
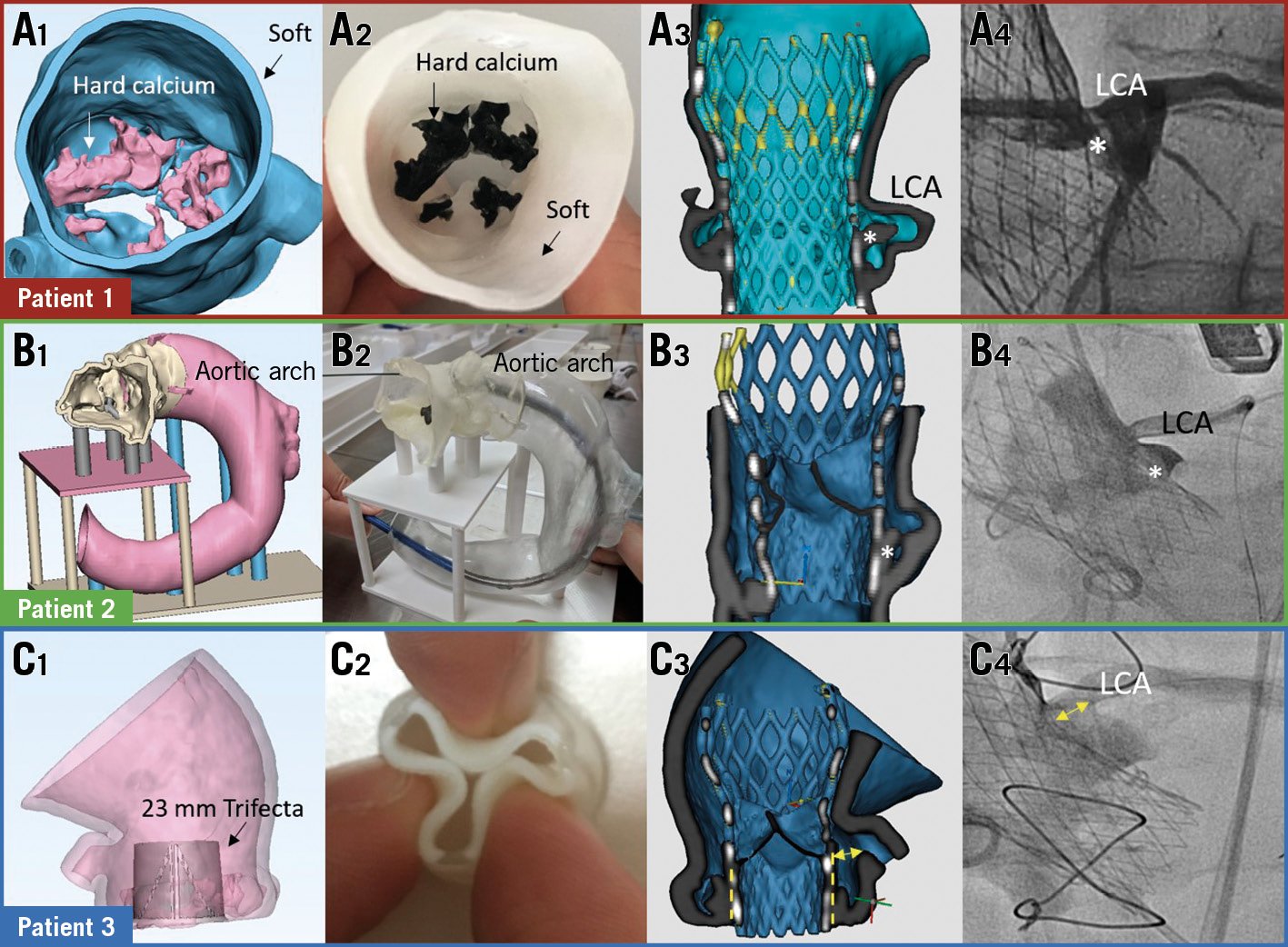
Figure 1. Simulation of TAVI with 3D printing. Digital and 3D-printed models (A1-C1, A2-C2). Re-imaged models and angiograms showing CO (A3, A4) and no CO (B3, B4, C3, C4). * native leaflet; double arrow=distance between Trifecta (dotted lines) and coronary ostium. CO: coronary obstruction; LCA: left coronary artery
Simulation of transcatheter aortic valve implantation (TAVI) on three-dimensional (3D) printed models was performed in three patients with high-risk anatomy for coronary obstruction (CO). Patient 1 had a narrow sinus (29.2 mm) and low coronary height (7.3 mm); patient 2 had a low coronary height (6.4 mm) and a horizontal aorta (71°); patient 3 had an externally mounted bioprosthesis (Trifecta™; Abbott Vascular, Santa Clara, CA, USA) undergoing valve-in-valve procedure. Digital models of the AV, aortic root and bioprosthesis were created using dedicated software (Mimics 19.0) based on computed tomography (CT) data sets (Figure 1A1, Figure 1B1, Figure 1C1). Physical models were fabricated with a PolyJet printer (Stratasys, Rehovot, Israel) using tissue-mimicking materials for calcifications (VeroBlack), Trifecta stent frame (VeroClear), valve leaflets and aortic wall (Agilus 30) (Figure 1A2, Figure 1B2, Figure 1C2). Selected Evolut™ prostheses (Medtronic, Minneapolis, MN, USA) were placed in each model (Moving image 1) and re-imaged using CT to assess the proximity of the displaced leaflets to the coronary. Procedural simulation for patient 1 showed left main CO by the displaced leaflet (Figure 1A3, Moving image 2). The actual procedure was performed with coronary protection; prosthesis deployment was complicated by CO, which was successfully treated with “chimney” stenting (Figure 1A4). Procedural simulation for patients 2 and 3 showed no CO despite anatomical risks (Figure 1B3, Figure 1C3, Moving image 3, Moving image 4). There was no CO during the actual procedure (Figure 1B4, Figure 1C4).
Conventional CT-based assessment involves integrating multiple anatomical parameters and is limited by its inability to assess the interaction between the transcatheter valve and the patient-specific aortic platform. Simulation on 3D-printed models provides important information regarding mechanical interactions between the transcatheter and native valves, although digital simulation may provide similar information but demands complex finite element analysis with boundary assumptions. 3D printing would therefore be a more practical and potentially more accurate approach to predict the mechanical complications of TAVI.
Funding
This work was funded by the Hong Kong Special Administrative Region Government Health and Medical Research Fund (05160976) and the Innovation and Technology Fund (ITS/025/16).
Conflict of interest statement
A. Lee has received research grants from Abbott and Medtronic. E. Wu is a proctor for Abbott and Boston Scientific and has received research grants from Asahi Intecc and Vascular Solutions. The other authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.
Moving image 1. TAVI simulation on a 3D model.
Moving image 2. CT of post-implant model of patient 1 showing LCA obstruction.
Moving image 3. CT of post-implant models of patient 2 showing no CO.
Moving image 4. CT of post-implant models of patient 3 showing no CO.

