A 74-year-old female with exertional breathlessness and a metal allergy underwent angiography following positive stress echocardiography. This demonstrated severe calcific disease of the left anterior descending artery (LAD) (Figure 1A). Percutaneous intervention (PCI) was offered after the failure of medical therapy.
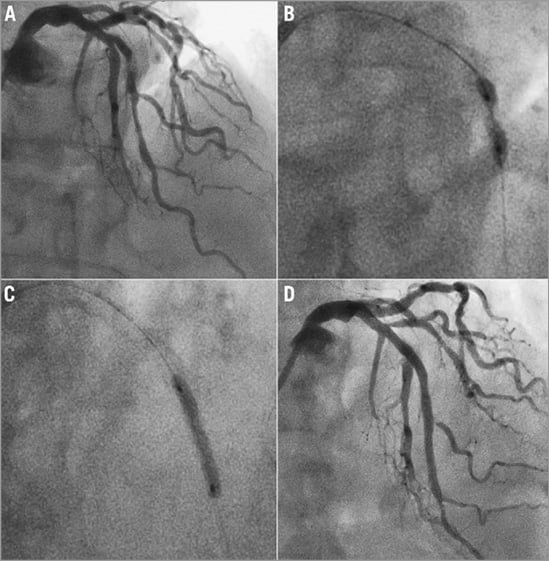
Figure 1. A) Calcified mid-LAD lesion. B) Unsuccessful predilation (significant wasting). C) Post-rotablation lesion expansion. D) Result following two overlapping Abbott BVS.
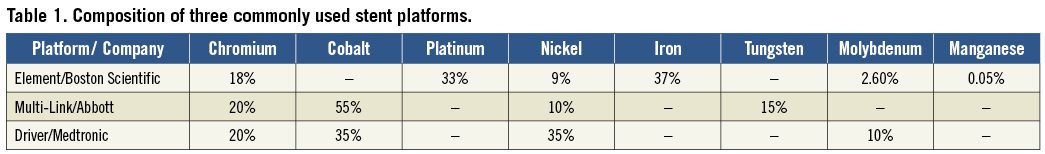
Patch testing confirmed allergy to nickel and cobalt. Most stent platforms contain alloys that include nickel (Table 1), which may contribute to restenosis in this context.
Balloon predilation was unsuccessful (Figure 1B). Therefore, rotablation with a 1.5 mm burr (Boston Scientific, Natick, MA, USA) was performed. After further predilation (Figure 1C) and QCA, overlapping 3×28 mm and 3.5×28 mm ABSORB Revision 1.1 bioabsorbable scaffolds (BVS) (Abbott Vascular, Santa Clara, CA, USA) (Figure 1C) were implanted. Postdilation with a non-compliant 3.25 mm balloon was performed (Figure 1D). The patient was symptom free at six-month review.
Although nickel allergy is not a conventional contraindication to stenting, bioabsorbable scaffolds such as the poly-L-lactide Abbott BVS now provide an alternative interventional strategy to metallic stents in this situation. Additionally, this case exemplifies the use of this technology in complex lesions.
Conflict of interest statement
The authors have no conflicts of interest to declare.

