Abstract
Aims: To investigate the incidence of cardiac events in octogenarians who underwent percutaneous coronary intervention (PCI) with stenting, as well as to evaluate the efficacy and safety of drug-eluting stents (DES) in this population.
Methods and results: The study included 6,129 consecutive patients who underwent PCI with stenting from 2000 to 2005 in our centre, of whom 291 (4.7%) were octogenarians. After adjusting for confounders, age ≥80 years appeared a significant predictor of high mortality at 30 days (adjusted hazard ratio [aHR] 1.92, 95% CI 1.23-3.01), and four years (aHR 2.25, 95% CI 1.77-2.85). No differences were seen with respect to incident myocardial infarction (MI), but target lesion (63.2 vs. 32.6 per 1,000 person-years at one year and 27.9 vs. 16.6 per 1,000 person-years at four years) and vessel (83.1 vs. 52.9 per 1,000 person-years at one year and 37.7 vs. 25.0 per 1,000 person-years at four years) revascularisation rates were lower in octogenarians. When comparing DES with bare metal stents (BMS) in octogenarians, mortality and MI rates were comparable, but there was a significantly lower incidence of target lesion revascularisation at one- (9.5 vs. 0.6 per 1,000 person-years, aHR 0.07, 95% CI 0.01-0.57) and four-year (3.4 vs. 0.7 per 1,000 person-years, aHR 0.16, 95% CI 0.04-0.59) follow-up in patients who received a DES.
Conclusions: Octogenarians undergoing PCI with stenting have an increased mortality risk, whereas the rates of repeat revascularisation in octogenarians are lower. This study suggests that the benefit of DES in reducing revascularisation rates is extended to elderly patients.
Introduction
The worldwide phenomenon of the ageing of the population has resulted in an increase in the number of elderly patients seeking treatment for cardiovascular disease. Recent reports on temporal trends in acute myocardial infarction (MI) showed an increase in the average age of patients1-3. Elderly subjects are characterised by a high prevalence of coronary artery disease (CAD) but also by a worse prognosis following cardiac events1.
Despite the advent of new therapeutic devices and adjunctive treatments for CAD, management of elderly patients remains a major therapeutic challenge. Many elderly patients present with absence of chest pain, atypical symptoms, abnormal baseline electrocardiograms (ECG) or comorbidities that may all contribute to clinical uncertainty and late referral for revascularisation. The higher incidence of side effects and complications, in addition to paucity of information regarding the safety and efficacy of therapies in elderly people, result in underutilisation of medical therapy and early invasive care, even in patients with no apparent contraindications1,3,4. Indeed, elderly patients remain highly under-represented in the published literature. Most clinical trials that allow the inclusion of the elderly, exclude patients aged 80 years or older, as well as elderly subjects with significant comorbidities. Obviously, these design features limit the translation of the trial results to routine clinical practice1,5.
There is a lack of information on clinical outcomes in octogenarians undergoing percutaneous coronary intervention (PCI) with intracoronary stenting. In particular, little data is available on the short- and long-term results after the use of drug-eluting stents (DES) as compared to bare metal stents (BMS) in this clinically relevant cohort. This study, which is based on a large series of consecutive and unselected PCI patients with systematic follow-up, aims to provide more insight into the incidence of cardiac events in octogenarians up to four years after the intervention. We also studied the effectiveness of DES in reducing this incidence.
Methods
Study design and patient selection
This study is based on a prospective cohort study that includes all consecutive patients (stable CAD and acute coronary syndrome patients, including unstable angina, ST segment elevation MI and non-ST segment elevation MI) who underwent PCI at Erasmus MC, Rotterdam, The Netherlands, from 1 January 2000 to 31 December 2005 (Figure 1). During this period, a total of 7,217 PCI were performed in our institution using BMS, sirolimus-eluting stents (SES) or paclitaxel-eluting stents (PES). From January 2000 until April 2002, 2,681 PCI were performed using exclusively BMS. From April 2002 until February 2003, 1,035 interventions were performed using SES (Cypher®; Cordis, Johnson & Johnson, Warren, NJ, USA) as part of the RESEARCH registry6, and from February 2003 to December 2005, 3,339 interventions using PES (TAXUS®; Boston Scientific, Natick, MA, USA) as part of the T-SEARCH registry7. The current analysis excluded the 162 patients who underwent procedures in which two different types of stents were used. Patients who underwent multiple procedures (n=784) were maintained in the cohort (BMS, SES or PES) in which they enrolled initially, even if a repeat intervention was performed using a different type of stent.
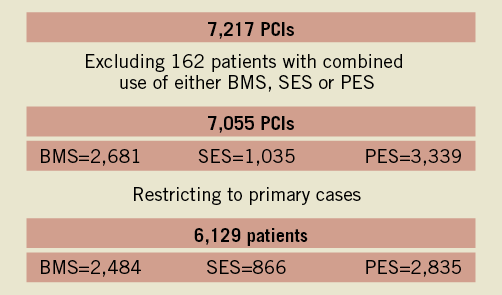
Figure 1. Flowchart depicting the number of procedures and the number of patients fulfilling the inclusion criteria. Primary cases indicates the number of patients undergoing their first intervention in the study period (2000-2005). PCI: percutaneous coronary intervention; SES: sirolimus-eluting stent; BMS: bare metal stent; PES: plaxitel-eluting stent. Modified from: Daemen J et al, EuroIntervention, 2009.
Procedures and post-intervention treatment
Patient management was in accordance with the American College of Cardiology (ACC)/American Heart Association (AHA) and European Society of Cardiology (ESC) treatment guidelines that were relevant at the moment that the patient was included. The final interventional strategy (including use of glycoprotein IIb/IIIa inhibitors, direct stenting, post-dilatation and intravascular ultrasound) was at the discretion of the interventional cardiologist, and was not specified in the research protocol.
After the procedure, all patients were advised to maintain lifelong aspirin. Clopidogrel was prescribed (75 mg/day) for at least one month for patients treated with BMS (median treatment time two [interquartile range (IQR) 1-3] months), at least three months for patients treated with SES (six [IQR 2-6] months) and at least six months for patients treated with PES (six [IQR 6-6] months).
Definitions
Cardiovascular risk factors were systematically assessed in all patients. Hypertension was defined as blood pressure ≥140 mmHg systolic or ≥90 mmHg diastolic or based on the current use of antihypertensive treatment. Diabetes was defined by treatment with antidiabetic agents, insulin or through diet. Hypercholesterolaemia was defined as fasting serum cholesterol level higher than 5.5 mmol/L or use of lipid-lowering therapy at the time of the procedure. Renal impairment was arbitrarily defined as creatinine clearance lower than 70 mL/min, estimated by the Cockroft-Gault formula8. Cardiogenic shock was defined as systolic blood pressure persistently lower than 90 mmHg or the need for inotropic support or intra-aortic balloon pump implantation in order to maintain blood pressure higher than 90 mmHg in the presence of adequate left ventricular filling pressures and systemic hypoperfusion. Clinical success was defined as anatomic (achievement of a minimum stenosis diameter reduction to less than 30% in the presence of grade 3 Thrombolysis In Myocardial Infarction [TIMI] flow) and procedural success (angiographic success without major complications: death, myocardial infarction and emergency coronary arterial bypass surgery within 48 hours after PCI) with relief of signs and symptoms of myocardial ischaemia9.
Endpoint definitions
The primary endpoint of the current analysis was all-cause mortality, which was evaluated at 30 days, one year and four years after the procedure. Secondary endpoints were non-fatal MI, target vessel revascularisation (TVR), major adverse clinical endpoints (major adverse cardiac events [MACE], defined as the composite of all-cause death, non-fatal MI or TVR), target lesion revascularisation (TLR) and stent thrombosis at one-year and four-year follow-up. PCI-related MI was diagnosed by an increase in creatine kinase-MB (CK-MB) fraction greater than three times the upper limit of normal in accordance with the universal definition of MI10. MI during follow-up was diagnosed by symptoms of ischaemia and/or new electrocardiographic changes indicative of ischaemia in association with elevation of cardiac biomarkers (CK-MB or troponine) above the upper reference limit. TVR was defined as a repeat intervention (surgical or percutaneous) to any lesion located in the same epicardial vessel treated at the index procedure in the presence of angina symptoms or signs of myocardial ischaemia. TLR was defined as treatment of a lesion within the stent or within 5 mm of the stent borders. Stent thrombosis was defined as angiographically documented thrombosis with complete occlusion (TIMI flow grade 0 or 1) or flow-limiting thrombus (TIMI flow grade 1 or 2) in a previously successfully treated artery, accompanied by symptoms of ischaemia.
Clinical follow-up
Clinical status was documented yearly until October 2007 by checking municipal civil registries. A questionnaire, consisting of queries regarding MACE, was sent to all living patients. In case of an event, the hospital medical records of our hospital or the referring institution were systematically reviewed. Most of the repeat interventions were done in our hospital, due to the fact that it is the only tertiary centre in the region. The general practitioners or the local cardiologists were called for more information if necessary. The Central Bureau of Statistics (The Hague, The Netherlands) was contacted for the cause of death, if death occurred outside the hospital.
Statistical analysis
The statistical analysis was done using SPSS statistical software release 17.0 (SPSS Inc., Chicago, IL, USA). Patients were divided in two age groups according to their age: <80 years and ≥80 years (octogenarians). Distributions of data were examined for normality by using the Kolmogorov-Smirnov test. All continuous variables had non-normal distribution, so they were expressed as median (interquartile range [IQR]) and differences between octogenarians and younger patients were evaluated using the Wilcoxon-Mann-Whitney test. Categorical variables were summarised as numbers and percentages and differences between the two groups were evaluated using the chi-square test or Fisher’s exact test.
The cumulative incidence of adverse events was estimated according to the Kaplan-Meier method, and differences between octogenarians and younger patients, as well as differences between octogenarians who were treated by BMS and DES were compared using the log-rank test. Patients lost to follow-up were considered at risk until the date of last contact, at which point they were censored.
Cox proportional hazards survival models were used to further assess the impact of age ≥80 years on mortality and other cardiac outcomes. Baseline and procedural characteristics listed in Table 1 and Table 2 were considered as potential confounders. The proportional hazards assumption was checked by a plotting log (-log[survival probability]) for age ≥80 years and potential confounders against time. Simple imputation was performed for covariates with missing data (15.5% of patients had a missing value of at least one of the measured covariates). In order to determine if age equal or higher than 80 years was an independent predictor of cardiac outcomes, a multivariable model was constructed for each cardiac endpoint by including all variables with a univariate p-value <0.50. The maximum number of covariates considered as potential confounders was one tenth of the number of events. Variables were eliminated via backward selection, and all variables with a final p-value<0.30 were retained in the model. Thus, we accepted a type I error of 30% for each variable in the model to avoid missing clinically relevant confounders. The final results were presented as adjusted hazard ratios with 95% confidence intervals.
All statistical tests were two-sided, and a p-value<0.05 was considered statistically significant.
Results
Baseline and procedural characteristics
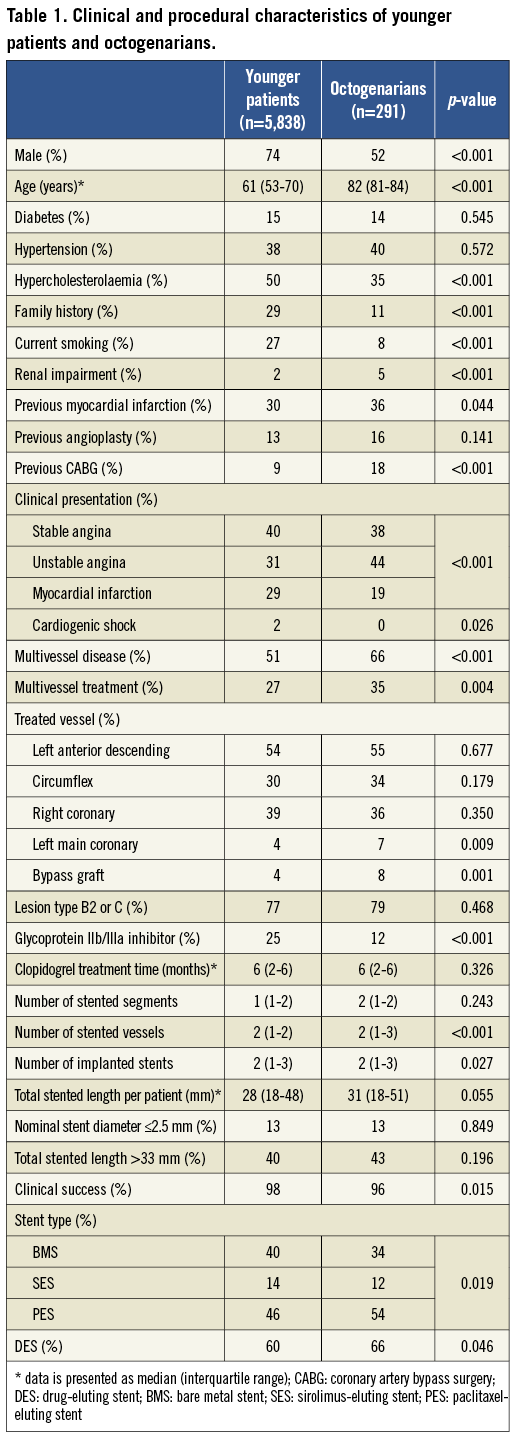
A total of 6,129 patients were included in the study, of whom 291 (4.7%) were octogenarians (median 82 [81-84] years) and 5,838 were under 80 years of age (median 61 [53-70] years). The majority of patients were male (73%). Regarding the type of stent, 40% were treated with BMS, 14% were treated with SES and 46% were treated with PES.
There were significant differences in risk factors and comorbidities between the two groups (Table 1). Younger patients were more likely to be male, smokers, and more often had hypercholesterolaemia and a family history of coronary artery disease. In contrast, octogenarians exhibited a higher prevalence of renal impairment, previous MI and previous coronary revascularisation. The percentage of patients who underwent PCI for acute MI treatment was 19% in octogenarians and 29% in the younger group (p<0.001).
Octogenarians were characterised by a more severe angiographic picture than younger patients (Table 1). They had a higher number of left main stem and bypass grafts treated, were more likely to have multivessel disease and a higher number of stented segments, and they were less likely to be treated with glycoprotein IIb/IIIa inhibitors. Procedural success was high in both groups with TIMI grade 3 flow achieved in the culprit artery in the vast majority of patients, although it was significantly lower in octogenarians.
Follow-up
There was no difference in the completeness of follow-up information between octogenarians and younger patients (99.0 vs. 98.6%, p=1.0), and the median follow-up duration was 996 (IQR 533-1,461) days in octogenarians (926 patient-years) and 1,438 (IQR 903-1,461) days in younger patients (21,388 patient-years).
Clinical outcomes
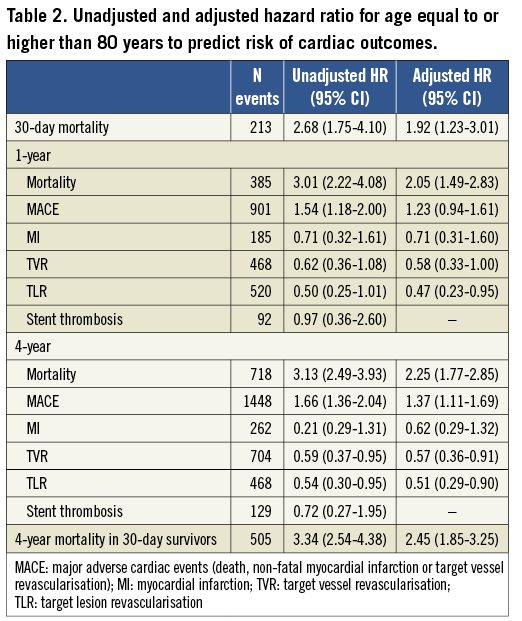
Mortality was significantly higher in octogenarians at 30 days (8.5 vs. 3.3%, p<0.001), one year (173.5 vs. 60.3 per 1,000 person-years, p<0.001) and four years (85.7 vs. 29.5 per 1,000 person-years, p<0.001) (Figure 2). Crude and adjusted hazard ratios (HR) of mortality and other outcomes are presented in Table 3. After adjusting for potential confounding factors, age equal to or higher than 80 years remained a significant predictor of higher risk of 30-day, one-year and four-year mortality. The adjusted HR of 30-day mortality (1.92 and 95% CI 1.23 to 3.01) and four-year (2.45 and 95% CI 1.85 to 3.25) mortality in 30-day survivors was similar (p=0.371), indicating that age over 80 years is a fairly constant risk factor for all-cause death in CAD subjects undergoing PCI with stenting.
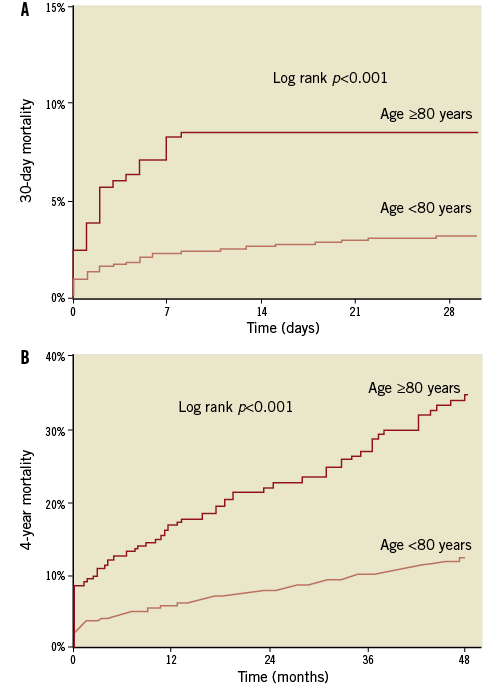
Figure 2. Cumulative incidence of 30-day (A) and 4-year (B) all-cause mortality. Age ≥80 years: n=291; age <80 years: n=5,838
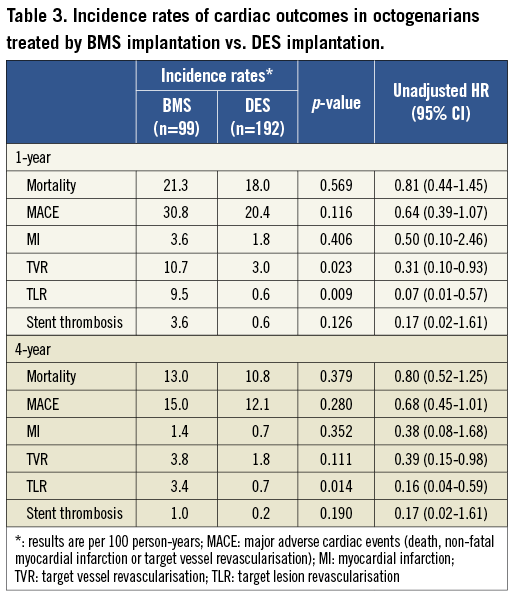
The majority of 30-day deaths were attributable to cardiac causes, but the proportion of cardiac deaths reduced over time. This reduction was more pronounced in octogenarians, in whom the importance of non-cardiac causes of death was progressively higher during long-term follow-up (Figure 3). However, this difference in the proportion of non-cardiac deaths in octogenarians when compared to the younger cohort at four-year follow-up did not reach statistical significance (72.2 vs. 57.5%, p=0.090).
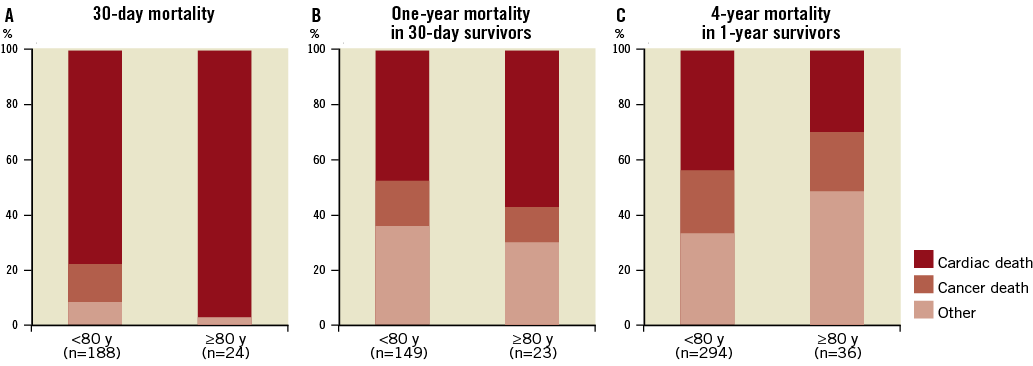
Figure 3. Causes of death at 30 days (A), 1 year (B) and 4 years (C) at 30-day survivors.
The cumulative incidence of MI was similar in younger patients and octogenarians (24.4 vs. 32.7 per 1,000 person-years at one year, p=0.423; and 9.7 vs. 14.0 per 1,000 person-years at four years, p=0.334). However, target lesion (32.6 vs. 63.2 per 1,000 person-years at one year and 16.6 vs. 27.9 per 1,000 person-years at four years) and vessel (52.9 vs. 83.1 per 1,000 person-years at one year and 25.0 vs. 37.7 per 1,000 person-years at four years) revascularisation rates were lower in octogenarians (Figure 4). The difference in the rate of repeat interventions between octogenarians and younger patients was maintained after multivariable adjustment for a range of potential confounders (Table 2).
Overall MACE was significantly higher in octogenarians (Figure 4), mostly attributable to higher mortality. The incidence of stent thrombosis was low and similar in both groups (16.3 vs. 16.1 per person-years at one year, p=0.984; 5.5 vs. 6.9 per 1,000 person-years at four years, p=0.665).
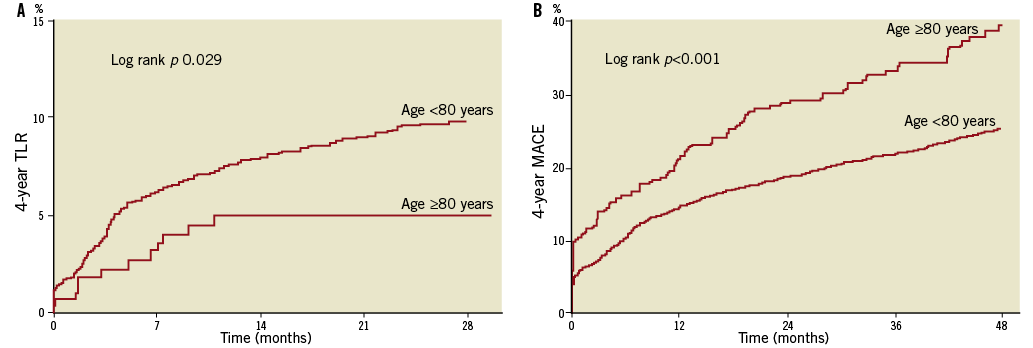
Figure 4. Cumulative incidence of 4-year target vessel revascularisation (TLR) (A) and major adverse outcomes (MACE) (B). Age ≥80 years: n=291; age <80 years: n=5,838
Subgroup analysis
Overall, mortality was significantly higher in octogenarians with acute coronary syndromes (ACS) at 30 days (12.3 vs. 2.8%, p=0.006), one year (23.5 vs. 5.5%, p<0.001) and four years (43.8 vs. 21.8%, p<0.001), when compared to octogenarians with stable CAD. However, when analysing patients with ACS versus patients with stable CAD, age equal to or higher than 80 years remained a significant predictor of a higher risk of mortality. Adjusted hazard ratios for 30-day (1.76 vs. 3.60 in patients with stable coronary artery disease, p-value=0.294), one year (2.19 vs. 1.85, p-value =0.712) and four-year mortality (3.38 vs. 1.77, p-value=0.327) were not significantly different between subgroups.
DES vs. BMS
Octogenarians who received a DES had somewhat different clinical and angiographic characteristics than those who received a BMS (Table 4). Particularly, a higher prevalence of hypercholesterolaemia and a longer clopidogrel usage, a lower frequency of glycoprotein IIb/IIIa inhibitors and a smaller average stent diameter were observed in DES patients. BMS patients had a higher frequency of previous PCI and revascularisation surgery, as well as a higher frequency of stented bypass graft and multivessel treatment.
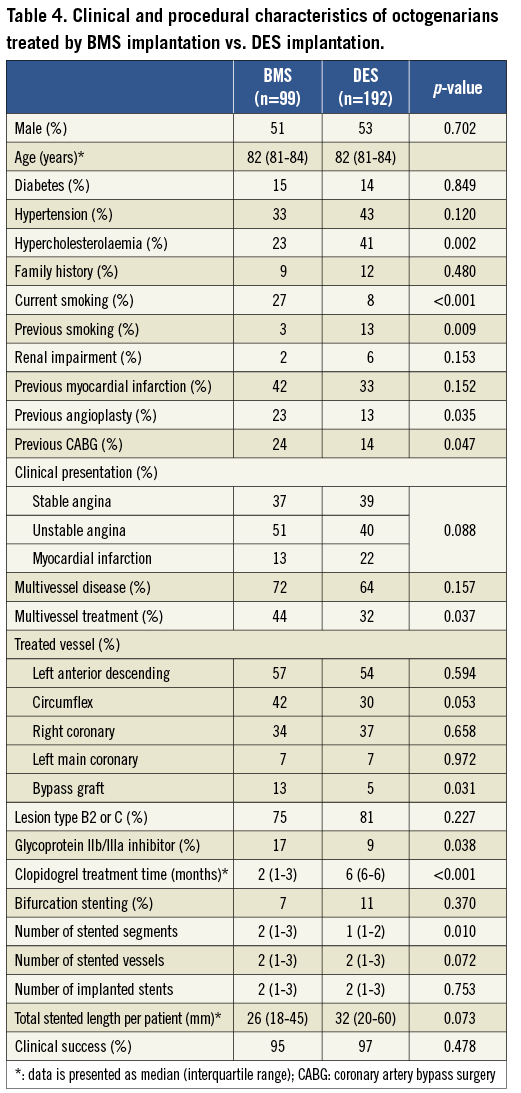
Mortality and MI rates were similar in octogenarians who received DES versus BMS (Figure 5). However, the incidence of TLR and TVR was significantly lower in octogenarians treated by DES implantation (Figure 5 and Table 3). There was also a tendency towards a lower incidence of stent thrombosis in DES patients, but the low number of events prohibits definitive conclusions. Because of this low number of events, we decided not to undertake a multivariate analysis for TLR, TVR and stent thrombosis.
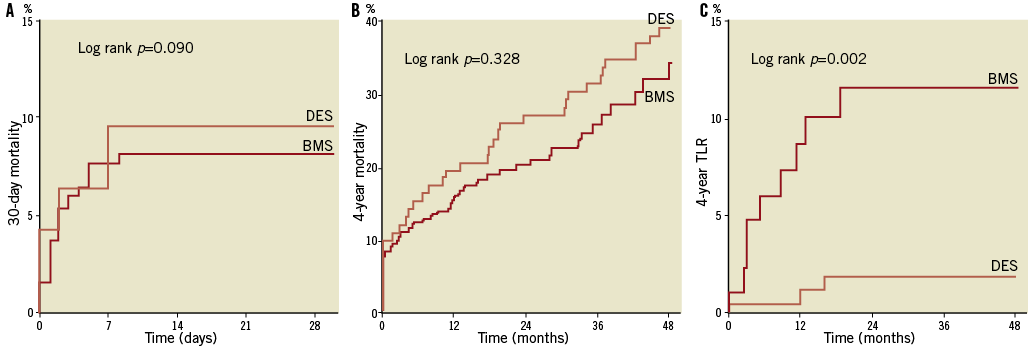
Figure 5. Cumulative incidence of 30-day (A) and 4-year (B) all-cause mortality and 4-year target lesion revascularisation (TLR) (C). Age ≥80 years: n=291; age <80 years: n=5,838
Discussion
Octogenarians undergoing PCI with stenting had higher short- and long-term mortality risks when compared with younger patients, but not an increased risk of non-fatal complications, including MI. The incidence of any MACE was also higher in octogenarians, mostly driven by the increased mortality rates. This study suggests that the benefit of DES in reducing repeat revascularisation rates, as observed in the overall population, can be extended to octogenarians, even during long-term follow-up.
Interestingly, the adjusted hazard ratio of all-cause death in relation to age was fairly constant throughout the entire follow-up period. Particularly, the hazard ratio was similar in the periprocedural phase and in the period thereafter. This observation suggests that age should be a factor to be considered when evaluating risk in patients undergoing PCI with stenting, but age in itself should not be considered a contraindication for that procedure. Indeed, it was recently shown that the relative mortality and morbidity reduction by primary PCI was independent of the patient’s age11.
There is conflicting evidence about the risk of revascularisation in the elderly. Despite all of the advances in the management of coronary artery disease regarding pharmacological treatment and revascularisation techniques, mortality rates are still high in this subgroup. The higher risk of death may be explained by several factors. It could reflect prolonged exposure to risk factors: this study observed higher prevalence of comorbidities (impaired renal function and previous myocardial infarction) and worse angiographic characteristics (higher frequency of left main coronary involvement and multivessel disease) in octogenarians. Although we adjusted for these factors in the multivariate analysis, it might not have been enough, or there might be other factors for which we have not adjusted. The increased mortality risk may also be considered a direct effect of age, as there are age-related alterations in vascular and cardiac structure and function, which have been increasingly recognised as risk factors for cardiovascular disease12. Other important determinants may be the late referral for revascularisation in ACS patients9 and underutilisation of medical therapy due to under-prescription by physicians or reduced compliance to treatment resulting from side-effects or cognitive impairment. The present study was not specifically designed to evaluate these factors, although it appeared that glycoprotein IIb/IIIa inhibitors were less frequently used in octogenarians, possibly due to concerns about bleeding in this population.
Early studies in patients undergoing PCI with BMS showed increased revascularisation rates in the elderly13, while more recent studies in the DES era observed rates similar to younger patients4,14. In the present study, however, octogenarians had lower repeat revascularisation rates and age equal to or higher than 80 years was an independent predictor of decreased risk of revascularisation, even though this group had worse angiographic characteristics. As they had higher mortality rates, this difference in revascularisation rates may result from censoring due to competing risks. Another possible explanation is that many events might have remained unrecognised, since no angiographic follow-up was performed and TLR and TVR were driven by symptoms. It is known that these patients frequently have atypical symptoms and non-diagnostic ECG, and may have a cardiac event in the setting of a period of acute or severe illness from another disease. Furthermore, these decreased rates might also be a case of confounding by indication, i.e., might be a reflection of the attending physician’s option to treat octogenarians conservatively, rather than expose them to the risks of a repeat revascularisation, regardless of whether there was or was not a formal indication for it.
There are limited data available concerning DES benefits specifically in this population15,16. Several studies have observed the benefits of DES in reducing the risk of TLR and TVR in short- and long-term follow-up in the overall population, with no significant differences in the risk of death, MI and stent thrombosis17,18. The present study suggests the benefits of DES in octogenarians with respect to a significant lower incidence of TLR and TVR when compared to patients receiving a BMS. This benefit was sustained at four years, which confirms a durable anti-stenotic effect of DES and shows that there is no “catch-up” effect even in elderly patients.
The main limitation of the present study is the fact that it is an observational study, and the enrolment was performed in a single centre. The proportion of octogenarians in the study population was lower than expected, which can be explained by a pre-selection bias: the pre-selection of the patients may be different in younger and elderly patients. The incidence of acute MI was lower than in the younger cohort (19% vs. 29%), although it was expected to be higher, and there was no elderly patient admitted with cardiogenic shock. A certain proportion of patients died before presentation to the hospital, but other cardiogenic shock patients were probably chosen to be treated conservatively due to a more restricted use of coronary angiography in this setting by the treating physicians19, even though primary PCI has been proven to be more effective20. However, our centre is a high-volume, tertiary cardiology hospital, and the study included all patients undergoing PCI in that period, except patients who underwent procedures in which two different types of stents were used (n=162). It seems unlikely that the higher mortality rates and the lower TVR and TLR rates observed in octogenarians are explained solely by improved selection.
Additionally, although the study suggests that the benefit of DES in reducing repeat revascularisation rates can be extended to octogenarians, the number of octogenarians included in the study precludes definitive conclusions about the comparison between DES and BMS. This issue should be studied more extensively. Elderly patients remain highly under-represented relative to the prevalence of their disease in clinical trials5, and the elderly included in trials are systematically different from those in the community (lower rates of cardiovascular risk factors, less comorbidity, better haemodynamics and renal function, lower progressive death rates with advancing age)1, so information from observational studies is of utmost importance to provide evidence applicable to everyday practice.
Another limitation is the fact that patients were not treated with the same protocol after hospital discharge, since this treatment was left at the discretion of the attending physician. Furthermore, the study was not powered to compare rates of infrequent adverse clinical events, such as stent thrombosis. This study also lacks routine measurement of ventricular function on admission, which has been observed to be a significant predictor of mortality21. It was not possible to evaluate if groups had an imbalance regarding this variable, as the high number of missing values precluded its inclusion in the analysis.
Conclusion
Octogenarians undergoing PCI with stenting in everyday practice have an increased short- and long-term mortality risk, and the risk of non-fatal events during long-term follow-up is comparable to younger patients. Rates of repeat revascularisation were lower in octogenarians in this study, but this may result from censoring due to competing risk, unrecognised events, or might be a reflection of the attending physician’s option to treat octogenarians conservatively. This data suggests that the benefit of DES in reducing revascularisation rates is extended to elderly patients and is persistent during long-term follow-up.
Acknowledgements
We would like to thank all the interventional cardiologists who performed the procedures during the study period: A. Dirkali, H.J. Duckers, M. van der Ent, P. de Feyter, A.L. Gaster, R.J. van Geuns, W.J. van der Giessen, P. de Jaegere, N. Kukreja, C. van Mieghem, S. Ramcharitar, E. Regar, P.W. Serruys, G. Sianos, A.G. de Vries, the cardiac catheterisation staff of the Thoraxcenter, Erasmus MC; and the staff of Havenziekenhuis, Maasstad Ziekenhuis, Sint Franciscus Gasthius, Ruwaard van Putten Ziekenhuis, Vlietland Ziekenhuis, Ijsselland Ziekenhuis hospitals in Rotterdam, The Netherlands, and Albert Schweizer Ziekenhuis in Dordrecht, The Netherlands, who collaborated in the data collection process.
Conflict of interest statement
The authors have no conflicts of interest to declare.

