This study was the first-in-human application of the novel hybrid imaging system. In addition to its good performance in image acquisition, the hybrid imaging catheter had a higher pullback speed, up to 40 mm/s, and a longer pullback length, up to 150 mm (Table 1). Hence, it could be concluded that the novel hybrid IVUS-OCT had achieved good clinical performance and reliability. In this study, the detection rate of tissue prolapse was significantly higher by stand-alone IVUS versus control IVUS. This finding might be explained by the fact that lumen was well flushed with contrast, and the detection capability of surface microstructures was significantly improved even if the frequency of the IVUS transducer was only 35 MHz (Central illustration C). Hence, not only did this novel imaging system realise complementary advantages with integrated imaging, it also provided high-definition IVUS images compared with state-of-the-art technologies. In our study, there were no device-related events that occurred. The higher pullback speed permitted a shorter imaging time with a longer pullback length, which objectively guaranteed the clinical safety of the hybrid imaging device.
Even though this was a small sample study, it provided extensive clinical application data for the novel equipment. Since all the hybrid imaging pullbacks were acquired after stent deployment, the passability and safety of the hybrid catheter in culprit lesions could not be assessed adequately. In addition, the evaluation of culprit plaque composition was also unavailable.
Our study demonstrated for the first time the feasibility and safety of a novel hybrid IVUS-OCT system. Each imaging modality of the hybrid system can provide clear intravascular images after stent implantation without procedure-related adverse cardiovascular events. This finding suggested that the novel hybrid IVUS-OCT system can be safely used in patients undergoing PCI.
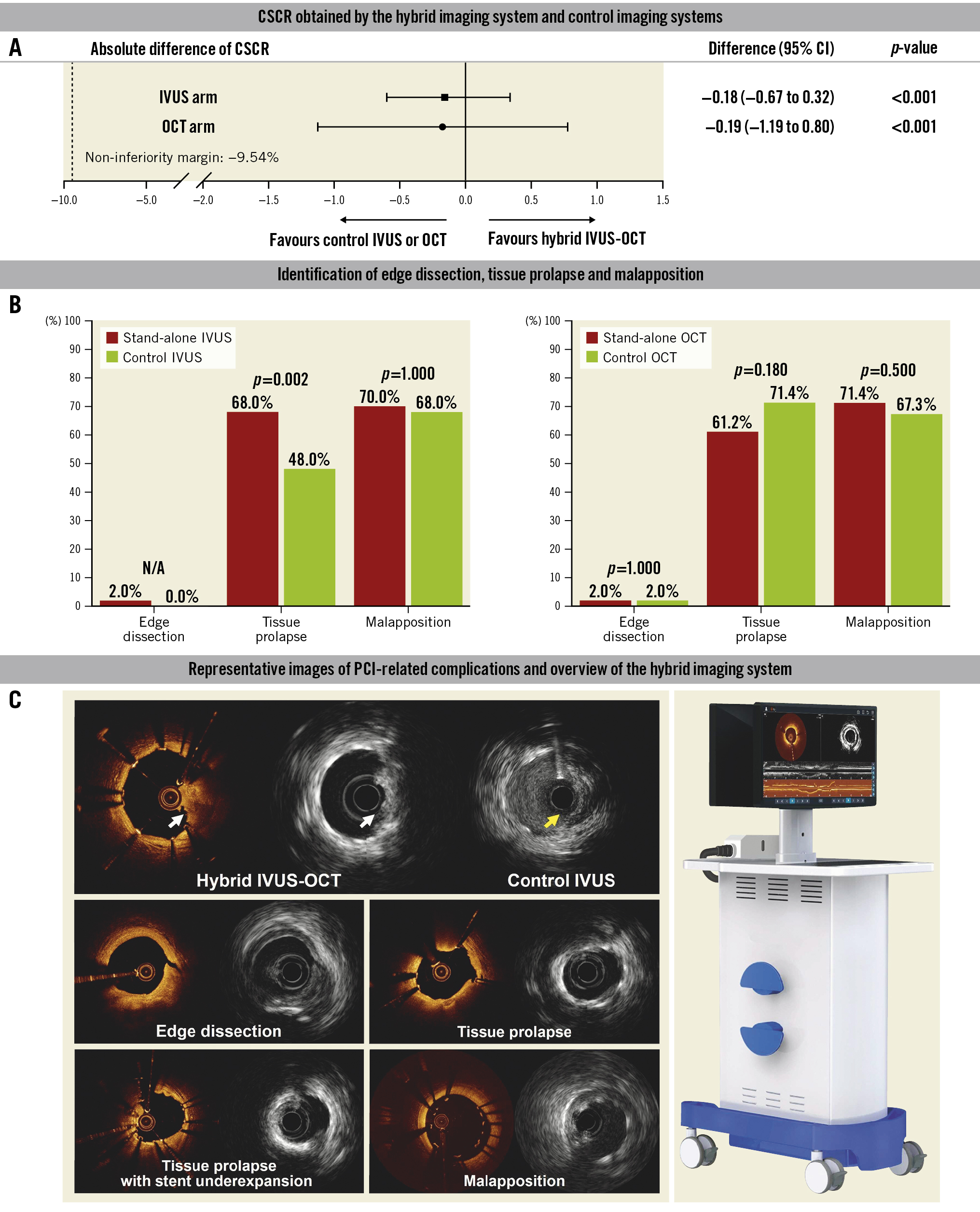
Central illustration. The main results of the PANOVISION trial. A) Each imaging modality of the hybrid IVUS-OCT demonstrated non-inferiority regarding the clear stent capture rate compared with control IVUS or control OCT. B) The proportion and difference in identification of edge dissection, tissue prolapse and malapposition between each imaging modality of the hybrid system and the control device are shown in this panel. C) Tissue prolapse was detected by stand-alone IVUS and coregistered OCT (white arrow), whereas it was indiscernible by control IVUS (yellow arrow). The representative coregistered images of edge dissection, tissue protrusion (with or without underexpansion), malapposition and overview of the hybrid imaging system are also shown in this panel. CI: confidence interval; CSCR: clear stent capture rate; IVUS: intravascular ultrasound; OCT: optical coherence tomography; PCI: percutaneous coronary intervention
Table 1. Specifications of the PANOVISION hybrid IVUS-OCT imaging system.
| o | IVUS | OCT |
|---|---|---|
| Frequency or wavelength | 35 MHz | 1,310 nm |
| Axial resolution | 100 μm | 20 μm |
| Lateral resolution | 150 μm | 30 μm |
| Frame rate (single imaging) | 20 or 100 fps | 20, 100, or 200 fps |
| Frame rate (hybrid imaging) | 20, 100, or 200 fps | |
| Pullback speed, mm/s | 1 and 4 at 20 fps; 20 at 20 fps or 100 fps | 20 at 20 fps or 100 fps; 40 at 20 fps or 200 fps |
| Hybrid use: 20 at 20 fps or 100 fps; 40 at 20 fps or 200 fps | ||
| Pullback length | Up to 150 mm | |
| Fusion imaging | Yes | |
| 3D reconstruction | No | Yes |
| 3D: three-dimensional; fps: frames per second; IVUS: intravascular ultrasound; OCT: optical coherence tomography | ||
Acknowledgements
The authors thank all the investigators, technicians, and participating staff members in this study.
Funding
This study was supported by the Natural Science Foundation of China (81827806 and 62135002 to B.Y., 81722025 and 82061130223 to H.J.), Natural Science Foundation of Hei Longjiang Province (TD2020H001 to B.Y.) and Panovision Co. Ltd., Beijing, People’s Republic of China.
Conflict of interest statement
The authors have no conflicts of interest declare.

