
CASE SUMMARY
BACKGROUND: A 68-year-old male, with a history of repeat percutaneous intervention for the treatment of severe calcific stenosis of the left circumflex artery and obtuse marginal branch bifurcation, presented with stable angina at a local hospital. He was found to have recurrent in-stent restenosis of this lesion and subsequently underwent a fifth reintervention; however, adequate balloon dilatation was not achievable in spite of non-compliant and cutting balloon use. In view of continuing symptoms despite optimal medical therapy, the patient was transferred to our hospital for further management.
INVESTIGATION: Coronary angiography, intravascular ultrasound.
DIAGNOSIS: Recurrent in-stent restenosis following prior repeat complex bifurcation stenting.
MANAGEMENT: Rotational atherectomy of the multilayered stent struts, followed by successful balloon dilatation with a cutting balloon at high pressure, and treatment with a drug-coated balloon.
KEYWORDS: bifurcation, calcification, in-stent restenosis, rotational atherectomy
PRESENTATION OF THE CASE
A 68-year-old male was admitted to a local hospital with stable angina. He had previously undergone multiple percutaneous coronary interventions (PCI). Eighteen years previously, following rotational atherectomy, two bare metal stents were implanted (T stenting) to treat a severe calcific stenosis of the left circumflex artery (LCX) and obtuse marginal branch (OM) bifurcation requiring further plain old balloon angioplasty (POBA) for restenosis five months later. Six years later, a late in-stent restenosis was treated with implantation of two TAXUS™ stents (LCX: 3.0×32 mm, OM: 3.0×28 mm; Boston Scientific, Marlborough, MA, USA), utilising the crush technique. A third restenosis occurred one year later requiring the implantation of a CYPHER® stent (3.5×33 mm; Cordis Corporation, Johnson & Johnson, Warren, NJ, USA) and a fourth after four years at the same site treated with PROMUS™ stent (3.5×23 mm; Boston Scientific) implantation, resulting in a bifurcation with six stent layers (Figure 1A).
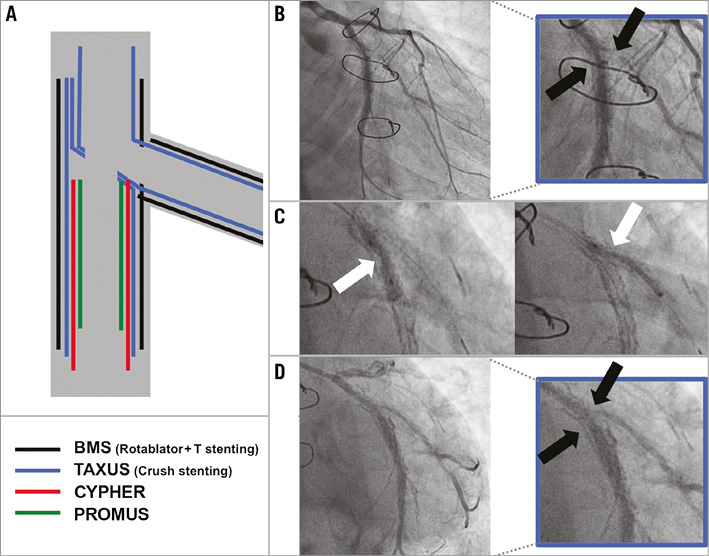
Figure 1. Illustration of recurrent in-stent restenosis following prior repeat complex bifurcation stenting. A) Schematic diagram illustrating the previously implanted stents at the bifurcation site. B) Baseline angiogram demonstrating recurrent restenosis at the LCX bifurcation (black arrows. C) Suboptimal balloon expansion at the site of LCX and OM side in bifurcation during 3.5 mm cutting balloon (LCX) and 2.5 mm balloon (OM) (white arrows) inflation. D) Final angiogram showing residual stenosis at the bifurcation site.
At the current admission, baseline angiography revealed a fifth restenosis at the site of the multilayered struts at the LCX bifurcation (Figure 1B, Moving image 1). Predilatation was initially attempted with a 3.0 mm non-compliant balloon and then a 3.5 mm cutting balloon to the LCX and a 2.5 mm balloon for the OM side branch. However, adequate balloon dilatation was not achievable (Figure 1C), with angiography confirming a significant residual stenosis (Figure 1D, Moving image 2). The procedure was therefore abandoned.
In view of continuing symptoms, despite maximal medical therapy, the patient was transferred to our hospital for further management of the LCX/OM bifurcation disease that was thought to be responsible for his angina.
How should this patient be treated?
How would I treat?
THE INVITED EXPERT’S OPINION

The authors present a patient with a fifth restenosis at the site of multilayered struts at the LCX/OM bifurcation.
Approximately 10% to 20% of patients with DES failure experience recurrent in-stent restenosis (ISR), with limited evidence about the treatment of recurrent multimetal-layered ISR, and the optimal management of these patients remains unknown1.
The management of stent underexpansion is generally straightforward, with most cases responding to simple increases in non-compliant (NC) balloon pressure or the use of larger-size balloons. The case reported here represents the extreme end of the spectrum because of multiple metal layers in the main LCX (four layers) and OM branch (two layers), following repeated use of two-stent bifurcation strategies (T-stenting and crush-stenting techniques). ISR lesions in both vessels did not respond to high-pressure NC balloon inflations.
The use of intravascular imaging (either intravascular ultrasound [IVUS] or optical coherence tomography [OCT]) is recommended when treating recurrent ISR lesions in order to identify the mechanism of restenosis and evaluate the underlying plaque morphology. The presence of multiple metallic layers occupies the space inside the vessels, preventing further dilatation, which may also be aggravated by underlying calcific plaque beneath the stent struts (the lesion was initially treated with rotational atherectomy [RA]).
If treating this lesion, I would consider partial removal of the excessive metallic layers present inside both vessels.
Longitudinal ablation of the stent (stent ablation) with RA (Rotablator®; Boston Scientific) has been described for the treatment of underexpanded or crushed stents embedded in rigid, calcified lesions, with a good acute and midterm outcome2-4. The RA burr is capable of ablating metallic stent struts and does that preferentially because of differential cutting (the abraded metallic particulate generated by stent ablation is similar in size to particles generated by calcific plaque rotablation)5. Some critical technical considerations are important prior to undertaking RA of metallic stents. The burr can “watermelon seed” or squirt through a stent without ablating the metal. If that occurs, the burr will be trapped distal to the stent and retrieval may be impossible. Also, excessive heat production can lead to thermal injury of the vessel. Therefore, using a stepped burr approach (starting with 1.5 mm burr), slow burr advancement avoiding extreme deceleration and using short periods of lesion/stent contact may be important for a successful PCI procedure3. For these reasons I would use an 8 Fr Launcher® EBU guiding catheter (Medtronic, Minneapolis, MN, USA) and femoral access in this case. Another approach for a refractory ISR lesion would be a high-energy excimer laser coronary angioplasty (ELCA) with contrast medium for disruption of calcified tissue beneath the stent struts6,7. However, this technology requires careful lesion selection and adequate training.
As a next step, repeated imaging guidance after RA is recommended before full lesion preparation with adequately sized NC balloons.
After sequential high-pressure dilatation of both vessels, we need to decide on definite treatment. According to the limited available evidence, it seems that restenotic bifurcation lesions should be approached as native bifurcation lesions when applicable, for which provisional stenting is the preferred technique8.
Considering the size and distribution of the two branches, my approach would be either a provisional stenting from the LCX to OM branch or selective OM stenting with the protective balloon in the LCX (similar to provisional T-stenting and small protrusion - TAP technique). Having accomplished this, the procedure should be finalised with kissing balloon inflation (with two NC balloons), and proximal optimisation would be performed (final POT).
Conflict of interest statement
The author has no conflicts of interest to declare.
How would I treat?
THE INVITED EXPERT’S OPINION

In recipients of the latest generation of drug-eluting stents (DES), treatment of in-stent restenosis ISR) accounts for a small number of the procedures carried out in interventional cardiology centres. This was not the case in the era of bare metal stents, especially in the setting of coronary bifurcation treatment.
The case reported here could be used to summarise the history of coronary bifurcation treatment over the past 20 years. The patient was treated for a circumflex bifurcation with two branches of similar diameter.
The initial treatment implemented was probably T stenting using BMS, starting with the marginal branch9, with a potential gap at the ostium of the marginal. Due to the occurrence of ISR, crush stenting10 was performed with TAXUS stents, starting with the mid circumflex. It was not specified whether the crush stenting procedure was followed by kissing balloon (KB) inflation, which is now considered indispensable11.
Stent implantation at the ostium of the circumflex was subsequently performed twice, with a CYPHER and a PROMUS stent, respectively. Once again, whether KB was carried out to correct carina shift is unknown. Implantation of six stents and balloon angioplasty were performed alternately. A total of six overlapping stents are present at the level of the carina. The last treatment using non-compliant (NC) balloons and cutting balloons failed to open a restenotic lesion which was diffuse in the marginal and focal in the mid circumflex opposite the carina.
IVUS is routinely used to identify factors inducing restenosis, such as small stent size, poor deployment and stent fracture. In the case reported here, the stents implanted in the circumflex and marginal branches are underexpanded.
The challenge is to achieve appropriate stent deployment and prevent the atherosclerotic material extruded by balloon angioplasty from re-entering the lumen12,13, before selecting a preventive treatment for recurrent ISR.
Rotational atherectomy (Rotablator) is a strategy implemented to ablate the struts of undeployable stents (acutely) and to correct inadequate stent deployment (chronically)3,14. Its mechanism involves metal abrasion and ablation of the restenotic material. The burr must be adapted to the stent lumen and should be exchanged for a larger burr whenever necessary. The ELCA laser can be used as an alternative. Its mechanism consists of pulverising the restenotic material whilst subsequently allowing stent expansion by balloon inflation. In current practice, the laser is activated during serum perfusion, but this can also be achieved with contrast media in order to increase the cavitation effect and soften the underlying plaque, even calcified7,15. Laser or Rotablator ablation should be completed by inflation of a NC, cutting or scoring balloon in order to achieve good angiographic results.
Preventive treatment of recurrent ISR includes implantation or use of a drug-eluting balloon (DEB)16. The latter option has been shown to be inferior to DES by several network meta-analyses, except perhaps in small-size arteries17. It was recently demonstrated that the effect of DEB was reinforced when prepared with a scoring balloon (ISAR-DESIRE IV) (TCT 2015).
In the case described here, deployment of new DES in both branches would appear to be inappropriate given the presence of the multiple stents previously implanted.
The use of a bioresorbable stent has been proposed for ISR treatment in order to avoid the addition of a metallic layer. However, stent placement over multiple layers of underexpanded stents could not only prove difficult but may also result in an excess risk of thrombosis18.
An important factor to be taken into account is the occurrence of restenosis in both branches of a coronary bifurcation. Coronary bifurcations are an anatomical and functional entity. The relationship between the diameters of the three bifurcation segments was defined according to a formula derived from Murray’s law (Finet formula: proximal diameter=(distal diameter+SB diameter)×0.678)19. Each diameter has a linear correlation with flow and myocardial mass. Blood flow distribution depends on the position of the carina.
Coronary bifurcations are proatherogenic. Atherosclerotic plaques develop opposite to the carina with low wall shear stress along the walls of the main vessel and the side branch. Following stent implantation, neointimal hyperplasia and neoatheroma develop at the same location. The best preventive method is to limit the surface of the low wall shear stress phenomenon, which is increased by the stent struts. This limitation is maximal when the initial three-diameter anatomy is reproduced20.
In the case described here, my technical strategy would be to use Rotablator via the 6 Fr transradial approach in both branches, starting with a 1.5 mm burr which could be exchanged for a 1.75 mm burr if balloon deployment proved impossible. The two distal branches would subsequently be dilated by means of KB inflation using NC balloons according to online QCA measurements (2.75 mm?). This would be followed by inflation of a short balloon of a larger size (3.25 mm?) in order to implement the proximal optimisation technique (POT) with the balloon distal marker facing the carina. Scoring balloons could be used if balloon deployment was deemed suboptimal. The procedure would be completed with the kissing deployment of two DEB of similar diameter to the preparatory balloons in the two distal branches without overlap followed by POT using a DEB or a standard balloon of suitable size.
Conflict of interest statement
The author has no conflicts of interest to declare.
How did I treat?
ACTUAL TREATMENT AND MANAGEMENT OF THE CASE
Following diagnostic angiography (Moving image 3, Figure 2A), in view of the failed attempt at predilatation with both non-compliant and cutting balloons, we proceeded to rotational atherectomy with a Rotablator 1.75 mm burr (Boston Scientific) to both the LCX and OM branches, taking every precaution to minimise the risk of the burr getting stuck within the multilayered stent struts. Successful balloon angioplasty was then performed with a 3.0 mm cutting balloon at 25 atmospheres (LCX) and a 2.5 mm cutting balloon at 22 atmospheres (OM) (Figure 2B). Finally, treatment with an Agent™ drug-coated balloon (LCX: 3.0 mm, OM: 2.5 mm; Boston Scientific) was performed, resulting in a good final angiographic result (Figure 2C, Moving image 4). Intravascular ultrasound imaging confirmed symmetric expansion, and minimal lumen areas were 4.72 mm2 (LCX) and 4.35 mm2 (OM), respectively.
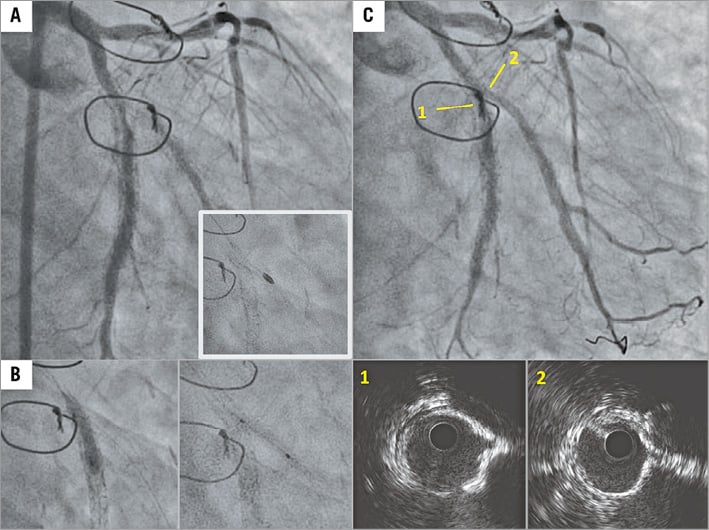
Figure 2. Baseline and final angiogram following intervention. A) Baseline angiogram showing significant stenosis at the site of bifurcation, and rotational atherectomy with 1.75 mm burr of OM (inset). B) Adequate balloon expansion after rotational atherectomy with a 3.0 mm cutting balloon in LCX and 2.5 mm non-compliant balloon in OM. C) Final angiogram and IVUS demonstrate an excellent result. Panels 1 and 2 are IVUS images at indicated segments of the angiogram.
Coronary calcification is an important contributing factor for stent underexpansion21, which increases the risks of subsequent adverse events including stent thrombosis and restenosis. When treating a non-dilatable lesion due to severe calcification, the existence of previously implanted stents makes additional intervention challenging. In such situations, an alternative option could be to use an excimer laser7 that could modify the tissue outside of the implanted stents.
There have been previous reports of stent ablation using rotational atherectomy2,3,22, although this case was further complicated by the presence of multiple stent layers at the site of a bifurcation. We here demonstrate that, utilising rotational atherectomy, a successful immediate result can be obtained, while we remain uncertain regarding long-term follow-up.
The problems related to multiple stent layers could be an issue of the past with the introduction in clinical practice of bioresorbable scaffolds.
Conflict of interest statement
A. Latib serves on the advisory board for Medtronic. The other authors have no conflicts of interest to declare.
Supplementary data
Moving image 1. Baseline angiogram at a local hospital.
Moving image 2. Final angiogram at a local hospital.
Moving image 3. Baseline angiogram at our hospital.
Moving image 4. Final angiogram at our hospital.
Supplementary data
To read the full content of this article, please download the PDF.
Baseline angiogram at a local hospital.
Final angiogram at a local hospital.
Baseline angiogram at our hospital.
Final angiogram at our hospital.

