
Abstract
Aims: Irregular protrusion identified by optical coherence tomography (OCT) immediately following stent implantation has been reported as an independent predictor for adverse cardiac events at one year. However, baseline characteristics for the development of irregular protrusion are unknown. The aim of the study was to identify predictors for irregular protrusion following stent implantation.
Methods and results: A total of 900 lesions in 786 patients who had post-stent OCT imaging were analysed. The patients were divided into two groups: those with (484 lesions, 456 patients) and those without (416 lesions, 330 patients) irregular protrusion. Baseline characteristics (n=786) as well as pre-stent angiogram (n=786), intravascular ultrasound (IVUS; n=31) and OCT (n=159) findings were compared between the two groups. Multivariate analysis showed that patients with irregular protrusion had higher low-density lipoprotein (LDL) cholesterol levels and lesions with greater stent length, greater percent atheroma volume on IVUS, greater lipid content and a higher prevalence of thrombus on OCT before stent implantation.
Conclusions: Irregular protrusion was found more frequently in patients with higher LDL cholesterol levels. In addition, lesions treated with longer stents, with greater plaque burden, larger lipid content and a higher prevalence of thrombus were identified as predictors for irregular protrusion following stent implantation.
Abbreviations
AMI: acute myocardial infarction
EEM: external elastic membrane
FCT: fibrous cap thickness
IVUS: intravascular ultrasound
LDL: low-density lipoprotein
MACE: major adverse cardiac events
MLD: minimal lumen diameter
MSA: minimal stent area
OCT: optical coherence tomography
PAV: percent atheroma volume
PCI: percutaneous coronary intervention
TCFA: thin-cap fibroatheroma
TIMI: Thrombolysis In Myocardial Infarction
Introduction
Although optical coherence tomography (OCT) has been increasingly used during percutaneous coronary intervention (PCI), its value to improve clinical outcome is limited. Recently, our group reported the OCT predictors for major adverse cardiac events (MACE): small minimal stent area (MSA) and irregular protrusion1. Small MSA can be measured by intravascular ultrasound (IVUS). However, irregular protrusion, which is defined as a protrusion of material with an irregular surface into the lumen of the vessel between stent struts can only be identified by OCT due to its high spatial resolution. Irregular protrusion was present in more than a half of patients and the odds ratio for MACE in one year was 2.6. Identifying predictors for irregular protrusion may help us to develop strategies to avoid this deep vascular injury and to reduce MACE. The aim of this study was to identify the predictors for irregular protrusion after stent implantation.
Methods
STUDY POPULATION
Subjects were selected from the Massachusetts General Hospital (MGH) OCT Registry. The MGH OCT Registry is an ongoing international multicentre registry of patients who underwent OCT of the coronary arteries and involves 20 sites across six countries (http://www. clinicaltrials.gov: NCT01110538). For this study, we used the previously published database1. A total of 900 lesions from 786 patients were analysed. Of 900 lesions, 159 had preprocedural OCT and 31 lesions had preprocedural IVUS imaging. The registry was approved by the institutional review board at each participating site. Written informed consent was obtained from all the patients before enrolment.
IMAGE ACQUISITION AND ANALYSIS
Coronary angiograms performed before intervention and at the end of the procedure were analysed. Coronary angiograms were analysed by offline quantitative coronary angiography software (CAAS, version 5. 10. 1; Pie Medical Imaging BV, Maastricht, the Netherlands). The reference diameter, minimal lumen diameter (MLD), % diameter stenosis, and lesion length at baseline and post procedure were measured.
In IVUS measurements, external elastic membrane (EEM) and lumen were traced manually using planimetry software (EchoPlaque 4.0; INDEC Systems, Santa Clara, CA, USA) in accordance with guidelines for IVUS from the American College of Cardiology Clinical Expert Consensus Document on Standards for Acquisition, Measurement and Reporting of Intravascular Ultrasound Studies2. The percent atheroma volume (PAV) was calculated as: PAV=100 [Σ(EEM area–lumen area)/Σ(EEM area)].
A frequency-domain OCT system (FD-OCT; St. Jude Medical, St. Paul, MN, USA) and a time-domain OCT system (TD-OCT; LightLab Imaging Inc, Westford, MA, USA) were used. The technique of intracoronary OCT imaging has been previously described3. All images were de-identified, digitally stored, and submitted to the Massachusetts General Hospital (Boston, MA, USA). Pre-stent OCT images were analysed at the corresponding segment of the vessel. All OCT images were analysed at every 1 mm interval.
Lipid-rich plaque was defined as a plaque with maximum lipid arc >90 degrees. Lipid index was calculated as mean lipid arc multiplied by lipid length4. In addition, the presence or absence of thin-cap fibroatheroma (TCFA), macrophage accumulation, cholesterol crystal, and microvessel was noted. Fibrous cap thickness (FCT) was measured at the thinnest part three times, and the average value was calculated. TCFA was defined as a lipid-rich plaque with fibrous cap thickness ≤65 µm5. Macrophage accumulations on the OCT images were identified by an increased signal intensity within the plaque, accompanied by heterogeneous back shadows6. Cholesterol crystals were characterised as a high-intensity thin linear region beside a lipid core. Microvessel was defined as a small vesicular or tubular structure with a diameter of 50 to 300 µm. Calcification was also recorded, when an area with low backscatter and a sharp border was identified inside a plaque7. Irregular protrusion was defined as a protrusion of material with an irregular surface into the lumen between stent struts. OCT images were analysed by two investigators who were blinded to the subject’s information.
Statistical analysis
Categorical variables are presented as counts and percentages (%). The normality of distributions of continuous variables was examined by using the Kolmogorov-Smirnov test. The mean with standard deviation is reported for normally distributed data, and the median (25th-75th percentiles) is reported when data are not normally distributed. Fisher’s exact test or chi-square test was used for categorical variables, and the Student’s t-test or Mann-Whitney U test was used to compare continuous variables. Multivariable logistic regression analyses were performed to determine the independent predictors for irregular protrusion. Univariate analysis was performed for all variables in the study, then the variables with p<0.05 were included in the multivariable models for adjusted analysis. Adjusted multivariable models were performed separately for patients’ characteristics, and angiography and OCT analysis. A generalised estimating equations approach was applied to fit the models in order to take into account the within-subject correlation of the outcomes due to multiple lesions analysed within a single patient. All statistical analyses were performed with SPSS, Version 23.0 (IBM Corp., Armonk, NY, USA).
Results
BASELINE CHARACTERISTICS
Among 900 lesions treated with stenting, irregular protrusion was observed in 484 lesions (53.8%), 456 patients. Baseline characteristics are shown in Table 1. Patients with irregular protrusion had higher body mass index and less frequent prior PCI. Those patients more frequently presented with acute myocardial infarction (AMI), and had higher total and low-density lipoprotein (LDL) cholesterol levels. In adjusted analysis, the only significant baseline independent predictor for irregular protrusion was a high LDL cholesterol level (Figure 1).
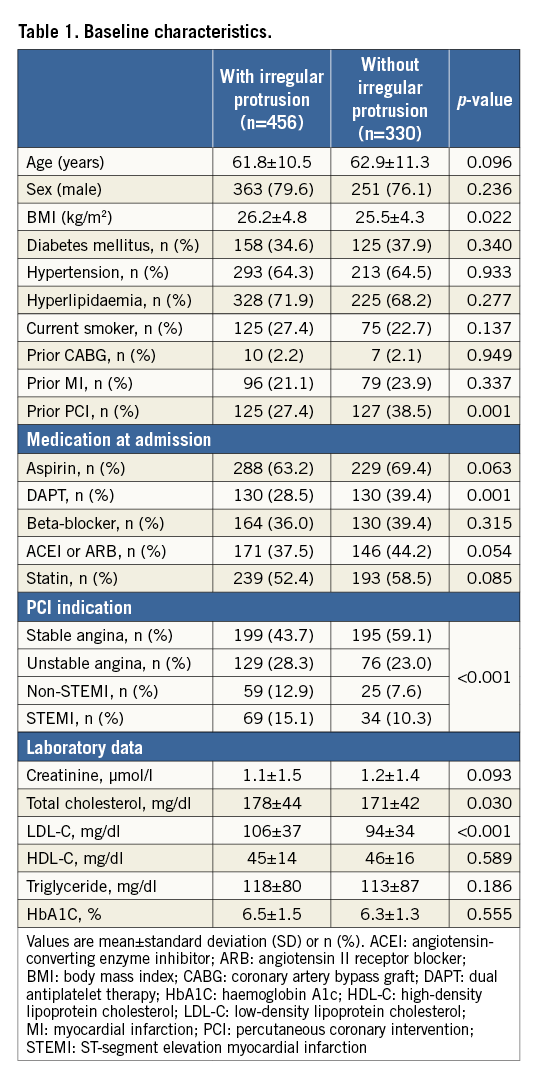
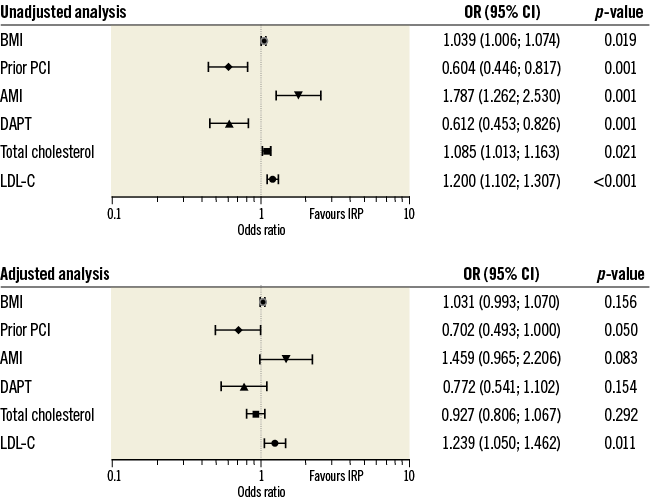
Figure 1. Baseline predictors for irregular protrusion. Patient-level analyses were performed in 786 patients with clinical data. AMI: acute myocardial infarction; BMI: body mass index; CI: confidence interval; DAPT: dual antiplatelet therapy; IRP: irregular protrusion; LDL-C: low-density lipoprotein cholesterol; OR: odds ratio; PCI: percutaneous coronary intervention. OR for BMI were calculated for each 1 kg/m2 increase. OR for total cholesterol and LDL-C were calculated for each 20 mg/dl increase.
ANGIOGRAPHY
Baseline angiography and procedural findings are shown in Table 2. Lesions with irregular protrusion had a longer lesion as well as stent length, smaller minimal lumen diameter, and greater reference diameter, consistent with greater plaque burden. Lower Thrombolysis In Myocardial Infarction (TIMI) flow grade requiring more frequent use of thrombectomy and greater need for predilatation indicated acute and severe culprit lesions. Greater post-stent residual diameter stenosis is also consistent with greater plaque burden. Interestingly, those lesions treated with overlapping stents had a lower incidence of irregular protrusion. Adjusted analysis showed that stent length, reference diameter, and post-PCI % stenosis were independent positive predictors and overlapping stents a negative predictor for irregular protrusion (Figure 2). As a linear relationship occurred between lesion length and stent length, only stent length was included in adjusted analysis as it was the stronger predictor.
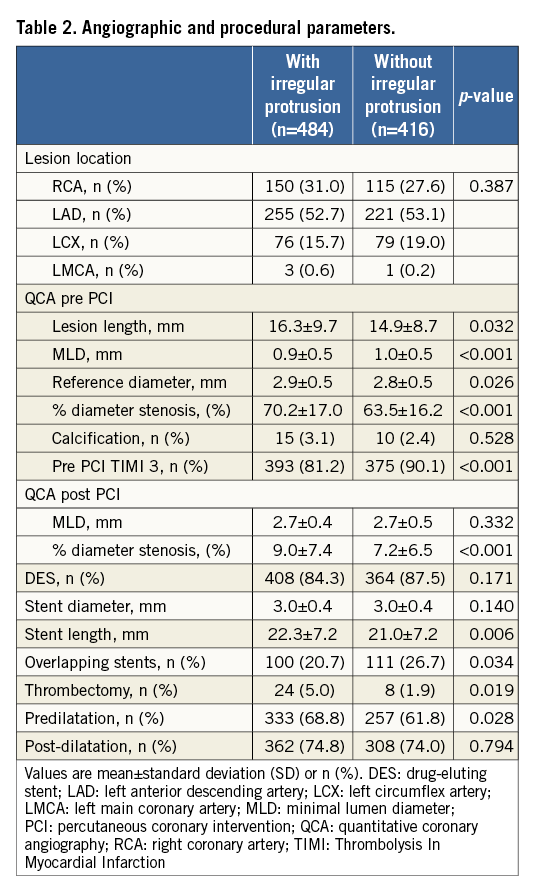
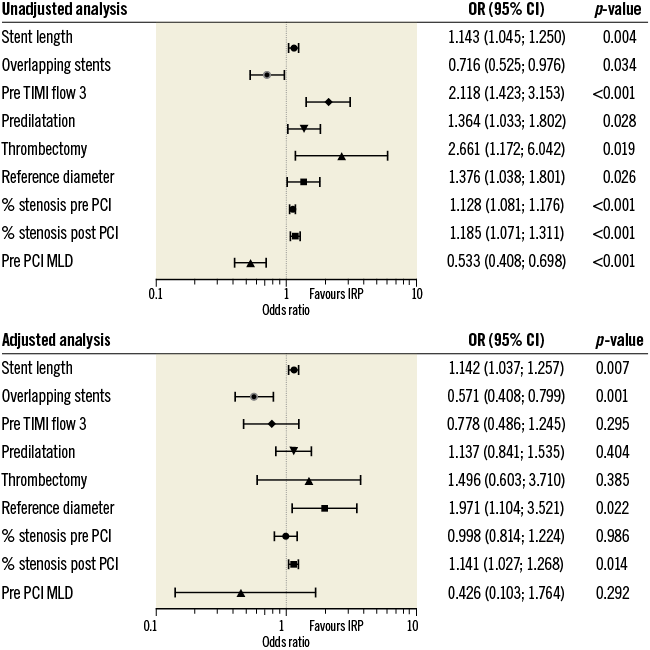
Figure 2. Angiographic and procedural predictors for irregular protrusion. Lesion-level analyses were performed in 900 lesions with angiography data. CI: confidence interval; IRP: irregular protrusion; MLD: minimal lumen diameter; OR: odds ratio; PCI: percutaneous coronary intervention; TIMI: Thrombolysis In Myocardial Infarction. OR for stent length were calculated for each 5 mm increase. OR for % stenosis pre and post PCI were calculated for each 5% increase. OR for MLD and reference diameter were calculated for each 1 mm increase.
IVUS ANALYSIS
IVUS analysis of 31 patients revealed that PAV (67.1±5.7% vs. 58.4±6.3%; p=0.002) was greater in lesions with irregular protrusion. MLA tended to be smaller in lesions with irregular protrusion (2.2±0.6 vs. 2.9±0.6 mm2), although not statistically significant (p=0.056).
OCT
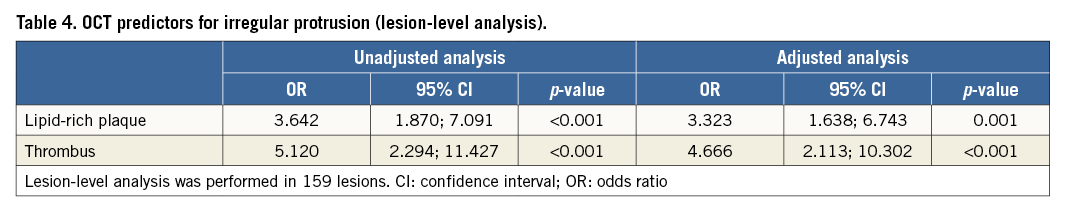
Among 900 lesions, 159 had pre-PCI OCT. Pre-procedure OCT analysis showed that the lesions with irregular protrusion had a higher prevalence of lipid-rich plaque and thrombus (Table 3, Figure 3). In lipid-rich plaques, lipid index and lipid arc were higher in lesions with irregular protrusion (Table 3). The presence of lipid-rich plaque, and thrombus were independent factors for irregular protrusion in adjusted analysis (Table 4). Lipid index, normalised lipid index, maximal lipid arc and mean lipid arc were excluded from the adjusted analysis as a linear relationship occurred between them.
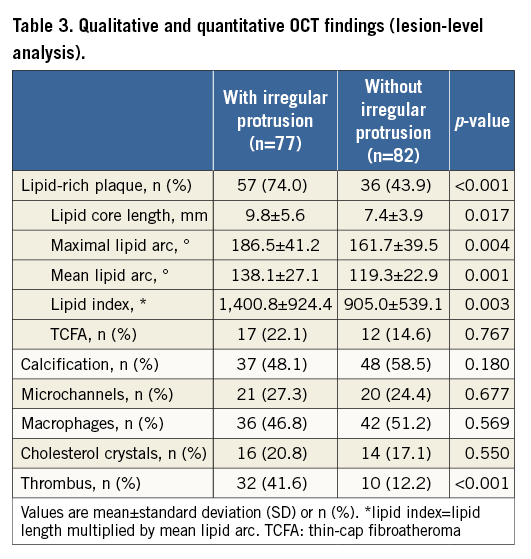
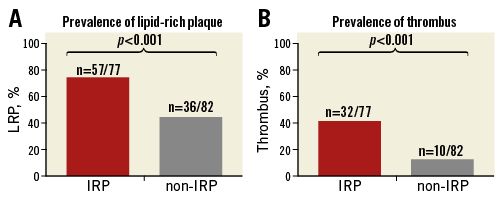
Figure 3. Prevalence of lipid-rich plaque and thrombus before percutaneous coronary intervention in lesions with irregular protrusion (IRP) compared with lesions without irregular protrusion (non-IRP). A) Out of 77 lesions with irregular protrusion, 57 (74.0%) had lipid-rich plaque (LRP) on optical coherence tomography before stenting. LRP was observed in 36 (43.9%) of 82 lesions without irregular protrusion (non-IRP), (p<0.001). B) Compared with 32 (41.6%) lesions with thrombus before stenting in the IRP group, only 10 (12.2%) lesions had thrombus before stenting in the non-IRP group (p<0.001).
Discussion
To our knowledge, this study is the first report on the predictors for irregular protrusion. The main findings of this study are that: LDL-cholesterol levels, stent length, PAV, lipid-rich plaque and thrombus are the independent predictors for irregular protrusion.
The unsurpassed spatial resolution of OCT offers unique opportunities for the in vivo assessment of the vessel wall injury caused by stent deployment. The importance of our findings is reflected in a target lesion revascularisation rate of 2.2% during one-year follow-up in the absence of OCT predictors of MACE, small MSA and irregular protrusion1. The presence of irregular protrusion increased the risk of MACE by 150%. In this study, we sought to find predictors for irregular protrusion using modalities such as laboratory test, angiography, OCT and IVUS.
PREDICTORS FOR IRREGULAR PROTRUSION
Although irregular protrusion was more frequently found in patients who presented with AMI, AMI was not found to be an independent predictor in a multivariable model. OCT studies showed that patients with AMI had a higher prevalence of lipid-rich plaque and thrombus8,9. Therefore, it is expected that patients with AMI would have a higher risk for developing irregular protrusion after stenting. Interestingly, prior PCI was a protective factor for irregular protrusion. Patients after PCI were probably treated with more aggressive statin therapy as well as a change of lifestyle, which might have contributed to stabilisation of plaques in the whole coronary vasculature10.
Among baseline demographic and laboratory variables, LDL-cholesterol levels were found to be the only independent predictor for irregular protrusion. Sugiyama et al also showed a correlation between LDL-cholesterol levels and volume of plaque prolapse, although they did not report detailed morphologic characteristics of prolapse11. There are possible explanations linking LDL cholesterol to irregular protrusion. An increased blood concentration of LDL cholesterol is a known factor for atherosclerosis. Several studies showed that LDL-cholesterol levels are positively correlated with plaque burden12-14. Large plaque burden may prohibit full expansion of stents and increase the risk of tissue protrusion between struts. More importantly, higher LDL-cholesterol levels are positively correlated with major cardiac events and plaque vulnerability itself15-17. When a stent is placed over a plaque with a large necrotic core, it is conceivable that fibrous cap overlying lipid would be easily disrupted and protrude between stent struts, resulting in irregular protrusion.
ANGIOGRAPHIC PREDICTORS FOR IRREGULAR PROTRUSION
The longer length of the stent was positively correlated with irregular protrusion. In the current era of drug-eluting stents, full coverage of diseased segments by stents is generally attempted. Longer stents are used for longer lesions, which contain a higher plaque burden. Moreover, longer stenting increases the incidence of vessel wall injury due to uneven distribution of inflation pressure along the length of the stent18. The longer the stent, the greater the difference between proximal and distal reference area - higher stent to artery ratio which occurs at the distal fragment of a stent may lead to a higher penetration of stent struts into the vessel19. When one stent is placed on another, different forces are applied to the vessel wall when compared to stenting the lesion without previous stents. Moreover, two layers of metal may prevent tissue from protruding. Thus, overlapping stents may be a protective factor against irregular protrusion. The reference diameter was higher in lesions with irregular protrusion. In lesions with a larger reference diameter and a greater pre-PCI stenosis, stent implantation may result in greater vascular injury and a higher incidence of irregular protrusion. We showed that post-PCI stenosis was an independent predictor for irregular protrusion. The difference in stenosis between groups with and without irregular protrusion was only 2%. Irregular protrusion itself and thrombus associated with it might have caused the smaller lumen area.
PLAQUE BURDEN ON IVUS
More severe lesions with greater plaque burden may require more aggressive balloon dilatation in order to achieve optimal final results20. The aggressive dilatation required to obtain adequate lumen dimension will increase the risk of deeper vessel wall injury, resulting in irregular protrusion. In our study, PAV was 10% greater in lesions with irregular protrusion compared to those without protrusion. This result is consistent with the recent IVUS study by Qiu et al21. By analysing 893 pre-PCI lesions they showed that patients with tissue protrusion after coronary artery stenting more often presented with AMI. Moreover, stented lesions had greater plaque burden and more plaque vulnerability. In their study, tissue protrusion was not associated with worse clinical outcomes. It is important to note that they did not distinguish irregular protrusion from other types of protrusion due to the limited resolution of IVUS. Of note, greater plaque burden is associated with higher plaque vulnerability, including larger lipid content21,22.
LIPID-RICH PLAQUE ON OCT
Vessel wall injury during coronary intervention is caused by ballooning or stenting23. The severity of injury depends on various factors such as plaque burden, underlying plaque morphology, stent or balloon type, stent length or balloon inflation pressure. Lipid-rich plaque covered by thin fibrous cap has decreased maximal stress that it can withstand, and is therefore prone to disruption. Farb et al showed that plaques with large lipid core areas and greater percentage of the plaques occupied by a lipid core were more likely to be penetrated by stent struts24. An OCT study from Sugiyama et al divided plaque prolapse volumes into tertiles and found that the highest tertile was associated with a higher prevalence of lipid-rich plaque, as well as greater lipid index11. Though their study did not evaluate the quality of plaque prolapse, these findings are in line with our study in which lipid-rich plaque was an independent predictor for post-stent irregular protrusion. We showed that the lesions with irregular protrusion had an almost two times higher prevalence of lipid-rich plaque than those without. Moreover, the lipid index, which corresponds to the size of lipid in the plaque, was statistically higher in the irregular protrusion group.
THROMBUS ON OCT
Adjusted logistic regression analysis demonstrated that lesions with thrombus before stent implantation had a four times higher risk of irregular protrusion after stenting. The presence of thrombus before PCI may be seen at an AMI culprit lesion. A recent meta-analysis showed that AMI is associated with a high frequency of plaque rupture25. Ruptured plaque releases thrombogenic substrates and accelerates thrombosis26. It is not always easy to distinguish thrombus from protruding material. In our study, when distinction between tissue protrusion and thrombus was not clear, we included it in the irregular protrusion category. Moreover, the lesions with plaque rupture are correlated with lipid plaque and greater plaque burden27. Since the lesions with thrombus have a large lipid core, such lesions have a higher risk of irregular protrusion than those without.
CLINICAL IMPACT
Our study has several clinical implications. First, more aggressive LDL cholesterol-lowering therapy before stenting may reduce the incidence of irregular protrusion. Second, when implanting long stents in lesions with great plaque burden and lipid content, post-stent OCT may be particularly helpful to identify irregular protrusion. Although no effective treatment is known to reduce future cardiac events in the presence of irregular protrusion, preliminary data suggest that additional balloon inflation may reduce the incidence of final irregular protrusion. When large thrombus is visualised, more aggressive manual thrombectomy may reduce the incidence of irregular protrusion.
Study limitations
Several limitations should be noted. First, although data were collected prospectively, this study was a retrospective analysis. Though all patients undergoing PCI at participating sites were eligible for inclusion in the registry, OCT was performed at the operator’s discretion and therefore there was a potential selection bias. Second, both FD-OCT and TD-OCT were used. Although the axial resolution of TD-OCT is lower than that of FD-OCT, it is unlikely that this difference influenced the results of this study. Third, irregular protrusion has not yet been validated by histology. Fourth, only 18% of all lesions were assessed by OCT before stenting. Finally, the number of patients with baseline IVUS was small.
Conclusions
Predictors for irregular protrusion include high LDL-cholesterol level, stent length, greater plaque burden, lipid-rich plaques and thrombus. They may be associated with deep vascular injury during stent implantation which then manifests as irregular protrusion. Whether modifying these predictors will decrease the incidence of irregular protrusion remains to be studied.
| Impact on daily practice Deep vessel wall injury manifests as irregular protrusion. Patients with lesions with irregular protrusion are in greater danger of adverse cardiac events in the future. Knowing the predictors for irregular protrusion may allow a decrease in the rate of cardiac events by reducing the incidence of deep vessel wall injury and therefore the incidence of irregular protrusion. |
Acknowledgements
The authors thank all the investigators and all supporting staff. I. Jang’s research was supported by “Mr and Mrs Michael and Kathryn Park”, and by “Mrs and Mr Gill and Allan Gray.” T. Zanchin is supported by the Swiss National Science Foundation (grant number 323530_171146).
Conflict of interest statement
I. Jang has received a fellowship grant and consulting fees from St. Jude Medical. The other authors have no conflicts of interest to declare.

