
Abstract
Aims: The aim of this study was to compare the hyperaemic flow and vasomotor response to endothelium-dependent stimuli between bioresorbable vascular scaffolds (BVS) and metallic everolimus-eluting stents (EES) at 13 months.
Methods and results: Seventy non-diabetic patients aiming to achieve complete revascularisation were randomised 1:1 to BVS or EES implantation. At 13 months, invasive coronary angiography was performed using intracoronary pressure and Doppler ultrasound measurements at rest and maximal hyperaemia. A vasomotor test to endothelium-dependent (acetylcholine) and independent (nitroglycerine) stimuli and optical coherence tomography (OCT) were also performed. Fifty-nine patients (30 BVS and 29 EES) underwent 13-month examination. Doppler ultrasound average peak velocity (49.0±17.5 vs 49.3±18.3 cm/sec; p=0.95), coronary blood flow (97.4±53.5 vs 88.3±46.7 ml/min; p=0.51), coronary flow reserve (2.6±0.9 vs 2.7±0.8; p=0.84) and fractional flow reserve (0.92±0.06 vs 0.94±0.04; p=0.17) were similar between the groups. The vasomotor test showed vasoconstriction response to acetylcholine in 75.6% proximal and 72.2% distal peri-scaffold segments without differences between study devices. BVS had larger in-scaffold vasoconstriction than EES (60.0% vs 27.6%; p=0.01) despite similar neointima response as assessed by OCT.
Conclusions: BVS and EES had similar microcirculatory response to hyperaemia and predominant vasoconstrictive response in the peri-scaffold segments to endothelium-dependent stimuli. However, BVS exhibited larger vasoconstriction to endothelium-dependent stimuli in the scaffold segment.
Introduction
In healthy coronary vessels, the adenosine-mediated vasodilatation of the intramyocardial microcirculation is the main regulator of the coronary flow1. However, epicardial coronary arteries also play a role in the regulation of the coronary flow. When flow changes, epicardial arteries react to maintain a given level of blood pressure at the origin of the microcirculation. This mechanism is mainly mediated by the endothelial synthesis of nitric oxide in response to local shear stress forces2. The goal of these complex processes between the different coronary segments is to match the coronary flow with the oxygen requirements of the myocardium1,2.
Obstructive coronary artery disease (CAD) is the main cause of angina3. Epicardial and intramyocardial coronary circulation present adaptive mechanisms to overcome the limited coronary flow in patients with obstructive CAD. These mechanisms, such as the emergence of collateral flow and a certain degree of compensatory microcirculatory vasodilation, are capable of adapting the coronary flow to hyperaemia, even in cases with severe coronary obstruction1,2. However, the relevance of these adaptive mechanisms and the patient’s tolerance to pain may have different clinical presentations. Current revascularisation guidelines recommend surgical or percutaneous revascularisation in patients with stable CAD when there is persistence of symptoms despite optimal medical treatment (OMT) and/or there is evidence of prognosis improvement by coronary revascularisation3. Several studies have shown greater relief of angina symptoms and better quality of life with myocardial revascularisation than with OMT alone4,5. However, in around 20-30% of patients with obstructive CAD achieving complete revascularisation, angina symptoms persist despite OMT5,6,7. Vasomotor dysfunction of the intramyocardial microcirculation and native or reactive (to stent implantation) endothelial dysfunction of the epicardial coronary arteries have been hypothesised to cause most of the cases with persistent angina7.
Bioresorbable vascular scaffolds (BVS) theoretically allow the restoration of the vasomotor response to local shear stress forces. When the scaffold loses its radial force, coronary segments treated with BVS present with geometric changes, react to pulsatile flow and present with vasomotor response to endothelium-dependent factors8,9,10,11. However, it is uncertain if the restoration of these epicardial coronary conditions is associated with better coronary flow in patients treated with BVS compared to patients treated with everolimus-eluting metallic stents (EES) at maximal hyperaemia.
The primary objective of the study was to compare the coronary blood flow (CBF; as assessed by Doppler ultrasound average peak velocity measurement) between everolimus-eluting BVS and EES at maximal hyperaemia. The secondary objective was to compare the vasomotor change of the scaffold segment to endothelium-dependent vasomotor stimuli.
Methods
STUDY DESIGN
The BVS-FLOW study was a prospective, randomised, controlled, multicentre clinical trial (NCT02738658). A total of 35 patients per group were requested to assess a difference in Doppler ultrasound average peak velocity (APV) larger than 12.0 cm/sec at maximal hyperaemia with 80% power and a two-tailed p-value of 0.05.
In order to minimise non-device-related factors of angina, microcirculatory dysfunction and endothelial dysfunction, the present study included only selected patients with obstructive CAD in whom complete angiographic revascularisation was achieved. All coronary lesions had ≥60% visually estimated stenosis suitable to be treated with a single stent of 12 to 28 mm length and 2.5 to 3.5 mm diameter. The following characteristics were exclusion criteria of the present study: incomplete revascularisation, diabetes mellitus, acute coronary syndromes with ≥5 times increase of ultra-sensitive cardiac troponin, patients with chronic total occlusions, prior myocardial infarction of the culprit artery, previous revascularisation of the culprit vessel and ejection fraction <50%. This study was approved by the local ethics committee of all participating institutions and was carried out in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients.
BASELINE PROCEDURE
Coronary angiography was performed according to standard procedures in each institution. Predilatation with a compliant or non-compliant balloon to achieve a balloon/artery ratio ≥0.75 was mandatory prior to randomisation. After successful predilatation, patients were randomised to one of the study devices via an interactive web response system. All stents were implanted according to standard procedures and scaffold/stent size was visually estimated. Post-dilatation and use of post-procedural intravascular imaging were left to the operator’s discretion. Post-dilatation was recommended in cases with residual diameter stenosis ≥20%. Patients were treated with at least one year of dual antiplatelet therapy. Angina status was obtained <72 hours after stent implantation, at six months and at 13 months by dedicated personnel using the Seattle Angina Questionnaire (SAQ).
13-MONTH INVASIVE CORONARY PROCEDURE
A detailed explanation of the 13-month coronary angiography, endothelial function test, microcirculatory examination and OCT imaging can be found in Supplementary Appendix 1.
ANGIOGRAPHIC AND OCT ANALYSES
Quantitative coronary angiography (QCA) and OCT analyses were performed by a dedicated core laboratory (BARCICORE-lab, Barcelona, Spain) according to our previous publication10. Endothelium-dependent and independent QCA vasomotor response of the in-scaffold and the 5 mm proximal and distal peri-scaffold segments was measured taking into account the core laboratory variability. Significant vasomotor changes (vasodilation or vasoconstriction) were defined in case of >4% change of the mean lumen diameter with respect to the 13-month baseline coronary angiography10,12.
STATISTICAL ANALYSIS
Categorical variables are presented as counts and percentages, and continuous variables as mean±standard deviation. Comparisons of categorical variables were estimated with the chi-square test. Comparisons of continuous variables between groups were estimated with the t-test for independent samples or with the non-parametric Mann-Whitney U test as appropriate. Comparisons of paired continuous data (such as mean lumen diameter changes) were estimated with generalised linear models for repeated measures. OCT strut-level analysis was performed considering the clustering nature of the OCT data with generalised estimating equations according to our previous publication10. A two-sided p-value ≤0.05 was considered statistically significant. Analysis of the five components of the SAQ was performed by means of an analysis of covariance (ANCOVA) test using the Bonferroni correction for multiple testing of the five components. Statistical analysis was performed with the SPSS software, Version 20.0 (IBM Corp., Armonk, NY, USA).
Results
POPULATION
A total of 70 patients (35 BVS and 35 EES) were included. There were no statistically significant differences regarding the main clinical and procedural characteristics. Baseline characteristics are shown in Table 1. One patient in the BVS group presented with coronary perforation after BVS implantation and required additional stent implantation of two intracoronary covered devices. One patient in the BVS group, treated with aspirin and clopidogrel, died from intracranial bleeding and nine patients refused the 13-month invasive procedure. Finally, 59 patients underwent invasive control (30 BVS and 29 EES). Three patients in the BVS group and one in the EES group presented with target vessel restenosis and did not undergo functional and imaging examination. There was no BVS or EES thrombosis at 13 months. A flow chart of the study is shown in Figure 1.
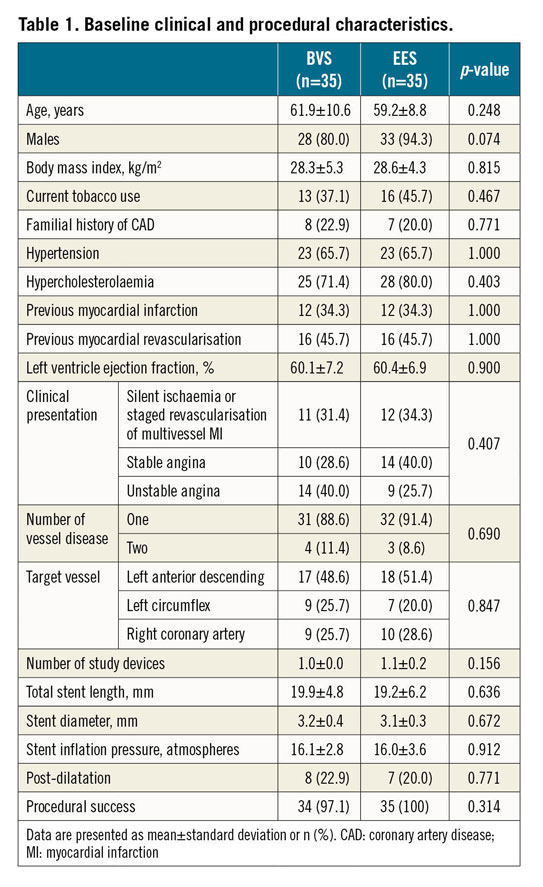
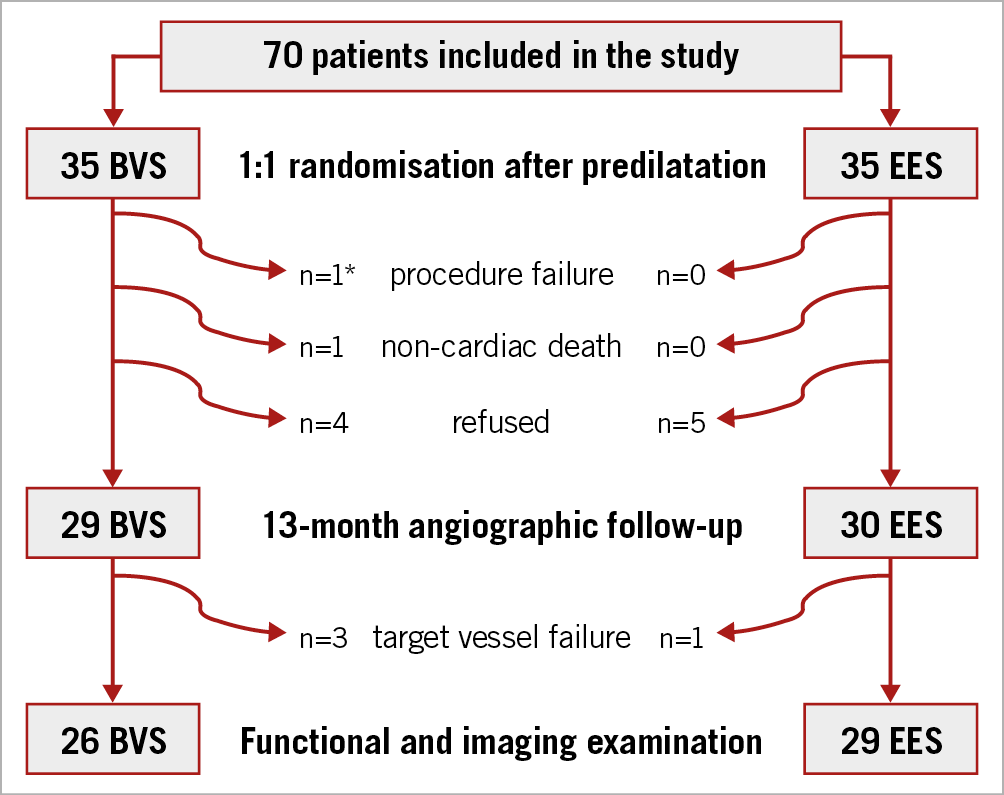
Figure 1. Study flow chart. * One patient presented with coronary perforation after scaffold implantation and required implantation of two covered stents.
ANGIOGRAPHIC ANALYSIS
Table 2 shows the baseline and 13-month QCA results. At baseline, pre-intervention study lesions were similar between BVS and EES. However, in-scaffold diameter stenosis post intervention was significantly larger with BVS (9.6±6.1%) than with EES (3.5±7.1%); p=0.001.
At 13 months, BVS had smaller in-scaffold minimal lumen diameter (2.30±0.57 mm vs 2.57±0.39 mm; p=0.046), larger lumen loss (0.30±0.43 mm vs 0.11±0.16 mm; p=0.026) and larger diameter stenosis (16.9±15.5% vs 6.1±6.1%; p=0.001).
EPICARDIAL VASOMOTOR RESULTS
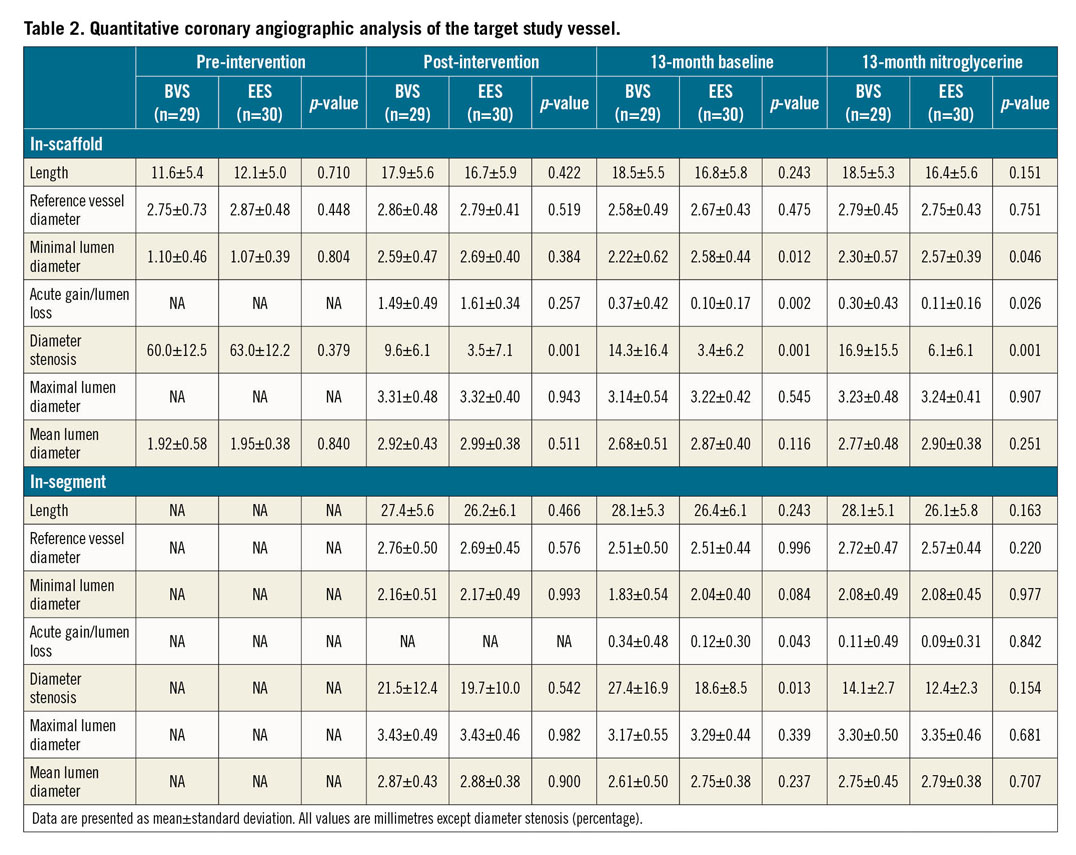
Epicardial vasomotor responses to endothelium-dependent and independent stimuli are shown in Table 3 and Figure 2. One patient did not undergo the vasomotor test because of severe spasm of the coronary artery during catheterisation. Moreover, nine proximal peri-scaffold segments were not analysed due to anatomical reasons.
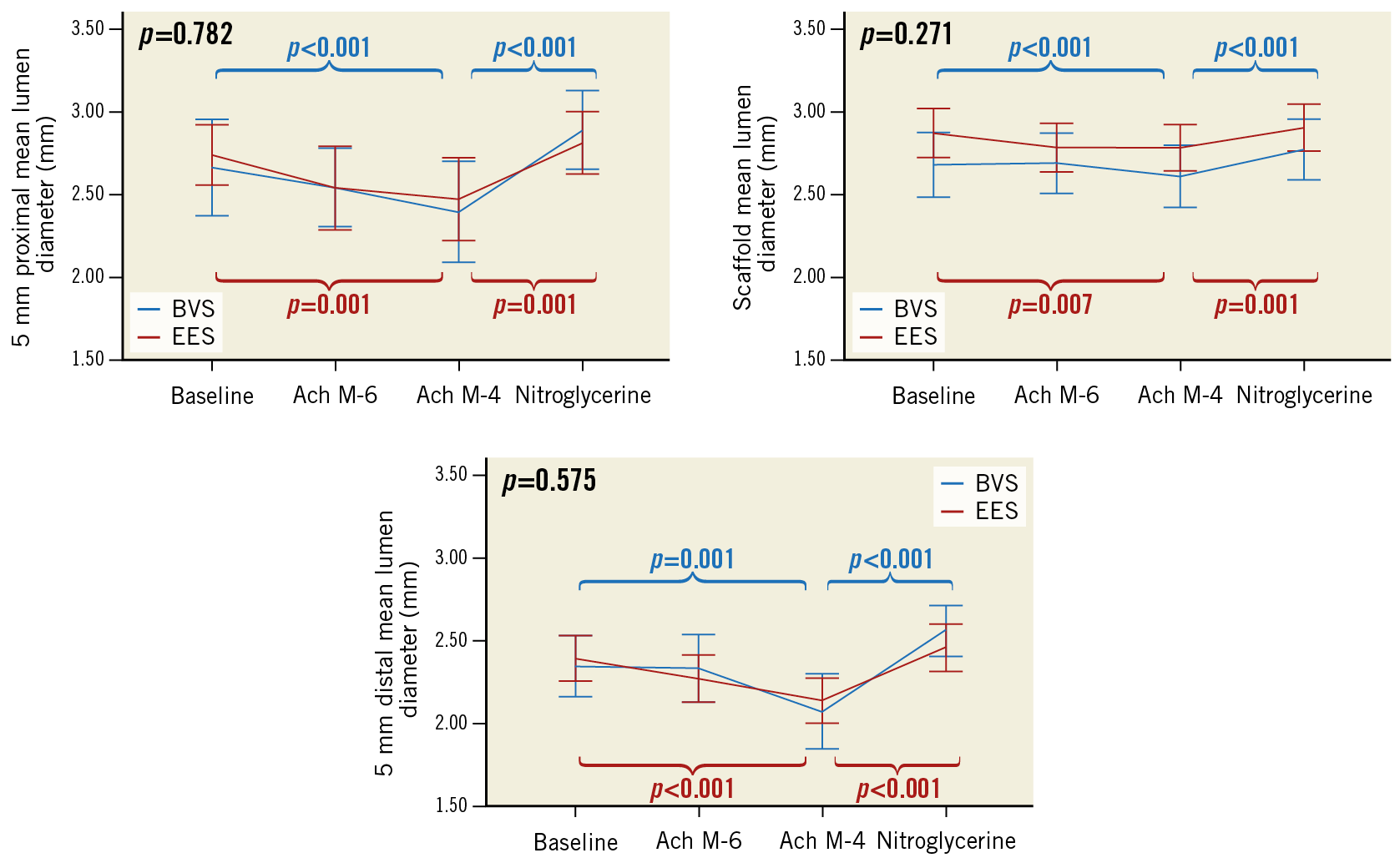
Figure 2. Endothelial and non-endothelial mediated vasomotor response at 13 months.
Proximal and distal peri-scaffold segments presented with vasoconstriction response to acetylcholine in 75.6% and 72.2% of patients, respectively. Nitroglycerine caused vasodilation in 80.0% and 87.0% of proximal and distal segments, respectively. BVS presented with larger vasoconstriction to acetylcholine in the scaffold segment than EES (60.0% vs 27.6%; p=0.010). The relative difference was −4.9±5.7% with BVS and −1.9±3.5% with EES (p=0.017). However, vasodilation with nitroglycerine was similar in both groups (8.3±6.4% vs 5.8±4.6%, respectively; p=0.184). Figure 3 shows the patient with the largest in-scaffold vasomotor change to acetylcholine and nitroglycerine.
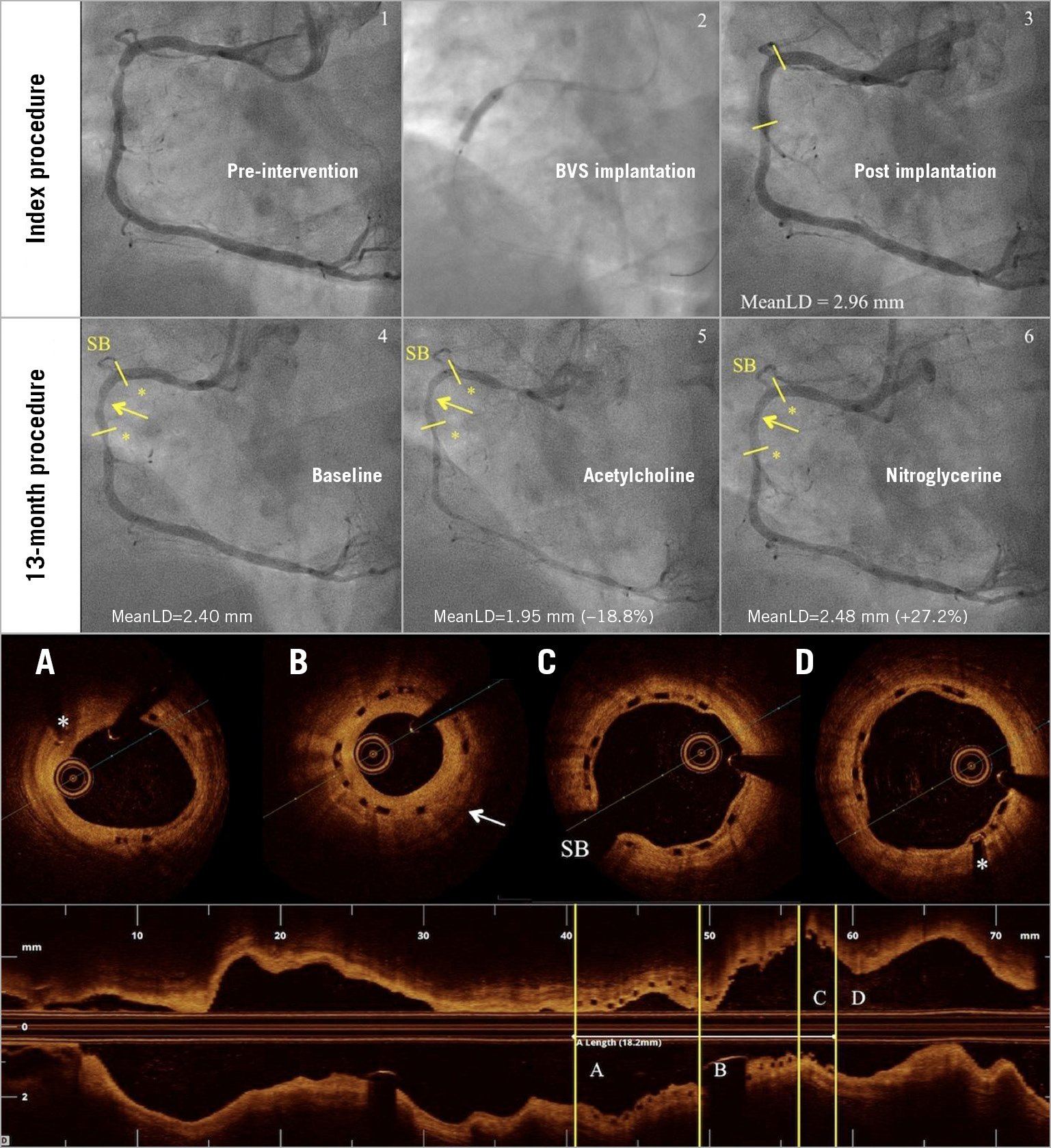
Figure 3. Patient with largest in-scaffold changes to vasomotor test. A 50-year-old male underwent catheterisation due to silent ischaemia. The angiography showed single-vessel disease in the right coronary artery that was treated with a 3.5×18 mm BVS (1-3). At 13 months, quantitative coronary angiography (4-6) showed vasoconstriction to acetylcholine and vasodilation to nitroglycerine. Optical coherence tomography (A-D) showed moderate neointima proliferation (arrow). Coronary flow at rest and at maximal hyperaemia was 62 ml/min and 88 ml/min, respectively. Fractional flow reserve and coronary flow reserve were 0.94 and 1.42, respectively. MeanLD: mean lumen diameter; SB: side branch
DOPPLER AND PRESSURE RESULTS
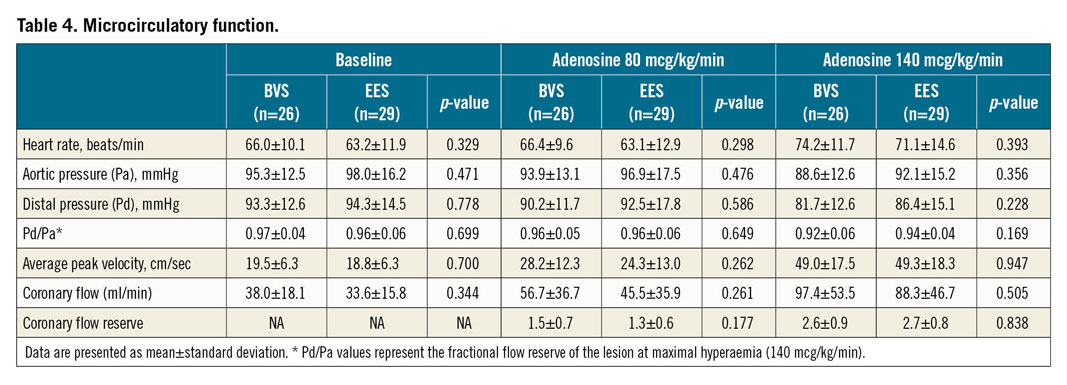
Functional examination of the coronary flow is shown in Table 4. There were no statistically significant differences regarding 13-month baseline pressure and Doppler ultrasound measurements. At maximal hyperaemic doses of adenosine, fractional flow reserve (FFR) was similar with BVS (0.92±0.06) and EES (0.94±0.04); p=0.169. A total of 4 patients (7.2%; 3 BVS and 1 EES) presented with ischaemic values of FFR (≤0.80) at 13 months. The APV (49.0±17.5 vs 49.3±18.3 cm/sec; p=0.947), CBF (97.4±53.5 vs 88.3±46.7 ml/min; p=0.505) and coronary flow reserve (CFR) (2.6±0.9 vs 2.7±0.8; p=0.838) were also similar in both groups.
ANGINA STATUS
Figure 4 shows the SAQ results at different time points in the study. There were no statistically significant differences between the two study devices regarding the five main aspects of the test: 1) physical limitations due to angina; 2) recent changes in the severity of angina; 3) angina frequency; 4) satisfaction with the anti-angina treatment; and finally 5) patient’s quality of life. At 13 months, the rate of patients with self-reported persistent angina was numerically lower with BVS (20.7%) than with EES (33.3%); p=0.275.
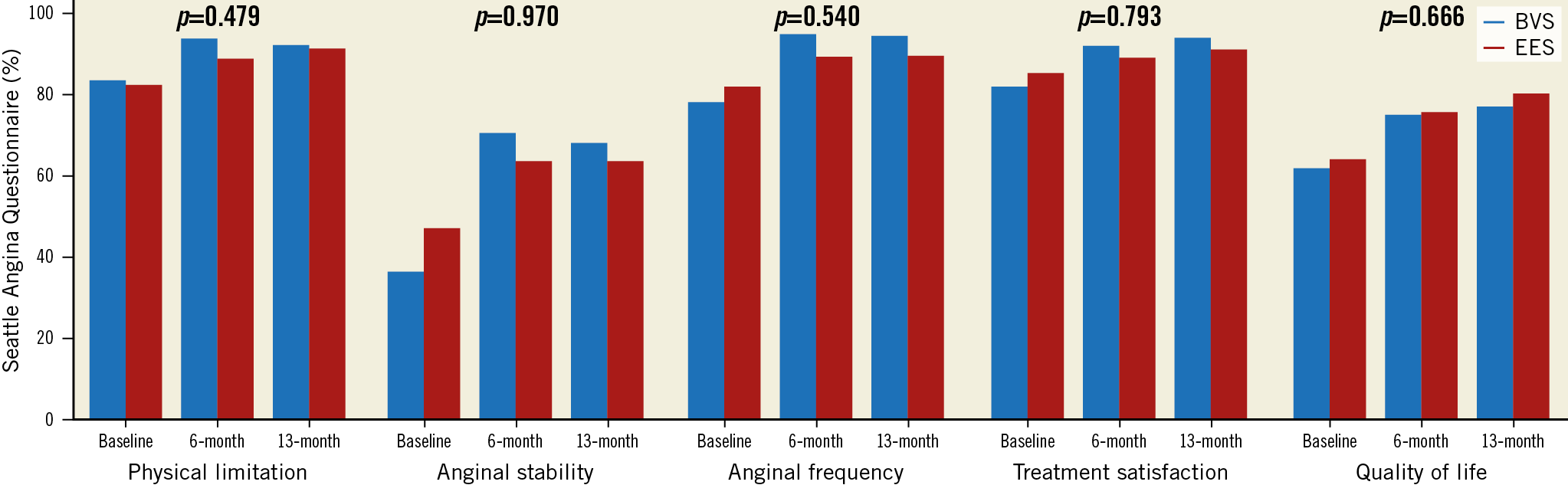
Figure 4. Seattle Angina Questionnaire results. P-value indicates the difference between the study devices. Scores range from 0 to 100 with higher scores indicating better health status.
OCT RESULTS
There were two patients (one BVS and one EES) with no OCT imaging due to technical issues. OCT results are shown in Table 5. At 13 months, reference lumen area was numerically larger with BVS than with EES (8.34±3.05 mm2 vs 7.48±2.40 mm2; p=0.238), and minimal lumen area was numerically smaller with BVS than with EES (5.06±2.36 mm2 vs 5.37±1.70 mm2; p=0.568). This caused larger lumen area stenosis with BVS than with EES (34.9±37.0% vs 27.2±10.7%; p=0.021). Neointima tissue characteristics were similar between groups regarding the predominant homogeneous pattern (71.4% vs 72.4%; p=0.505) and observation of neoatherosclerotic plaques (3.6% vs 3.4%; p=0.980).
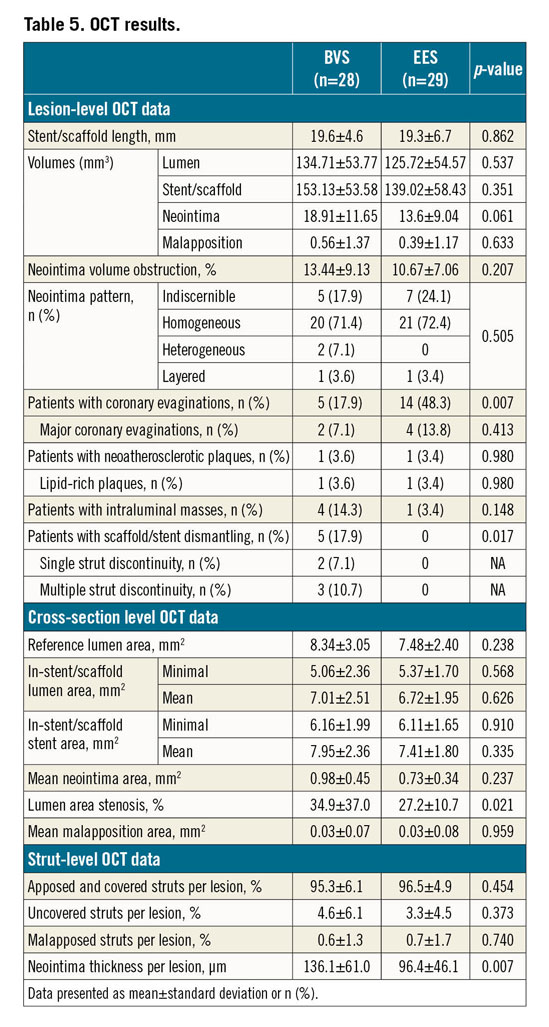
Uncovered struts (4.6% vs 3.3%; p=0.373) and malapposed struts (0.6% vs 0.7%; p=0.740) were also similar between groups. Scaffold discontinuities were observed in 35.7% of BVS patients and in no patients of the EES group (p<0.001). Fractured struts protruding into the lumen (so-called scaffold dismantling) were observed in 17.9% of patients in the BVS group.
Discussion
The main findings of the present study are: 1) in a highly selected group of patients, BVS presented with similar hyperaemic coronary flow to EES at 13 months; 2) BVS were associated with larger endothelium-dependent vasomotor changes of the scaffold segment than EES at 13 months; 3) these changes in the scaffold segment were in line with the response observed in the 5 mm proximal and distal segments to the scaffold edges but to a lower degree; 4) OCT imaging showed similar neointima response in both groups but a remarkable amount of BVS dismantling was observed at 13 months; and finally 5) angina status and quality-of-life parameters also improved similarly in both groups during follow-up.
In the ABSORB II study, the cumulative rate of angina reported by the investigators was 22% with BVS and 30% with EES at one year (p=0.04)6. Although there were no statistically significant differences in the SAQ parameters, angina symptoms during the exercise test were numerically lower with BVS (6.0% vs 8.5%; p=0.25) and fewer patients needed treatment with nitrates (19.5% vs 26.0%; p=0.09) compared to EES6. The authors hypothesised that restoration of coronary vasomotion could explain this potential benefit of bioresorbable technology. However, in the ABSORB IV trial, a total of 2,604 patients were randomised to BVS versus EES implantation using novel masking techniques to blind patients and clinical assessors to the randomisation group. In this study, there were no differences regarding the percentage of angina at one year. It is noteworthy that almost 21% of patients presented with persistent angina at one year, despite the fact that the need for repeat revascularisation was only 5%13. Similarly, in the present study, only 7% of patients presented with obstructive CAD at the invasive control, but 27.1% of patients reported angina at 13 months.
Microcirculatory dysfunction has been associated with cardiovascular risk factors and specific myocardial diseases1. Several studies have also reported the presence of microcirculatory dysfunction in around 40% of patients with symptoms and no evidence of obstructive CAD14.
On the other hand, in healthy coronary epicardial vessels, local shear stress stimulates the synthesis of endothelial factors (such as nitric oxide) in order to dilate or constrict epicardial coronary arteries. Coronary flow and vessel geometry are the main determinants of local shear stress15. After acetylcholine infusion, vessels and segments with an intact endothelium vasodilate, mediated by the release of nitric oxide, whereas vessels and segments with dysfunctional or disrupted endothelium respond with vasoconstriction as a result of direct activation of muscarinic receptors on vascular smooth muscle cells15.
Drug-eluting stent implantation causes denudation of the endothelial cells and inhibits their recovery by the release of the antiproliferative drug. Drug-eluting stents have been associated with a greater degree of endothelial dysfunction in the peri-stent segments than bare metal stents12. In the present study, both groups presented with 70% vasoconstriction to acetylcholine in the peri-scaffold segments. Moreover, the scaffold segment also presented with significant vasoconstriction to acetylcholine in 60% of patients treated with BVS. However, these vasomotor changes in the scaffold segment were smaller than those observed in the peri-scaffold segments. Coronary spasm, defined as a focal or diffuse epicardial lumen diameter reduction ≥90% during intracoronary acetylcholine administration compared to the relaxed state following intracoronary nitroglycerine, associated with the reproduction of symptoms and ischaemic ECG changes, is observed in around 50% of patients with persistent angina7. Although the current protocol did not register the 12-lead electrocardiogram (ECG), angiographic definition of coronary spasm was observed in the culprit vessel in 13% of patients (20% BVS vs 7.0% EES; p=0.153).
In the randomised VANISH study (Impact of Vascular Reparative Therapy on Vasomotor Function and Myocardial Perfusion), myocardial flow to hyperaemia and cold pressor testing (to assess myocardial flow during endothelial stimulation) of BVS and EES was assessed by positron emission tomography at one-month, one-year and three-year follow-up16. Coronary flow decreased similarly in both groups over time and was also similar during the different physiologic states (hyperaemic and endothelial stimulation)16. Therefore, the present study is in line with the results of the VANISH study.
Finally, around 10-20% of patients undergoing complete angiography-guided revascularisation present with residual FFR indicative of ischaemia (≤0.80)17. This has been associated with persistent angina and worse outcomes17. In the present study, two out of four patients with ischaemic FFR values also had angina symptoms at 13 months.
Limitations
The present study has several limitations. First, there were four patients with target vessel restenosis at 13 months. It is unknown if the endothelial and microcirculatory dysfunction was related to the restenosis process in these patients since there was no post-procedural functional examination. Second, an endothelium-independent vasomotor test (with nitroglycerine) was conducted after the acetylcholine test. Although a two-minute washout period was requested per protocol, it is possible that the vasomotor response to nitroglycerine was influenced by the previous response to acetylcholine. Finally, the present study did not measure the coronary flow during acetylcholine infusion, and therefore the impact of the endothelial function on the coronary flow is still unknown. However, the assessment of the coronary flow during maximal hyperaemia already accounts for the endothelium-dependent function of the epicardial segment due to the stimulation of the endothelium by the hyperaemic flow.
Conclusions
The presence of endothelial dysfunction in highly selected patients with a low burden of CAD treated with scaffold implantation is remarkable with both BVS and EES at 13 months. BVS mildly restore the vasomotor endothelium-dependent epicardial function within the scaffold segment. However, these epicardial vasomotor changes do not have any influence in the coronary flow at rest and at maximal hyperaemia at 13 months.
|
Impact on daily practice Persistent angina after complete revascularisation with stent implantation is estimated to be present in around 20-30% of patients. The causes of persistent angina are mostly vasomotor and microcirculatory dysfunction. According to the present study, BVS present with larger endothelial dysfunction in the scaffold segment than EES. Moreover, the hyperaemic coronary flow and rate of persistent angina are similar between the devices at 13 months. Therefore, further generations of BVS may not prevent persistent angina as compared to current permanent metallic drug-eluting scaffolds. |
Funding
This work was supported by a grant of the “Fundació La Marató de TV3” (reference number 201518-10).
Appendix. Study collaborators
Valentina León, MD; Hospital Universitari de Bellvitge, L’Hospitalet de Llobregat, Spain. Lara Fuentes, MD; Hospital Universitari de Bellvitge, L’Hospitalet de Llobregat, Spain. Albert Ariza, MD, PhD; Hospital Universitari de Bellvitge, L’Hospitalet de Llobregat, Spain. Raul Millan, MD; Grup de Recerca en Malalties del Cor, Hospital del Mar, Barcelona, Spain. Luis Ortega-Paz, MD; Hospital Clínic, Institut d’Investigacions Biomèdiques August Pi i Sunyer, Universitat de Barcelona, Barcelona, Spain.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.

