
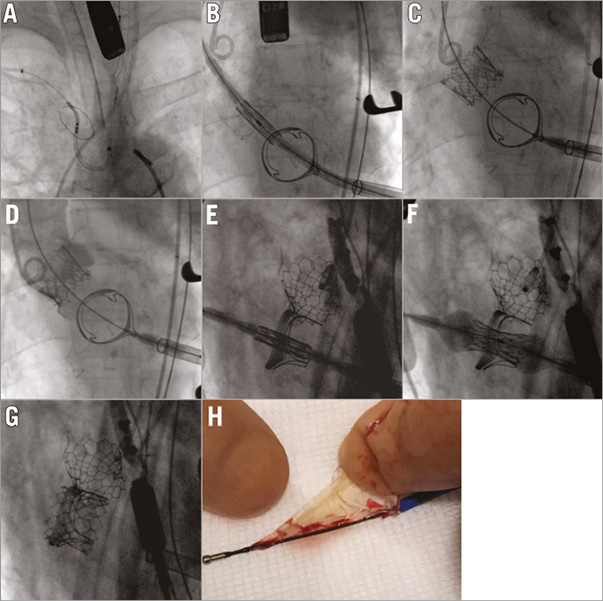
A patient with degenerated aortic (Trifecta™ 23 mm; St. Jude Medical, St. Paul, MN, USA) and mitral (Edwards PERIMOUNT 25 mm; Edwards Lifesciences, Irvine, CA, USA) valve bioprostheses was referred and deemed high risk for open heart surgery (logistic EuroSCORE 17.6% and frailty). The Heart Team decided on transapical double valve-in-valve.
Due to the heavy calcification on the degenerated leaflets, a cerebral protection device (Sentinel CPS; Claret Medical, Santa Rosa, CA, USA) was used (Panel A). Two 23 mm Edwards SAPIEN 3 valves (Edwards Lifesciences) were directly deployed (no balloon valvuloplasty) in the failing aortic prosthesis (reduction of mean gradient from 40 mmHg to 10 mmHg, trace AR) (Panels B-D, Moving images 1-3) and failing mitral prosthesis (reduction of mean transvalvular gradient from 22 mmHg to 3.5 mmHg) (Panels E-G, Moving images 4-6). A thrombotic mass was found in the filter retrieved from the left carotid artery (Panel H). There was no evidence of periprocedural stroke. The patient was discharged at day 14, and 30-day follow-up was uneventful.
Conflict of interest statement
A. Wolf and T. Schmitz work as in-house proctors for Edwards Lifesciences. The other authors have no conflicts of interest to declare.
Supplementary data
Moving image 1. Positioning of 23 mm Edwards SAPIEN 3 valve in degenerated aortic bioprosthesis.
Moving image 2. Implantation in aortic position.
Moving image 3. Aortography.
Moving image 4. Positioning of 23 mm Edwards SAPIEN 3 valve in degenerated mitral bioprosthesis.
Moving image 5. Implantation in mitral position.
Moving image 6. Rotational view of implanted valves.
Supplementary data
To read the full content of this article, please download the PDF.
Moving image 1. Positioning of 23 mm Edwards SAPIEN 3 valve in degenerated aortic bioprosthesis.
Moving image 2. Implantation in aortic position.
Moving image 3. Aortography.
Moving image 4. Positioning of 23 mm Edwards SAPIEN 3 valve in degenerated mitral bioprosthesis.
Moving image 5. Implantation in mitral position.
Moving image 6. Rotational view of implanted valves.

