Abstract
The European Association of Percutaneous Cardiovascular Interventions (EAPCI), the European Heart Rhythm Association (EHRA), the European Association of Cardiovascular Imaging (EACVI), the European Society of Cardiology (ESC) Regulatory Affairs Committee and Women as One support continuous review and improvement, not only in the practice of assuring patients a high quality of care but also in providing health professionals with support documents to help them in their career and enhance gender equity. Recent surveys have revealed that radiation exposure is commonly reported as the primary barrier for women pursuing a career in interventional cardiology or cardiac electrophysiology (EP). The fear of foetal exposure to radiation during pregnancy may lead to a prolonged interruption in their career. Accordingly, this joint statement aims to provide a clear statement on radiation risk and the existing data on the experience of radiation-exposed cardiologists who continue to work in catheterisation laboratories (cath labs) throughout their pregnancies. In order to reduce the barrier preventing women from accessing these careers, increased knowledge in the community is warranted. Finally, by going beyond simple observations and review of the literature, our document suggests proposals for improving workplace safety and for encouraging equity.
Introduction
Why is a collaborative statement on radiation exposure during pregnancy required? Interventional cardiology and electrophysiology are the two subspecialities with the lowest female representation in cardiology1234. Radiation exposure is commonly identified as a major barrier for women considering a career in interventional cardiology and electrophysiology45, and concerns regarding occupational radiation exposure during pregnancy are likely to lead to missed career development opportunities6. Amongst European Union countries, there are substantial variations in occupational radiation regulations and practices, despite the recent definition of safe dose limits and safety standards for foetal exposure from the European Directive, as well as the repeal of previous Euratom directives7. The 2011 Society for Cardiovascular Angiography and Interventions (SCAI) and 2017 European Heart Rhythm Association consensus documents and 2018 European Association of Percutaneous Cardiovascular Interventions (EAPCI) survey report289 endorse the safe continuation of work with occupational radiation exposure while pregnant; yet, in some EU countries, pregnant women are not permitted to work in the cardiac catheterisation laboratory, while in other EU countries, policies are more permissive10 (Table 1). Nevertheless, there remains a lack of precise data on the real risks to the foetus and awareness of the existing directives, not only amongst heads of departments but also amongst women working as physicians in interventional cardiology11. Therefore, while European, North American, Japanese, and Australian directives allow women to work if closely monitored with an abdominal dosimeter to ensure that the foetus does not exceed recommended dose limits during pregnancy, in some countries, pregnant women are not allowed to work. There is also a disparity regarding the maximum foetal dose allowed across countries: 1 mSv in Europe7, Australia12 and Israel, 2 mSv in Japan and 5 mSv in the US13 (Table 1). This lack of knowledge that persists among the interventional and electrophysiology communities contributes to workforce gender disparity. In order to reduce barriers preventing women from accessing a career in interventional cardiology, there should be clear statements on radiation risk and data collection on the current experiences of radiation-exposed cardiologists who continue to work in catheterisation laboratories throughout their pregnancies. The aim of the present document is to provide data and clear information on radiation risk in order to overcome the “radiation barrier”.
Table 1. Current legal requirements: EU, UK, USA, Australia, Israel. Overview of the current EU regulations and their local application demonstrating a general trend in more restrictive applicative directives usually limiting access to the catheterisation/electrophysiology lab and allowing a discretional power to the X-ray surveillance experts. Overview of the current non-EU regulation with focus on the UK, USA, Australia and Israel.
| The European Directive 2013/59/Euratom, Art.10 has been adopted in your Country. YES/NO | COUNTRY LAW | Exposure during pregnancy (cumulative dose over the pregnancy) | Exposure during breastfeeding | In-hospital discretionary allowance | |
|---|---|---|---|---|---|
| AUSTRIA | YES | YES | Not allowed | Not allowed | NA |
| BELGIUM | YES | ARBIS 20-07-2001 | Yes, should be <1 mSv and ALARA | Yes, allowed | Risk analysis of occupational exposure for every pregnant woman with arbitrary allowance according to the X-ray surveillance expert. |
| CYPRUS | YES | 2018 | Yes, should be <1 mSv | Yes, allowed | NA |
| DENMARK | YES | BEK nr 669 af 01/07/2019 (equivalent to EU directive) | Yes, should be <1 mSv after pregnancy is recognised − special dosimetry is encouraged (day-to-day monitoring) | Ok, but exposure should be limited | NA |
| FRANCE | YES | Articles D. 4152-5 and R. 4451-45 of French Labour Code | Yes, should be <1 mSv | No specific indications | Possibility of shifting the pregnant woman to an occupation with less radiation exposure or keeping her in her current assignment if the total expected radiation dose between pregnancy confirmation and childbirth is <1 mSv |
| GERMANY | YES | Strahlenschutzgesetz (StrlSchG): German Radiation Protection Law dated June 27, 2017 (Federal Law Gazette Part I, p. 1,966), last amended by Article 2 of the law of May 20, 2021 (Federal Law Gazette Part I p. 1,194): §78Strahlenschutzverordnung (StrlSchV): German Radiation Protection Ordinance dated November 29, 2018 (Federal Law Gazette Part I, p. 2,034-2,036), last amended by Article 6 of the law of May 20, 2021 (Federal Law Gazette Part I p. 1,194): §55 | Yes, should be <1 mSv, weekly monitoring | Yes, allowed | Risk analysis of occupational exposure for every pregnant woman with arbitrary allowance according to the X-ray surveillance expert. |
| HUNGARY | YES, in a restricted manner (no exposure when pregnant/breastfeeding) | Government Decree 487/2015 (XII. 30.) on the protection against ionising radiation and the corresponding licensing, reporting (notification) and inspection system | Not allowed | Not allowed | The pregnant woman shall have her work reorganised to avoid any risk (including infections, radiation, etc.).The pregnant woman cannot be dismissed/suspended because of her status. |
| IRELAND | YES | RADIOLOGICAL PROTECTION ACT 1991 (IONISING RADIATION) REGULATIONS 2019 | Yes, should be <1 mSv | Yes, allowed | NA |
| ITALY | YES, in a restricted manner (no exposure when pregnant/breastfeeding) | DL 101 July 2020 | Not allowed | Not allowed | Not allowed |
| NETHERLANDS | YES | Yes, should be <1 mSv | Yes, allowed | Risk analysis of occupational exposure for every pregnant woman with arbitrary allowance according to the X-ray surveillance expert | |
| PORTUGAL | YES (108/2018, article 69) 102/2009 | 102/2009, 10 September | Not allowed | Not allowed | Not allowed |
| ROMANIA | YES | LAW 154/2015 | Not allowed | Not allowed | The employer must change the working conditions, so that there is no radiation exposure |
| SLOVENIA | ZVISJV-1, December 2017 | Yes, should be <1 mSv, monthly monitoring | Yes, allowed | Institution is obliged to organise a different workplace or reorganise working time under exposure so the goal <1 mSv can be met | |
| SPAIN | YES, current laws, although enacted before this directive was issued, are in accordance with it | Royal Decree 298/2009, of 6th March, which modifies the Royal Decree 39/1997, of 17th January | Yes, should be <1 mSv, monthly monitoring | Yes, allowed. During the breastfeeding period the worker will not be employed in a job with radionuclides and contaminants | Even though current law allows women to work provided the dose is <1 mSv for the whole pregnancy, in some cases, the the X-ray surveillance department or the occupational risk department do not allow a woman to work |
| SWEDEN | YES | SFS 2018:396 | Yes, should be <1 mSv | Yes, allowed | How a foetal dose below 1 mSv is ensured is up to the authority, i.e., the public healthcare provider. There may be different ways of calculating the foetal dose between hospitals. Currently a factor of 5 is used. |
| NON-EU | |||||
| AUSTRALIA | NO, the current Australian codes and standards are based on the 2020 guidelines of the International Commission on Ionising Radiation Protection (ICNIRP) | Australian Radiation Protection and Nuclear Safety Agency Radiation Protection Diagnostic and Interventional Cardiology, series 14.1. Australian Radiation Health Committee. 2008 | Yes, dose to foetus should be <1 mSv from declaration of pregnancy to delivery | Yes, allowed (no threshold specified) | NA |
| ISRAEL | NO | 1992 Law by the Ministry of Labor, Social Affairs and Social Services | Yes, dose should be <1 mSv/9 months | Yes, allowed. No limitations | At 4 months pregnancy − medical examination by occupational medicine |
| UK | YES | The Ionising Radiation Regulations 2017 (IRR 17) | 1 mSv limit | None for radiation(only radionuclides and contaminants) | 1 mSv limit |
| USA | Pregnancy Discrimination Act and the American Disabilities Act: pregnant workers have a legal right to work adjustments that allow them to do their job without jeopardising their health | Individual states have Pregnant Workers Fairness laws | No legal specifications regarding exposure.NCRP recommends a dose <5 mSv/9 months | No legal specifications regarding exposure | Yes |
| ALARA: as low as reasonably achievable; NA: not applicable; NCRP: National Council on Radiation Protection and Measurements | |||||
Risks: biological evidence of radiation exposure during pregnancy
Ionising radiation exposure during pregnancy and the potential impact of prenatal radiation is a major concern for women in the cardiac catheterisation laboratory1415.
Prenatal radiation risks include pregnancy loss, congenital malformations, developmental delay, and carcinogenesis. Some risks are dependent on the radiation dose. These hazards can be stochastic, including the risk of childhood cancer and genetic disorders1617, or deterministic, which have a radiation dose threshold, such as intrauterine growth retardation, miscarriage, and congenital defects17. The greatest risk of pregnancy loss from radiation exposure is during the first 2 weeks of pregnancy, while between 2-8 weeks after conception, the embryo is most susceptible to the development of congenital malformations because this is the period of organogenesis18.
Table 2 shows the main deterministic irradiation effects on the embryo/foetus at each pregnancy stage. As can be appreciated in the table, the foetal radiation doses that have been related to the occurrence of abortion, malformations or intelligence quotient reductions are 100 times higher than those allowed during the entire pregnancy for an interventional cardiologist. Since the threshold dose for these deterministic effects is well above that which an invasive or interventional cardiologist would receive under a protective apron, the use of standard radiation protection techniques would result in negligible risk to the foetus. With respect to stochastic effects, which have no threshold dose, the embryo/foetus is assumed to be at about the same risk for potential carcinogenic effects of radiation as children throughout most of the pregnancy13. Table 3 shows the spontaneous probability of a newborn having a congenital malformation or childhood cancer and how this probability has a negligible increase when the occupational exposure range of the mother respects the 1, 2 or 5 mSv limits during pregnancy919.
However, understanding the mechanisms that underlie the pathogenesis of radiation-induced birth defects is difficult since possible radiation damages are linked to a multifactorial process which has not been well researched. There are no randomised trials assessing the risks of prenatal radiation. The most conclusive studies have been on massive radiation exposure such as after the atomic bombs used in World War II. There have also been estimates made from population studies of prenatal radiation exposure. The probability that a child will be born with a congenital abnormality or cancer increases from 4.07% with background radiation to 4.078% with 1 mSv conceptus exposure and to 4.12% with 5 mSv exposure20. There are no studies that show an increased risk of non-cancerous effects from prenatal radiation exposure below 50 mSv21. Occupational radiation exposure during pregnancy is regulated in Europe by directives from both the EU and each country, and in the USA by federal and state law, which take into account the recommendations from the International Commission on Radiological Protection (ICRP)13. The current National Council on Radiation Protection and Measurements (NCRP) recommends a monthly maximal exposure of <0.5 mSv, and the ICRP recommends a dose <1 mSv for the entire pregnancy. The limit of safe foetal radiation exposure of 1 mSv throughout pregnancy for the EU member states is based on the Directive 96/29/Euratom, which establishes that the protection of the foetus shall be comparable to that provided for members of the public, and thus it should not receive more than 1 mSv/year9. However, in the USA, regulation is less restrictive, and the NCRP in Report No. 174 recommends limiting occupational exposure of the foetus to not exceed 5 mSv throughout the entire pregnancy and 0.5 mSv per month of the pregnancy. This is because, in the USA, American employers are required to treat their female employees equally with respect to “pregnancy, childbirth or related medical conditions”. Therefore, the radioprotection policies for pregnant workers in the USA prioritise the pregnant worker’s rights from an anti-discriminatory perspective, while the European legislation policies prioritise mostly the safety rights of the unborn child9. In any case, the general instructions of the European directives continue stating that, once the woman has declared pregnancy, the employer must ensure that the equivalent dose limit for a foetus remains ≤1 mSv. Thus, while pregnancy does not require removing the exposed professional from work, a careful review of working conditions in order to comply with current regulations is warranted. However, nowadays there are still countries in the EU which prevent pregnant staff from working in the cath lab. Thus, we encourage the national interventional and electrophysiology (EP) societies of those countries to work together with their national health system authorities to promote changes in regulation and to repeal sexist laws that disincentive women to choose interventional subspecialities and expose the foetus to unnecessary risks by promoting late pregnancy declaration.
The amount of radiation exposure to the conceptus is measured by wearing a radiation badge under a lead apron at waist level. This amount should be assessed in all professionals who work in an environment with ionising radiation exposure, but all the more so when those professionals include pregnant women. Furthermore, operators can use the same technique to measure radiation exposure prior to pregnancy in order to estimate the likely occupational risk during pregnancy. Since the first trimester is when the conceptus is at highest risk to radiation exposure and since most women may not know they are pregnant during this time, it is important to practise universal radiation safety at all times.
Although the foetal radiation doses that have been related to the occurrence of malformations/childhood cancer are much higher than those allowed for the entire pregnancy for an interventional cardiologist10, little information is available in the literature on the dose received by pregnant employees exposed to ionising radiation. In order for interventional and EP cardiologists to make informed decisions, we encourage national interventional and EP societies to collect and publish data regarding radiation exposure and pregnancy outcomes in pregnant cardiologists working in the cath/EP labs.
Finally, the impact of radiation exposure on men should also be considered. In fact, chronic occupational radiation exposure among male workers is correlated with a higher prevalence of low birth weight in offspring and instability in the Y chromosome azoospermia factor c (AZFc) region, responsible for male infertility22.
Table 2. Main deterministic/stochastic irradiation effects on the embryo/foetus at each pregnancy stage (Adapted from9).
| Pregnancy stage | Main irradiation effect | Doses at which effects have been described | Risk at occupational dose (range ≤1-5 mSv exposure to the mother) | Spontaneous risk24 |
|---|---|---|---|---|
| Preconception gonadal irradiation | Has not been shown to result in increased cancer or malformations in children13 | |||
| Preimplantation(First two weeks post-conception) | Abortion | Doses over 100 mGy25 | Death of the conceptus due to radiation is not described28 | Risk of spontaneous abortion in known pregnant women 1/7 |
| Period of major organogenesis(weeks 3-8 post-conception) | Risk of malformation or growth retardation | Dose threshold of around 100 mGy26 | Not expected below 100 mGy26 | Risk of major congenital malformations 1/33Risk of growth retardation 1/33 |
| Early foetal period (weeks 8-25 post-conception) | Risk of reducing the intelligence quotient | Dose threshold of around 120-200 mGy weeks 8-15Dose threshold of around 500 mGy weeks 16-25 | Doses under 100 mGy and in the mother’s occupational exposure range (<5 mSv) would be of no practical significance26 | |
| Third trimester | Risk of malformation, growth retardation or reduced intelligence quotient not expectedPossible fatal or non-fatal cancer of any type (solid tumours and leukaemia) | Lifetime cancer risk following in utero exposure will be similar to that following radiation in early childhood | Lifetime cancer risk around 1/500 for 5 mSv in utero exposure2728, and 1/2,500 for 1 mSv in utero exposure2728 | Risk of childhood leukaemia per year 1/25,000/year |
Table 3. Probability of a child to be born with a congenital malformation or to develop childhood cancer spontaneously and after ionising radiation exposure (summarised from20).
| Foetal dose added to the background radiation (mSv) | Probability of a child having a congenital malformation (%)2930 | Probability of a child developing childhood cancer (%)2931 | Probability of a child having a congenital malformation or childhood cancer (%)32 |
|---|---|---|---|
| 0 (spontaneous risk) | 4.000 | 0.070 | 4.070 |
| 0.5 | 4.001 | 0.074 | 4.072 |
| 1 | 4.002 | 0.079 | 4.078 |
| 2.5 | 4.005 | 0.092 | 4.090 |
| 5 | 4.010 | 0.110 | 4.120 |
| 10 | 4.020 | 0.160 | 4.170 |
Proposal for better practice
Operators using fluoroscopy must be guided by the “as low as reasonably achievable” (ALARA) principle, in which the obtention of optimal images must be balanced with prodecure safety. The practice of radiation safety is grounded in an understanding of external radiation protection measures as well as technical considerations in operating the X-ray system. Minimising a pregnant operator’s radiation exposure follows similar principles to general working practices in a radiation environment. Scatter radiation emitted from the patient is the greatest source of radiation exposure to the operator and personnel. Therefore, methods to reduce radiation exposure to the patient will automatically reduce operator and personnel exposure. Furthermore, appropriate standardised operating procedures must be in place to prevent unintentional exposure.
The three fundamentals of radiation safety to an operator include 1) time, 2) distance and 3) shielding and dosimeter monitoring. Time refers to the amount of time the operator spends using the X-ray system, where less usage equals less radiation exposure23. Operators must maximise their distance from the X-ray source as radiation intensity follows the inverse square law: if the operator’s distance from the X-ray source increases from 40 cm to 80 cm, radiation intensity reduces by a factor of 4. Shielding is used in the form of personal, tableside or external protection with each form having a degree of lead equivalence defining its radiation protective effect. Personal shielding includes a lead apron with or without shoulder covers for breast shielding, a thyroid collar and lead glasses. The lead apron should be of at least 0.35 mm thickness, attenuating approximately 95-96% of the scatter radiation. The use of 0.5 mm thickness attenuates 98.0-99.5% of the scatter radiation dose. An overhead movable lead shield of 1 mm thickness positioned close to the patient and between the operator and the entry of the X-ray source can reduce radiation exposure by 95%. Available data show that the majority of foetal radiation dose exposure rarely exceeds 0.3 mSv (Table 4, Table 5). The key principles of radiation safety for pregnant staff are summarised in the Central illustration.
Table 4. Current published and unpublished data from practice or indirect studies.
| Author | Settings | Attitude during pregnancy and dose received | Outcome | |
|---|---|---|---|---|
| Spain(Velázquez et al, 201710) | Interventional cardiologists and electrophysiologists (n=5) | Background radiation in 80% of all pregnancies, 0.2 mSv in one pregnancy | 4 normal pregnancy outcomes, 1 pregnancy with placental insufficiency | |
| New Zealand* (Unpublished anecdotal accounts only) | Multiple trainees and interventional cardiologists in New Zealand | Unpublished accounts of case-by-case and monthly foetal monitoring with radiation dose well below safe pregnancy thresholds* | Normal pregnancy outcomes reported | |
| Australia* (unpublished anecdotal accounts only)(*Burgess S; on behalf of Women in Interventional Cardiology of Australia and New Zealand (WIICAN). Unpublished data on pregancy in Female Interventional Cardiologists of Australia and New Zealand 2021) |
Multiple trainees and interventional cardiologists in Australia(n=11) | Unpublished accounts of case-by-case and monthly foetal monitoring with radiation dose well below safe pregnancy thresholdsIn detail: Of 19 female interventional cardiologists (IC) in Australia and New Zealand2, 13/19 (68%) known to the author* were directly contacted by phone or email, 11/13 responded (85%). Amongst responders 11/11 (100%) had at least one pregnancy during advanced training, fellowship or consultancy. A total of 21 pregnancies were included. Amongst responders, during 86% of all pregnancies, doctors, including 82% of IC, remained in the cath lab with appropriate shielding and without any adjustment of schedule or cath lab exclusion. Of the remaining 2 IC, one doctor was excluded from weeks 8-15 in 1 of 2 pregnancies but remained in the lab without exclusion for her other pregnancy, and the remaining doctor chose to self-exclude from the catheterisation laboratory from approximately 6-9 weeks.All 11/11 responders (100%) reported dose monitoring at the time of their pregnancies with radiation doses well below thresholds for safe pregnancy as defined by Australian and New Zealand policy and standards. | Pregnancy outcomes consistent with those of the general population were reported | |
| USA(Marx MV et al 199233) | Prospective study (n=30) of interventional radiologists and trainees (not pregnant, male and female) | The calculated average dose to pregnant interventional radiologists (in 1992) over a 40-week pregnancy with two layers of lead=0.4 mSv dose calculated to be 1.3 mSv with one layer of lead (NCRP occupational foetal dose limit=5 mSv) | ||
| France(Vautrin et al34) | Questionnaire among the female population in interventional cardiology in France (n=14) | 14/26 women had children. Half of them continued to work during the first trimester of pregnancy, and 2 continued until 7 months. Four wore an additional lead apron for double lead protection of the abdomen. Dose received was not asked in the questionnaire. | Outcomes not reported | |
| USA(Sarma AA et al6) | Survey of the women in cardiology section of the American College of Cardiology (n=501 women) | 47% of women tried to avoid pregnancy during periods when they would be exposed to radiation and 57% of women experienced radiation during a pregnancy, without a difference between trainees (49%) vs attending physicians (58%; p=0.28). Those under 50 at the time of the survey were actually more likely to have avoided pregnancy during periods of radiation exposure as compared with those >50 (50% vs 39%; p=0.03), suggesting that younger cardiologists are more concerned about radiation.Women were relatively uninformed about whether their department had an official policy regarding radiation exposure during pregnancy: 34% saying they didn’t know if their department had a policy, 32% reporting that their department did not, and 34% reporting that their department did (no difference between women >50 years of age and women <50 years of age, potentially limiting senior cardiologists from providing mentorship on this issue to younger colleagues).Among women who experienced pregnancy radiation exposure, only 20% used foetal radiation badges, 24% used additional lead, and 42% increased their distance from radiation sources. Thus, despite a high rate of concern, pregnant cardiologists underuse radiation reduction and monitoring strategies.Dose received during pregnancies was not asked in the survey. | Outcomes not reported | |
| *Published and unpublished data from interventional cardiologists that continued to work in the cardiac catheterisation laboratories during their pregnancies. While numerous interventional cardiologists from Europe, North America, Australia and New Zealand report safely working and training throughout pregnancy without interruption using various radiation monitoring techniques, little published data exist. | ||||
Table 5. Unpublished data from practice.
| Country | Pregnancies with radiation exposure, standard shift | Means of reducing foetal radiation exposure | Equivalent dose received during pregnancy | Outcome |
| Spain | 15 pregnancies/11 interventional cardiologists | Standard 1 vest+1 skirt 7/15Extra skirt or gonadal protection shield 8/15 | Background radiation 8/150.2 mSv 2/15<1 mSv 3/15Don’t recall 2/15 | 14 pregnancies: normal outcome1 pregnancy: placental insufficiency |
| France | 8 pregnancies/5 interventional cardiologists | Standard 1 vest+1 skirt 5/8Extra removable on-wheels shield protection 3/8 | 7/8 pregnancies: background radiation0.23 mSv in one pregnancy | Normal pregnancy outcomes reported for all pregnancies |
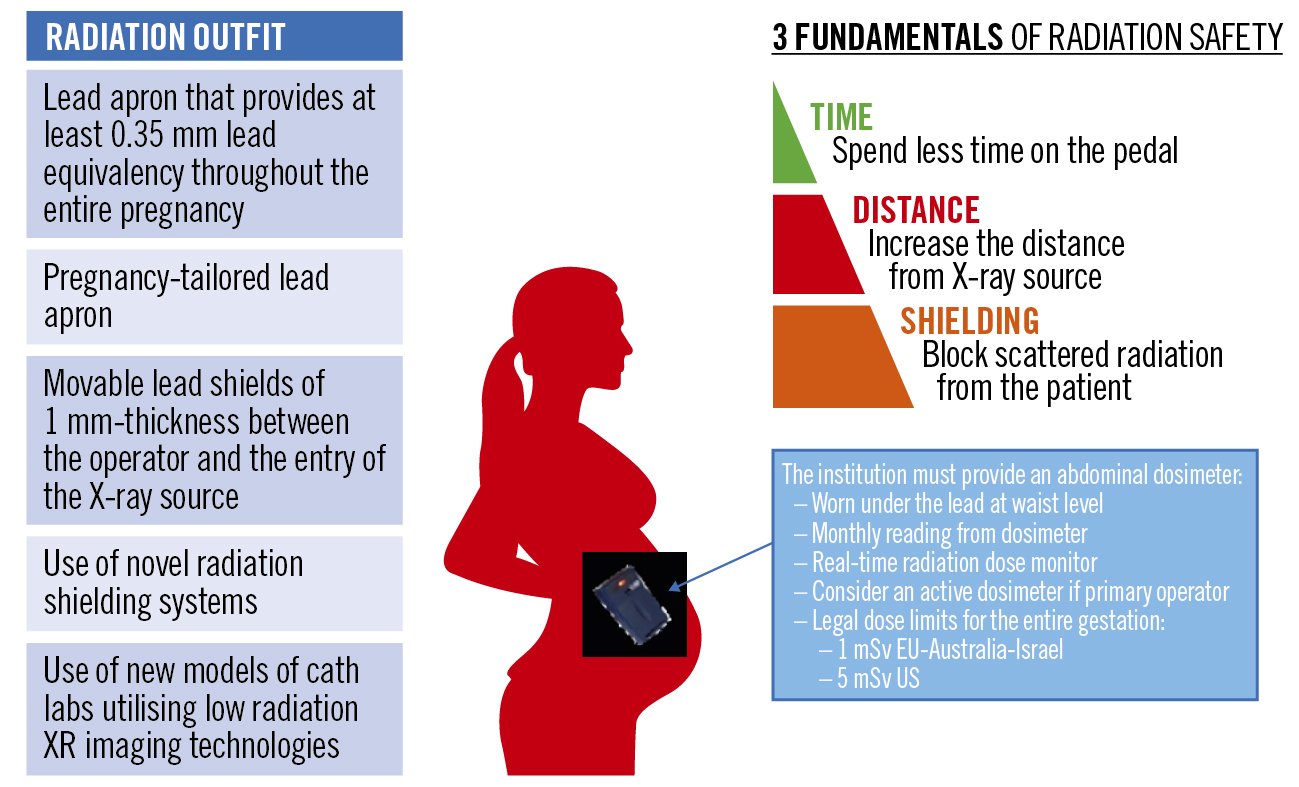
Central illustration. Managing the radiation safety of pregnant staff. (adapted from Women as One https://rad.womenasone.org)
Proposal for encouraging equity
The root causes of gender disparity in cardiology are numerous but related mainly to societal and cultural norms concerning the role of women. This is most apparent regarding parenting and family planning for women during training and early career, especially in invasive subspecialities. As medicine witnesses a cultural shift that overlaps with the changing societal biases towards gender, paradigm changes in practices must occur to increase female representation and narrow gender inequalities among trainees and faculty in invasive cardiology subspecialities.
The first change should target medical schools’ curricula, introducing medical students to clinical cardiology and supporting students interested in invasive cardiology subspecialities regardless of race or sex. Commonly heard fallacies must be clarified, particularly the ones related to the impact of radiation on women’s fertility and conception. Institutions should improve radiation counselling to address employees’ concerns adequately and train them in safe practices that minimise radiation exposure. Concurrently, efforts must be made to convert the workplace into a friendlier environment for families, especially pregnant interventionists. For instance, lighter protective garments must be introduced to avoid additional physical burdens on pregnant women and ensure that these protection suits are adjustable as the foetus grows. Emerging technologies to minimise radiation exposure in male and female operators (i.e., tailored shielding, lead offloading, robotics, 3-dimensional mapping systems in invasive electrophysiology, etc.) should be introduced in contemporary catheterisation laboratories. Furthermore, coverage for women who become parents during training should be implemented proactively, and standardised, so that the burden of guilt for the pregnant interventionalist is lifted and so that this becomes routine practice rather than an inconvenience to the rest of the trainees. These measures can include increased pay for those covering, as well as curricular advances to ensure that training for these women is not prolonged.
Providing active dosimeters to help reduce exposure would be an important step and would protect not only pregnant women but all operators.
Indeed, gender equity should be a common interest for both men and women. Male role models and mentors need to show support based on talent and accomplishments irrespective of gender. The Pygmalion effect, where prejudiced expectations impact outcomes, needs to be of less influence when recruiting or collaborating with female colleagues. Highly qualified female interventionists should be assumed to be as equally apt as their male counterparts in performing the job and should be offered similar opportunities with equal pay. There must be a joint effort to revoke gender as a criterion for promotion or leadership. The only way to achieve equity is through a collaborative leadership that ensures equality between men and women at all career and life stages.
Conclusions
Fear of foetal exposure to ionising radiation during pregnancy remains a barrier for women who wish to pursue a career in interventional cardiology. International expert commission recommendations and European directives clearly state that pregnant women can continue to work in an ionising radiation environment providing that the foetus does not exceed certain dose thresholds. Moreover, data from practice, although scarce, confirm radiation doses to be well below these limits. Despite this, many countries apply inappropriately restrictive directives. Specific institutional radiation protection programs should be established to help to overcome radiation barriers by including specific safety requirements for everyone; this would also ensure safe exposure during pregnancy. Key opinion makers in cardiovascular societies and ancillary institutions should promote awareness at both the local and national level to ensure a level playing field and a friendly environment for pregnant interventionists. This would help to facilitate the continuation of interventional work during pregnancy and go towards eliminating this cause of gender inequity in invasive cardiology subspecialities.
Conflict of interest statement
S. Manzo-Silberman has received consulting fees from Bayer, Organon, and Exeltis; lecture fees from Bayer, BMS, Exeltis, and Organon; and has served on the adjudication board for a study for Biotronik. S. Burgess received a research a grant from Women as One for a radiation safety project (paid to her to university research fund), and has received speaker honoraria from AstraZeneca, Women as One, Pfizer, and Novartis. S. Sahni has received lecture fees from Abbott Vascular. P. Best has served as co-Chair of SCAI-WIN and Chair of the SCAI CME Oversight Committee, and is a member at Mission Lifeline. R. Mehran reports institutional research payments from Abbott, Abiomed, Alleviant Medical, AM-Pharma, Amgen, Applied Therapeutics, Arena, AstraZeneca, AtriCure, Bayer, Biosensors, Biotronik, Boston Scientific, Bristol-Myers Squibb, CardiaWave, CeloNova, Chiesi, Concept Medical, CSL Behring, Cytosorbents, Daiichi Sankyo, Element Science, Faraday, Humacyte, Idorsia, Janssen, Magenta, Medtronic, Novartis, OrbusNeich, PhaseBio, Philips, Pi-Cardia, RenalPro, RM Global, Shockwave, Vivasure, and Zoll; personal fees from Cine-Med Research Institute and WebMD; has equity <1% in Applied Therapeutics, Elixir Medical, Stel, and ControlRad (spouse); is on the Scientific Advisory Board of AMA; is on the Board of Trustees of the American College of Cardiology; is a Women in Innovations Committee Member for SCAI; is Associate Editor of JAMA; and is a member of the Cardiovascular Research Foundation faculty (no fee). L. Vitali-Serdoz has received lecture fees from Abbott Vascular and Medtronic. A. Sarma has received a grant from CRICO Patient Safety Award. J. Mauri is a stakeholder of CERC CRO. P. Szymański has received lecture fees from Novartis and Abbott Vascular. G. Stefanini has received institutional grant support from Boston Scientific paid to his institution and, has received lecture fees from Abbott Vascular, Boston Scientific, and Pfizer/BMS. A. Gimelli has served on the Pfizer Advisory Board for amyloidosis and the GE Healthcare Advisory Board for the safety of regadenoson in paediatric patients. P. Maurovich-Horvat is shareholder of Neumann Medical Ltd. L. Boersma has received consulting fees from Medtronic, Boston Scientific, Adagio, Philips, and Abbott paid to his institution; has served in the Committee of Science and Innovation for the Dutch Society of Cardiology, as Associate Editor of Europace (no fee) and in the EHRA Scientific Documents Committee (no fee). G.L. Buchanan has received support for attending meetings and/or travel from Menarini. G. Pontone has received grants from GE Healthcare, Bracco, Boehringer Ingleheim, and HeartFlow; has received consulting and lecture fees from GE Healthcare, Bracco, and Boehringer; and payment for expert testimony from GE Healthcare. L. Holmvang has received lecture fees from MicroPort and Bayer, and support for attending meetings from Abbott. N. Karam has received consulting fees from Abbott Vascular, Medtronic, and Edwards Lifesciences; has received lecture fees from Abbott Vascular and Edwards Lifesciences; has received support for attending meetings from Abbott Vascular; and has served on an advisory board for Medtronic. A. Neylon holds shares/stock options in CERC Europe. M-C. Morice is CEO and shareholder of CERC (not involved in this article) and is a minor shareholder of Electroducer. G. Tarantini has received consulting fees from Medtronic, GADA, Edwards Lifesciences, Boston Scientific, and Abbott Vascular; and has received lectures fees from Medtronic, GADA, Edwards Lifesciences, Boston Scientific, and Abbott Vascular. A. Chieffo has received consulting fees from Abiomed; and speaker fees from Abiomed, Abbott Vascular, and Biosensors; and has served on an advisory board for Shockwave Medical. The other authors have no conflicts of interest to declare

