A 75-year-old male underwent transfemoral transcatheter aortic valve implantation (TAVI) with a 23 mm SAPIEN 3 (Edwards Lifesciences Inc., Irvine, CA, USA). One year after, he was admitted to our department for pulmonary oedema. Echocardiographic evaluation evidenced low placement of the SAPIEN 3 and 3-(4)+/4+ aortic regurgitation. ECG-gated multislice computed tomography (MSCT) was performed which confirmed low placement (Moving image 1) and underexpansion of the SAPIEN 3 with a perimeter of 62 mm and diameter of 20 mm (Figure 1A-Figure 1C). After Heart Team evaluation, a new TAVI procedure was preferred with the implantation of a 26 mm CoreValve® Evolut R™ (Medtronic, Minneapolis, MN, USA). Basal aortography evidenced significant paravalvular regurgitation (Moving image 2). Haemodynamic evaluation revealed a mean aortic gradient of 25 mmHg and an aortic regurgitation index of 22. After 26 mm Evolut R implantation, post-dilatation was performed. Final aortography showed trivial paravalvular regurgitation (Moving image 3). Haemodynamic assessment evidenced a mean transaortic gradient of 4 mmHg and an aortic regurgitation index of 44. Pre-discharge ECG-gated MSCT evidenced good Evolut R expansion inside the SAPIEN 3 (Figure 1D) and echocardiography showed trivial paravalvular regurgitation (Moving image 4).
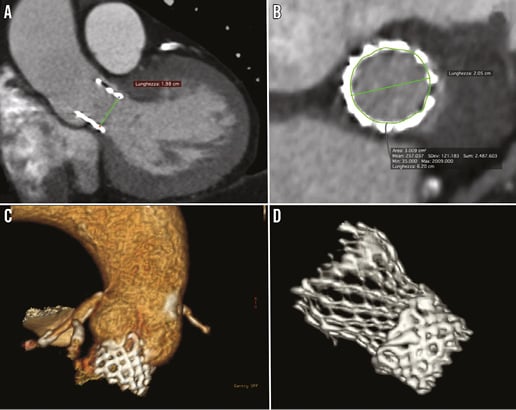
Figure 1. SAPIEN 3 ECG-gated multislice computed tomography evaluation and final results after valve-in-valve with CoreValve Evolut R. A) MSCT oblique coronal view that evidenced low SAPIEN 3 implantation and underexpansion with a valve diameter of 19.8 mm. B) Double oblique transverse reformatting of the annulus on MSCT with SAPIEN 3 area of 300 mm2 and perimeter of 62 mm. C) 3D MSCT evidenced low placement of the 23 mm SAPIEN 3 valve. D) 3D MSCT evidenced full Evolut R expansion inside the SAPIEN 3 valve.
Implantation of a CoreValve Evolut R prosthesis for the treatment of a dysfunctioning Edwards SAPIEN 3 prosthesis is feasible and is associated with a good functional result.
Conflict of interest statement
G. Bruschi is a consultant for Medtronic and Direct Flow Medical. The other authors have no conflicts of interest to declare.
Online data supplement
Moving image 1. 3D reconstruction of multislice computed tomography that evidenced low placement of the SAPIEN 3 valve.
Moving image 2. Basal aortography evidenced significant paravalvular regurgitation of the SAPIEN 3 valve.
Moving image 3. Final aortography showed trivial paravalvular regurgitation with good Evolut R expansion inside the SAPIEN 3 valve.
Supplementary data
To read the full content of this article, please download the PDF.
Moving image 1. 3D reconstruction of multislice computed tomography that evidenced low placement of the SAPIEN 3 valve.
Moving image 2. Basal aortography evidenced significant paravalvular regurgitation of the SAPIEN 3 valve.
Moving image 3. Final aortography showed trivial paravalvular regurgitation with good Evolut R expansion inside the SAPIEN 3 valve.
Moving image 4: Final aortography shown trivial para-valvular regurgitation with good Evolut R expansion inside the Sapien 3

