Abstract
Aims: We sought to explore the immediate and long-term outcome of combined percutaneous valvuloplasty of the mitral and/or aortic and/or tricuspid valves in a series of patients with rheumatic valvular stenosis.
Methods and results: A total of 11 patients (three underwent percutaneous mitral valvuloplasty [PMV], percutaneous aortic valvuloplasty [PAV] and percutaneous tricuspid valvuloplasty [PTV], six underwent PMV and PAV, and two underwent PMV and PTV) were enrolled. PMV was performed by the standard double balloon technique. PAV was always performed after PMV, employing the retrograde approach in eight patients and the antegrade approach in one patient. PTV was performed by the double balloon technique. Echocardiographic assessment was performed before and after the procedures. Follow-up was performed in all patients for a period that ranged from 12 and up to 60 months. PMV was successful in 10 out of 11 cases (91%); PAV was successful in all nine procedures (100%), while PTV was successful in four out of five cases (80%). At long-term follow-up, one case of restenosis occurred following PMV (9%), two following PTV (40%), and no restenosis occurred following PAV.
Conclusions: Percutaneous balloon dilatation of rheumatic valvular stenosis is feasible with fairly adequate immediate and long-term outcome.
Introduction
Rheumatic valvular disease continues to be endemic in developing countries, with mitral stenosis being the most frequent valve disorder1. During the past two decades, percutaneous mitral valvuloplasty (PMV) has emerged as the procedure of choice in most patients with symptomatic mitral stenosis2. Several large series reported an excellent outcome both in the short and long run3-7, as well as a low incidence of serious complications8. Moreover, in patients with mitral valve characteristics suitable for balloon valvuloplasty, results of randomised trials comparing PMV with surgical commissurotomy (both open and closed) have demonstrated comparable clinical, echocardiographic and haemodynamic outcome between the two therapeutic strategies, added to the shorter hospital stay and lower morbidity and mortality in case of PMV9-13.
Percutaneous aortic valvuloplasty (PAV) for degenerative calcific aortic stenosis has been associated with modest increase of valve area, along with a high toll of in-hospital mortality (3.5-13.5%) and morbidity (20-25%)14,15. Over and above, improvement of functional status after PAV has been moderate and transient, lasting only for a few months16,17. Percutaneous tricuspid valvuloplasty (PTV) is feasible; however, cases reported to date are so scarce that it is difficult to draw adequate conclusions regarding the safety and efficacy of the procedure on the short and long-term18.
Several previous small series and case reports about combined percutaneous mitral and tricuspid valvuloplasty have demonstrated feasibility, safety, efficacy of the procedure with excellent immediate outcome, and benefit being maintained on the intermediate term19-24. Case reports of combined aortic/mitral and combined aortic/tricuspid have also shown feasibility and safety25,26. A few cases of combined triple valvuloplasty (mitral, aortic and tricuspid) are reported in the literature to date27,28.
In a retrospective study design, we sought to explore the immediate and long-term outcome (assessed by echocardiography) of combined percutaneous valvuloplasty of the mitral and/or aortic and/or tricuspid valves in a series of patients with rheumatic multi-valvular stenosis.
Methods
Population
We enrolled 11 patients with moderate to severe rheumatic mitral stenosis (defined as mitral valve area less than 1.5 cm2) associated with severe aortic stenosis (defined as peak systolic pressure gradient across the aortic valve ≥ 60 mmHg by Doppler echocardiography) and/or significant tricuspid stenosis (defined as mean diastolic pressure gradient across the tricuspid valve ≥5 mmHg by Doppler echocardiography) admitted to our cathlabs during the period from December 1988 to June 1993, to undergo PMV. Patients were considered eligible for PMV if they had a Massachusetts General Hospital (MGH) score of the mitral valve ≤1119 with no more than grade 2/4 mitral regurgitation (MR), for PAV if they had no more than grade 1/4 aortic regurgitation (AR) with only minimal calcification of the valve (we excluded patients with aortic calcification evident by fluoroscopy), and for PTV if they had no more than grade 1/4 tricuspid regurgitation (TR) as assessed by echocardiography. We excluded patients with previous PMV or previous valve surgery, patients with concomitant valve disease that needs surgical intervention, patients indicated for coronary artery bypass surgery, and patients with limited life expectancy due to coexistent disease (for example: malignancy). Before inclusion, an informed consent was obtained from each patient and the study protocol was reviewed and approved by our local institutional human research committee as conforming to the ethical guidelines of the 1975 Declaration of Helsinki, as revised in 2000.
Methods
Before enrolment, all patients were assessed by transthoracic echocardiography within 24 hours of admission. Doppler echocardiography was performed using a Hewlett Packard Sonos 1000 cardiac ultrasound machine (Hewlett Packard, Palo Alto, CA, USA). A 2.5 MHz phased array probe was used to obtain standard 2D, and Doppler images. Patients were examined in the left lateral recumbent position using standard parasternal and apical views. The mitral, aortic and tricuspid valves were specially evaluated regarding the valve area, transvalvular pressure gradient and the presence – if any – and grade of valvular regurgitation. Scoring of the mitral valve was done by the MGH scoring system29. Calcification of the aortic valve was evaluated.
Beforehand, patients in atrial fibrillation received oral anticoagulants for six weeks before the procedure aiming at an INR 2-3, to minimise the risk of embolisation. Oral anticoagulants were stopped for several days before the procedure till the INR decreased below 1.5.
PMV was performed in all patients by the standard double balloon technique based on the method described by Al Zaibag et al30 and modified by Palacios et al31. Balloon size was chosen according to mitral annular diameter (measured by echocardiography) as follows: sum of diameters of the two balloons equals mitral annular diameter. Right and left heart haemodynamic data were recorded before and after the procedure.
PAV was performed always after PMV, employing the retrograde approach in eight patients and the antegrade approach in one patient, without additional heparin being given for the second procedure. Balloon size of 25 mm was adopted for all patients. Measurement of the peak systolic pressure gradient across the aortic valve was done before and after the procedure.
PTV was performed always following PMV without additional heparin being given for the second procedure, and in the three cases where all three valves were dilated; PMV and PAV were performed in one session while PTV was postponed to another separate session. PTV was performed by the double balloon technique. Balloon size of 20 mm was adopted for either balloon. Measurement of the mean diastolic pressure gradient across the tricuspid valve was done before and after the procedure.
Echocardiographic assessment was performed 24 hours following each procedure to evaluate the final valve area, transvalvular pressure gradient, and the presence and grade of valve regurgitation, if any, for the index valves.
Follow-up
Follow-up by clinical examination and echocardiographic assessment was performed in all patients for a period that ranged from 12, and up to 60 months. Echocardiographic assessment was done to evaluate the valve area, transvalvular pressure gradient, and the presence and grade of valve regurgitation, if any, for the index valves.
Definitions
Definitions of procedural success were based on transthoracic echocardiography findings. Procedural success of PMV was defined as an increase of 50% or more of mitral valve area assessed by planimetry method, with a final mitral valve area equal to, or more than, 1.5 cm2 and no more than 1 grade increment of MR severity, if any. Procedural success of PAV was defined as a drop of ≥50% of the peak transvalvular pressure gradient and no more than 1 grade increment of AR severity, if any. Procedural success of PTV was defined as a drop of 50% or more of the mean transvalvular pressure gradient with no more than 1 grade increment of TR severity, if any. Restenosis was defined as loss of 50% or more of the final valve area estimated immediately following the procedure, for the mitral valve and an increase of 50% or more of the transvalvular pressure gradient estimated immediately following the procedure for both the aortic and the tricuspid valves.
Statistical analysis
All continuous variables were presented as means±SD, if they were normally distributed. Differences in the normally distributed variables were assessed using the t-test and the paired t-test for dependent variables. Categorical variables were described with absolute and relative (percentage) frequencies. Comparisons between echocardiographic data at baseline and those immediately after the procedure were performed by the paired t-test for parametric and Wilcoxon’s test for non-parametric values. A probability value of P<0.05 was considered statistically significant. Analyses were performed with SPSS version 12.0 statistical package (SPSS Inc., Chicago, IL, USA).
Results
A total of 11 patients with moderate to severe mitral stenosis who underwent PMV were included in the current study. Of the total series, three patients underwent PMV, PAV and PTV, six patients underwent PMV and PAV, and only two patients underwent PMV and PTV (a total of 25 procedures). The procedures were well tolerated in all patients with no serious complications. Table 1 shows the baseline characteristics of the patients. The mean age of the study patients was 36.4±7.9 years, 45% (five patients) being females. Three patients (27.3%) were in atrial fibrillation. The mean value of MGH score of the mitral valve was 8±1.3.
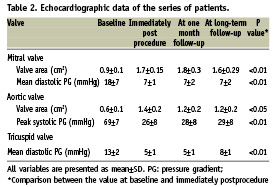
Table 2 shows the echocardiographic data of the series of patients before, immediately after the procedure, at one month and at long term follow-up. According to our prespecified definitions, the procedure of PMV was successful in 10 out of 11 cases (91%), with a mean final mitral valve area of 1.74±0.15 cm2, and a mean diastolic pressure gradient across the valve of 7±1.2 mmHg immediately after the procedure. One case had a suboptimal mitral valve area (1.4 cm2) immediately after the procedure, and one case developed new grade 1/4 MR. The procedure of PAV was successful in all nine procedures (100%), with a mean final aortic valve area of 1.4±0.2 cm2, and a peak systolic pressure gradient across the valve of 26±8.1 mmHg immediately after the procedure. In only one case, the AR grade increased from 1/4 to 2/4. The procedure of PTV was successful in four out of five cases (80%), with a mean diastolic pressure gradient across the valve of 5.4±1.1 mmHg immediately after the procedure. Tricuspid regurgitation increased by 1 grade in four out of five cases, and by 2 grades in one case (suboptimal result).
Follow-up period ranged from 12 and up to 60 months (mean 23.5±14.3, median 24 months). No mortality or serious complications occurred during follow-up, except for one case (35-years-old female with atrial fibrillation at baseline and a suboptimal result of PMV) who developed acute pulmonary embolism six months after the procedure of combined PMV and PTV, and improved later on. According to our prespecified definitions, one case of restenosis occurred following PMV (9%), two cases of restenosis occurred following PTV (40%), and no cases of restenosis occurred following PAV. Valve regurgitation grade at long-term follow-up remained the same as immediately following the procedures.
At long term follow-up, the mean mitral valve area was 1.64±0.29 cm2, and the mean diastolic pressure gradient across the valve was 7.2±2.1 mmHg, the mean aortic valve area was 2.1±0.2 cm2, and the peak systolic pressure gradient across the valve was 29.8±8.1 mmHg, while the mean diastolic pressure gradient across the tricuspid valve was 8±1.6 mmHg.
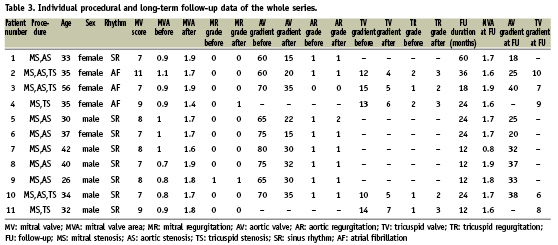
Individual procedural and long-term follow-up data of the whole series are presented in Table 3.
Discussion
This study demonstrated that percutaneous balloon valvuloplasty is both feasible and safe for stenosis of the mitral, aortic and tricuspid valves of rheumatic origin. It is well established that PMV is the procedure of choice in young patients with favourable anatomy18, or when surgery is contraindicated or very high risk32. Several parameters were previously put forward for selection of patients with “favourable anatomy”. At the time of this study, we adopted the MGH scoring system proposed by Wilkins et al, for predicting outcome of PMV, based on four parameters, namely, leaflet thickness, mobility restriction, calcification and subvalvular affection29. Recent reports have emphasised the importance of commissural morphology (chiefly commissural calcification) and subvalvular involvement in determining immediate outcome of PMV33-35. More recently, a novel scoring system was suggested based only on these two later parameters and better predicted outcome after the procedure36. Excluding other parameters (namely, leaflet thickness and mobility) not closely related to the outcome of PMV would have improved the immediate success rate of the procedure in the current series (91%), and reduced restenosis rate at long-term follow-up (9%).
PAV has been abandoned by many groups as a treatment option for severe degenerative calcific aortic stenosis of adults37. Others still consider this approach for critically-ill patients with cardiogenic shock and multiple organ failure38. According to a few reports, it can serve as a bridge to secondary operation with good midterm results39. It might also have a limited role in patients who must undergo emergency non-cardiac surgery, in those who refuse surgery, and as a palliative procedure in those with short term absolute contraindication for surgery who have significant disability18. In contrast, rheumatic aortic stenosis is characterised by commissural fusion which – in the absence of calcification or with minimal calcification – would easily yield under balloon dilatation, a mechanism exactly similar to PMV. This notion was clearly supported by the results of the current study that demonstrated good immediate results and absence of restenosis at long-term follow-up of up to five years, in patients with minimal calcification. In view of the current results, we would suggest that patients with rheumatic severe aortic stenosis and minimal calcification should be given a chance for PAV, particularly in young females with child-bearing potential, in order to defer the need for valve replacement whether by the surgical or percutaneous route.
Cases of PTV reported to date in literature are too rare to provide adequate data about the procedure outcome. The paucity of cases of tricuspid stenosis and its frequent association with TR may be responsible for the rare employment of the procedure. The results of the current study demonstrated modest decrease of the transvalvular mean pressure gradient, at the expense of increasing the grade of TR in all patients (in one case by 2 grades). Restenosis occurred in two cases out of five. Based on these results, the procedure of PTV cannot be recommended as a definitive treatment for rheumatic tricuspid stenosis, even in patients with no or mild TR. Yet, it might serve as an acceptable approach to defer surgery in selected patients with high surgical risk as those with marked liver insufficiency, a common consequence of severe tricuspid stenosis.
Previously, a few small series19-21 and sporadic case reports22-28 have demonstrated the feasibility and safety of the combined valvuloplasty procedure. To our knowledge, this series is the largest one of combined valvuloplasty procedure (25 procedures) reported to date in the literature. Being the most common, mitral stenosis was present in all patients. We started always with PMV before PAV in order to perform transseptal puncture (a pivotal step of PMV) well before heparin administration (necessary for PAV), to keep active the coagulation mechanism in case of inadvertent occurrence of haemopericardium as a complication of transseptal puncture. PTV was also performed after PMV in order to reassess the haemodynamic parameters (gradient across the tricuspid valve) which are liable to change after successful PMV and drop of the pulmonary artery pressure. When all three valves required valvuloplasty, we performed PMV and PAV in one setting and postponed PTV to another setting to avoid exhaustion of patients and operators.
Balloon size for PMV was chosen according to the mitral annular diameter measured by echocardiography. For PAV, we adopted a balloon size of 25 mm for all patients. The balloons used for PTV were not necessarily those used for PMV in the same patient, yet, two balloons (20 mm each) were always used for PTV, given the larger tricuspid valve area. Finally, we adopted always the double balloon technique for PMV, and it is not clear whether the Inoue technique would give a better outcome in the setting of combined valvuloplasty.
Conclusions
The procedure of multiple percutaneous valvuloplasty for rheumatic valvular stenosis is feasible and safe. Outcome is excellent for PMV, and for PAV in patients with minimal calcification of the aortic valve; both on the short and long term. PTV is associated with increase of TR grade and a high incidence of restenosis at long-term follow-up.
Study limitations
Our findings are based on a single centre study with a relatively small sample size of the cohort, a fact that makes it difficult to generalise our results to all patients with multi-valvular stenosis. Multicentre studies using the same protocol and examining a larger number of patients are needed before reaching solid conclusions. Moreover, all our patients underwent PMV with the double balloon technique, and it is unknown whether our data can be safely extrapolated to patients undergoing Inoue PMV and patients undergoing percutaneous metallic valvotomy. Finally, selection of patients with mitral stenosis did not employ the recently ascertained determinants of outcome such as commissural calcification, a fact that may certainly influence outcome.
Acknowledgements
We would like to express our deep gratitude to all the staff members of the cathlabs in the cardiology department, Ain Shams University Hospitals, who have sincerely and rigorously supported the performance of this work.

