Abstract
Background: Current guidelines recommend beta blockers after myocardial infarction (MI) regardless of left ventricular ejection fraction (LVEF), aiming to reduce reinfarction and ventricular arrhythmias. However, recent trials have challenged this practice in patients without reduced LVEF. Whether beta blocker withdrawal in these patients increases short-term or recurrent ischaemic events remains uncertain.
Aims: We aimed to evaluate the short-term ischaemic safety of beta blocker withholding or withdrawal at hospital discharge in patients with MI and LVEF >40% and to determine the effect of beta blocker therapy on a broad composite ischaemic endpoint.
Methods: This is a post hoc analysis of the REBOOT trial, in which patients with MI and LVEF >40% were randomised to beta blocker therapy or no beta blocker at discharge. The incidence of short-term (3-month) and recurrent ischaemic events (a composite of cardiac death, reinfarction, sustained ventricular tachycardia/fibrillation, resuscitated cardiac arrest, or unplanned revascularisation) was assessed overall and according to prior beta blocker use.
Results: From the 8,438 patients in the intention-to-treat population of the trial, information regarding beta blocker history was available for 8,401. Of these, 12.1% were on chronic beta blocker therapy before MI. Overall, withholding or withdrawing beta blockers was not associated with increased short-term ischaemic risk (hazard ratio [HR] 1.13, 95% confidence interval [CI]: 0.74-1.72). Over a median follow-up of 3.7 years, there were no differences in recurrent ischaemic events between groups (HR 0.98, 95% CI: 0.82-1.16), nor significant interactions with prior beta blocker therapy. In patients who were on a beta blocker before the index MI, randomisation to no beta blocker (withdrawal) was not associated with an increased risk of ischaemic events during trial follow-up (composite ischaemic endpoint HR 0.93, 95% CI: 0.64-1.34).
Conclusions: In patients with MI and LVEF >40%, beta blocker withholding or withdrawal at discharge was not associated with increased short-term or recurrent ischaemic events, supporting the safety of this strategy in contemporary clinical practice.
Beta blockers have long been a cornerstone of post-myocardial infarction (MI) therapy. For decades, both European and American clinical guidelines for the management of acute coronary syndrome (ACS) have recommended their use following acute MI, regardless of left ventricular ejection fraction (LVEF)12. However, these recommendations were largely based on evidence from trials conducted in an earlier era – prior to the widespread adoption of modern reperfusion strategies, potent dual antiplatelet therapy (DAPT), high-intensity statins, and comprehensive secondary prevention protocols3. Consequently, the current clinical relevance of beta blockers in this population has been increasingly called into question.
Two recent large randomised clinical trials – tREatment with Beta blockers after myOcardial infarction withOut reduced ejection fracTion (REBOOT) and Randomized Evaluation of Decreased Usage of Beta blockers after Acute Myocardial Infarction (REDUCE-AMI) – have specifically addressed this issue. Both studies demonstrated a lack of prognostic benefit from routine beta blocker therapy in patients without moderately or severely reduced LVEF after an MI45. Notably, in both trials, a substantial proportion of participants were already receiving beta blockers prior to the index infarction. The abrupt discontinuation of beta blockers in patients with known or suspected ischaemic heart disease has historically raised concerns due to the potential for rebound phenomenon, which may precipitate early ischaemic events6. In fact, the U.S. Food and Drug Administration (FDA) label for beta blockers includes a warning in this regard. However, this warning is based on older studies conducted in an era before routine reperfusion, complete revascularisation, and optimised secondary prevention pharmacotherapy (such as potent DAPT, high-intensity statins, etc.) were widely implemented3. To address this concern, the design of the REBOOT trial incorporated an early clinical follow-up visit at 3 months post-randomisation7. This was intended to capture any signal of early harm associated with beta blocker withholding or withdrawal, particularly during the vulnerable period immediately following the acute event, when patients are at the highest risk for recurrent ischaemic complications.
The recent 2025 ACC/AHA/ACEP/NAEMSP/SCAI ACS guidelines, which were released after the results of REDUCE-AMI but before those of REBOOT were public, continue to endorse beta blockers after MI (with the strongest class of recommendation), aiming “to reduce risk of reinfarction and ventricular arrhythmias”2. These are considered specific ischaemic events.
The present study had two main objectives: (1) to assess whether beta blocker withholding (no prescription at discharge) or withdrawal (abrupt discontinuation at discharge in patients previously on therapy) raised any short-term (3-month) ischaemic concerns and (2) to evaluate the effect of beta blocker therapy on a broad composite ischaemic endpoint, including recurrent events, throughout the entire study follow-up. To these aims, we used data from the REBOOT trial, the largest contemporaneous trial (n=8,505) randomising patients after MI without reduced LVEF to beta blocker therapy or control (no beta blocker). The main results of the trial have been published elsewhere4.
Methods
Study design, participants and study intervention
REBOOT was a pragmatic, controlled, prospective, randomised open blinded endpoint (PROBE) trial conducted in Spain and Italy7. Treatment allocation was not masked, but clinical outcomes were centrally adjudicated by a committee blinded to treatment allocation. Key information on the study design and their inclusion and exclusion criteria can be found elsewhere7. The clinical trial registration numbers are ClinicalTrials.gov: NCT03596385 and EudraCT: 2017-002485-40. Patients with myocardial infarction (with ST-segment elevation MI [STEMI] or non-STEMI [NSTEMI]) were eligible if they underwent invasive management during the index MI hospitalisation and had a left ventricular ejection fraction greater than 40%. Patients were excluded if they had a history of heart failure or already had either a contraindication to or an indication for beta blocker therapy. Participants were randomly assigned to receive either beta blocker therapy (type and dose of beta blocker at the discretion of the managing physician) or no beta blocker therapy (control). No target dose or guidance on dose adjustment based on heart rate was specified in the protocol. All patients received standard care. Randomisation was performed at the time of hospital discharge or within the subsequent 14 days.
Follow-up and clinical adjudicated outcomes
Follow-up assessments of clinical outcomes were conducted at 3±1, 15±3, 36±3, and 48±6 months after randomisation, as detailed elsewhere47. We used the first follow-up assessment at 3 months to evaluate the potential rebound phenomenon upon abrupt beta blocker withdrawal, as well as the entire follow-up period to evaluate the long-term effects of beta blockers on ischaemic endpoints. The original primary outcome was a composite of all-cause death, reinfarction, or heart failure admission7. In this article, we focused on the non-prespecified broader ischaemic composite endpoint including cardiac death, reinfarction, sustained ventricular tachycardia/fibrillation (VT/VF), resuscitated cardiac arrest, or unplanned revascularisation. In addition, we analysed the prespecified secondary outcomes of all-cause death, cardiac death, non-fatal reinfarction, unplanned revascularisation, stroke, and the composite of sustained VT/VF or resuscitated cardiac arrest.
Statistical analysis
Baseline characteristics by prior beta blocker therapy are described as frequencies (%) and were compared using chi-square tests for categorical data, whereas baseline continuous data are expressed as mean and standard deviation (SD) or median with interquartile range and were compared using t-tests or Wilcoxon rank-sum tests, as appropriate.
The short-term incidence of ischaemic endpoints was analysed up to 3-month follow-up using Cox proportional hazard models to produce unadjusted hazard ratios (HRs) and 95% confidence intervals (CIs), with a log-rank test of significance. Lin Wei Yang Ying (LWYY) models were used to assess the incidence of recurrent ischaemic events throughout the trial8. In addition, the order of total events was analysed (time to first, second, and third event) using the Wei-Lin-Weissfeld model9. Multiple events on the same day were counted as one event.
To assess whether the effect of beta blockers was different according to prior beta blocker therapy and other baseline characteristics of interest, subgroup analyses were performed by including an interaction term between the subgroup of interest and randomised therapy in the model. A p-value of less than 0.05 was considered statistically significant. All analyses were performed using Stata, version 18.5 (StataCorp).
Results
From the 8,505 patients enrolled in the REBOOT trial, 8,438 formed the intention-to-treat (ITT) population. Of the 8,401 MI patients without reduced LVEF (>40%) enrolled in the trial with available information on previous beta blocker treatment, 1,019 (12.1%) were on chronic beta blocker therapy before the index event. Compared with patients not on treatment before the index MI, those with prior beta blocker therapy were older, more frequently female, and presented with a higher burden of cardiovascular comorbidities including hypertension (72.8% vs 49.0%), diabetes (27.6% vs 20.5%), prior MI (37.7% vs 5.6%), and atrial fibrillation (6.8% vs 1.7%) (Table 1). They also more frequently presented with NSTEMI (62.4% vs 47.2%) and had lower rates of complete revascularisation during index admission (82.2% vs 89.1%). Patients on chronic beta blocker therapy before their index MI had a slightly lower mean LVEF (56.0% vs 57.3%; p<0.001) and lower estimated glomerular filtration rate (84.4 mL/min/1.73 m2 vs 90.6 mL/min/1.73 m2; p<0.001).
At discharge, the prescription rates of guideline-directed medical therapy were generally high in both groups, though among patients with a prior beta blocker, a smaller percentage were discharged on DAPT and statins and a higher proportion on angiotensin-converting enzyme (ACE) inhibitors or diuretics relative to those without a prior beta blocker. As expected, due to randomisation, the proportion of patients discharged on beta blockers was similar in both groups of patients − those taking beta blockers or not before the index MI − (49.7% vs 48.9%; p=0.64), although the distribution of agents and dosages differed significantly (Table 1).
Table 1. Baseline characteristics of patients stratified by prior beta blocker use and randomised treatment groups.
| Prior beta blockers | Without prior beta blockers | |||
|---|---|---|---|---|
| Beta blocker (N=510) | No beta blocker (N=509) | Beta blocker (N=3,682) | No beta blocker (N=3,700) | |
| Demographics | ||||
| Age, years | 64.4±10.9 | 65.4±11.5 | 60.9±11.1 | 60.8±10.9 |
| Sex | ||||
| Male | 399 (78.2) | 397 (78.0) | 2,982 (81.0) | 3,004 (81.2) |
| Female | 111 (21.8) | 112 (22.0) | 700 (19.0) | 696 (18.8) |
| Country of enrolment | ||||
| Spain | 224 (43.9) | 216 (42.4) | 3,021 (82.0) | 3,046 (82.3) |
| Italy | 286 (56.1) | 293 (57.6) | 661 (18.0) | 654 (17.7) |
| Medical history | ||||
| Hypertension | 374 (73.5) | 367 (72.1) | 1,802 (49.0) | 1,810 (49.0) |
| Diabetes mellitus | 143 (28.2) | 137 (27.0) | 753 (20.5) | 751 (20.4) |
| Dyslipidaemia | 319 (62.5) | 295 (58.1) | 1,830 (49.8) | 1,863 (50.4) |
| Smoking status | ||||
| Non-smoker | 182 (37.2) | 200 (40.1) | 1,132 (31.5) | 1,149 (31.9) |
| Smoker | 157 (32.1) | 161 (32.3) | 1,687 (46.9) | 1,660 (46.0) |
| Ex-smoker <1 yr | 30 (6.1) | 42 (8.4) | 219 (6.1) | 223 (6.2) |
| Ex-smoker >1 yr | 120 (24.5) | 96 (19.2) | 557 (15.5) | 574 (15.9) |
| Prior MI | 211 (41.5) | 172 (33.9) | 192 (5.2) | 218 (5.9) |
| Prior stroke | 19 (3.7) | 18 (3.5) | 65 (1.8) | 48 (1.3) |
| Atrial fibrillation prior to admission | 30 (5.9) | 39 (7.7) | 60 (1.6) | 62 (1.7) |
| Peripheral arterial disease | 36 (7.1) | 32 (6.3) | 91 (2.5) | 79 (2.1) |
| History of COPD | 23 (4.5) | 22 (4.3) | 122 (3.3) | 110 (3.0) |
| Index admission | ||||
| MI type | ||||
| STEMI | 183 (35.9) | 200 (39.3) | 1,959 (53.2) | 1,939 (52.4) |
| NSTEMI | 327 (64.1) | 309 (60.7) | 1,723 (46.8) | 1,761 (47.6) |
| Infarct-related artery | ||||
| None | 18 (3.5) | 16 (3.1) | 82 (2.2) | 95 (2.6) |
| Left anterior descending | 143 (28.1) | 123 (24.2) | 987 (26.9) | 1,014 (27.5) |
| Left circumflex | 68 (13.4) | 63 (12.4) | 462 (12.6) | 428 (11.6) |
| Right coronary artery | 121 (23.8) | 127 (25.0) | 961 (26.2) | 985 (26.7) |
| Secondary | 27 (5.3) | 18 (3.5) | 213 (5.8) | 200 (5.4) |
| Left main | 7 (1.4) | 6 (1.2) | 23 (0.6) | 24 (0.7) |
| Multivessel | 124 (24.4) | 156 (30.6) | 944 (25.7) | 944 (25.6) |
| Type of revascularisation | ||||
| None | 44 (8.7) | 33 (6.5) | 161 (4.4) | 156 (4.3) |
| PCI – stent | 433 (85.2) | 455 (89.7) | 3,400 (93.0) | 3,392 (92.5) |
| Complete revascularisation | 396 (82.2) | 401 (82.3) | 3,058 (88.8) | 3,071 (89.3) |
| Echo and laboratory tests | ||||
| LVEF, % | 56.0±6.8 | 56.1±7.0 | 57.1±7.1 | 57.4±7.1 |
| Creatinine prior to discharge, mg/dL | 1.0±0.4 | 1.0±0.5 | 0.9±0.3 | 0.9±0.4 |
| eGFR, mL/min/1.73 m2 | 85.6±20.2 | 83.3±21.1 | 90.3±18.1 | 91.0±17.7 |
| Haemoglobin prior to discharge, g/dL | 13.8±1.7 | 13.7±1.8 | 14.3±1.6 | 14.3±1.6 |
| Discharge medication | ||||
| Aspirin | 492 (96.5) | 490 (96.3) | 3,629 (98.7) | 3,656 (98.9) |
| P2Y12 inhibitor | 493 (96.7) | 491 (96.5) | 3,613 (98.2) | 3,618 (97.9) |
| ACEi/ARB | 393 (77.1) | 426 (83.9) | 2,639 (71.9) | 2,831 (76.6) |
| Statin | 493 (96.7) | 496 (97.4) | 3,622 (98.5) | 3,645 (98.6) |
| Aldosterone receptor antagonist | 20 (3.9) | 19 (3.7) | 73 (2.0) | 65 (1.8) |
| Oral anticoagulant | 42 (8.2) | 42 (8.3) | 127 (3.5) | 120 (3.3) |
| Diuretic | 71 (13.9) | 87 (17.1) | 292 (8.0) | 320 (8.7) |
| Type of beta blocker | ||||
| Atenolol | 14 (2.8) | 12 (0.3) | ||
| Bisoprolol | 345 (68.2) | 3,191 (88.4) | ||
| Carvedilol | 25 (4.9) | 103 (2.9) | ||
| Metoprolol | 85 (16.8) | 223 (6.2) | ||
| Nebivolol | 35 (6.9) | 78 (2.2) | ||
| Other | 2 (0.4) | 3 (0.1) | ||
| Beta blocker dose | ||||
| ≤Median dosage | 363 (71.9) | 3,168 (88.2) | ||
| >Median dosage | 142 (28.1) | 425 (11.8) | ||
| Data are mean±SD or n (%). ACEi: angiotensin-converting enzyme inhibitor; ARB: angiotensin II receptor blocker; COPD: chronic obstructive pulmonary disease; eGFR: estimated glomerular filtration rate; LVEF: left ventricular ejection fraction; MI: myocardial infarction; NSTEMI: non-ST-segment elevation myocardial infarction; PCI: percutaneous coronary intervention; SD: standard deviation; STEMI: ST-segment elevation myocardial infarction | ||||
Short-term incidence of ischaemic events
At 3 months, the incidence of ischaemic events was low and comparable between patients randomised to beta blockers or control. There were no significant differences in all-cause death, non-fatal reinfarction, or unplanned revascularisation between patients randomised to beta blockers or not, both in the unadjusted model and after adjustment for key variables (Table 2). The composite ischaemic endpoint of cardiac death, reinfarction, sustained VT/VF or resuscitated cardiac arrest, or unplanned revascularisation at 3 months occurred in 46 (rate: 44.8 per 1,000 patient-years) patients randomised to beta blockers versus 41 (39.6 per 1,000 patient-years) in those randomised to no beta blocker (HR 1.13, 95% CI: 0.74-1.72). Landmark analysis at 3 months is shown in the Central illustration. No treatment heterogeneity was identified across prespecified key subgroups (Figure 1).
Stratified analyses according to prior beta blocker use did not identify significant treatment-by-subgroup interactions. In patients with chronic beta blocker use before their index MI, beta blockers at discharge were not associated with significant benefit across individual or composite endpoints (Table 3). Similarly, no treatment effect was observed in those without prior beta blocker use.
Analyses of the primary endpoint based on beta blocker type and dose at 3 months (below or above the median) are shown in Table 4.
Table 2. Three-month rate of ischaemic endpoints.
| N (rate per 1,000 patient-years) | HR (95% CI) | p-value | ||
|---|---|---|---|---|
| Beta blocker | No beta blocker | |||
| Unadjusted | ||||
| All-cause death | 13 (12.6) | 15 (14.4) | 0.87 (0.41-1.83) | 0.72 |
| Cardiac death | 8 (7.7) | 10 (9.6) | 0.80 (0.32-2.04) | 0.65 |
| Non-fatal reinfarction | 22 (21.4) | 21 (20.3) | 1.05 (0.58-1.92) | 0.86 |
| Unplanned revascularisation | 21 (20.4) | 17 (16.4) | 1.24 (0.66-2.36) | 0.50 |
| Admission for stroke | 5 (4.8) | 3 (2.9) | 1.68 (0.40-7.02) | 0.47 |
| Sustained VT/VF or resuscitated cardiac arrest | 2 (1.9) | 0 (0) | - | 0.16 |
| Cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation | 46 (44.8) | 41 (39.6) | 1.13 (0.74-1.72) | 0.57 |
| Adjusted for country, age, sex, type of infarction, and diabetes mellitus | ||||
| All-cause death | 13 (12.6) | 15 (14.4) | 0.89 (0.42-1.87) | 0.76 |
| Cardiac death | 8 (7.7) | 10 (9.6) | 0.81 (0.32-2.05) | 0.65 |
| Non-fatal reinfarction | 22 (21.4) | 21 (20.3) | 1.05 (0.58-1.90) | 0.88 |
| Unplanned revascularisation | 21 (20.4) | 17 (16.4) | 1.23 (0.65-2.34) | 0.52 |
| Admission for stroke | 5 (4.8) | 3 (2.9) | 1.63 (0.39-6.82) | 0.50 |
| Sustained VT/VF or resuscitated cardiac arrest | 2 (1.9) | 0 (0) | - | - |
| Cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation | 46 (44.8) | 41 (39.6) | 1.12 (0.73-1.70) | 0.60 |
| Hazard ratios were estimated using Cox proportional hazard models; the models were used to estimate the effect of taking a beta blocker versus no beta blocker. CI: confidence interval; HR: hazard ratio; VF: ventricular fibrillation; VT: ventricular tachycardia | ||||
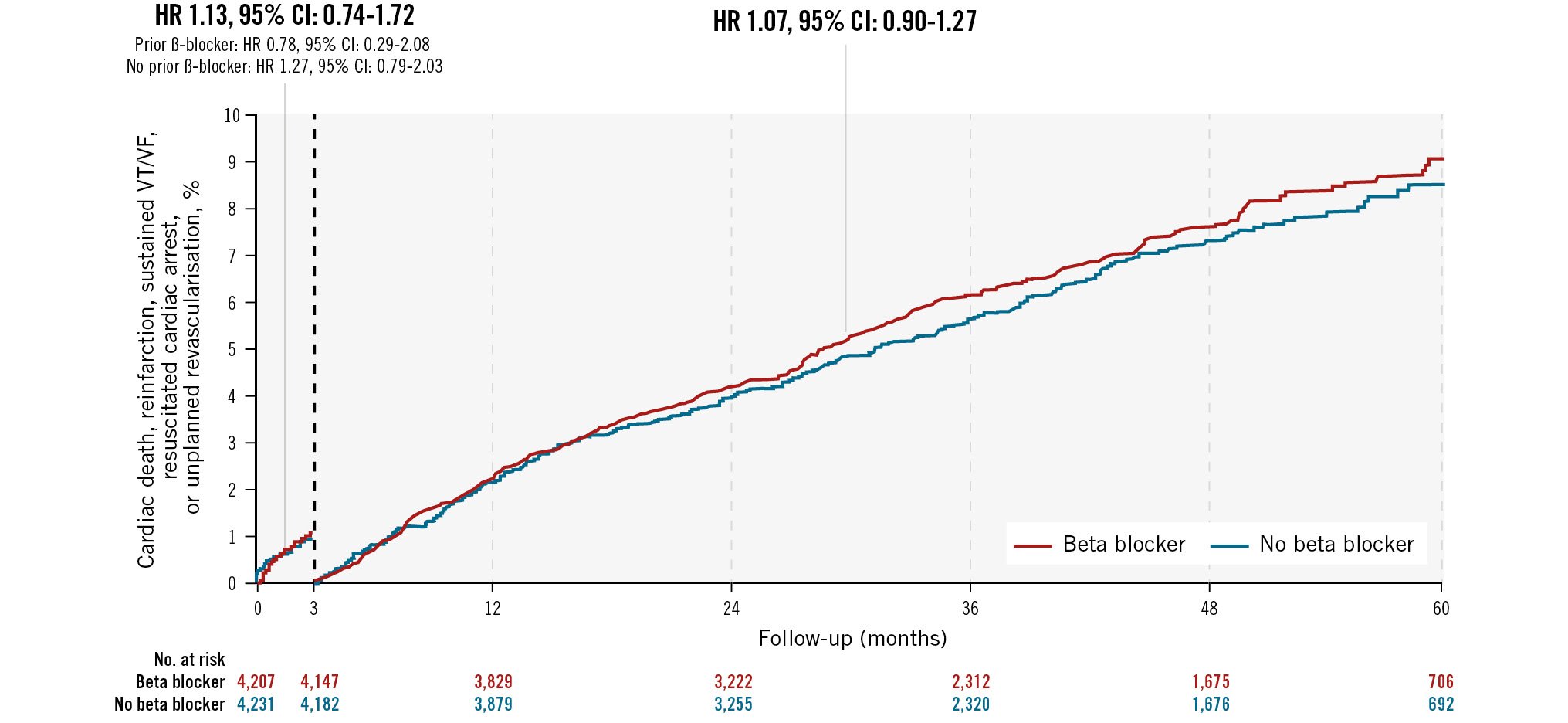
Central illustration. Kaplan-Meier curves for primary endpoint landmark analysis at 3 months Kaplan-Meier plot of the landmark analysis of cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation at 3 months. A hazard ratio (HR) below 1 shows a benefit of beta blocker therapy and above 1 shows a benefit of control. CI: confidence interval; VF: ventricular fibrillation; VT: ventricular tachycardia
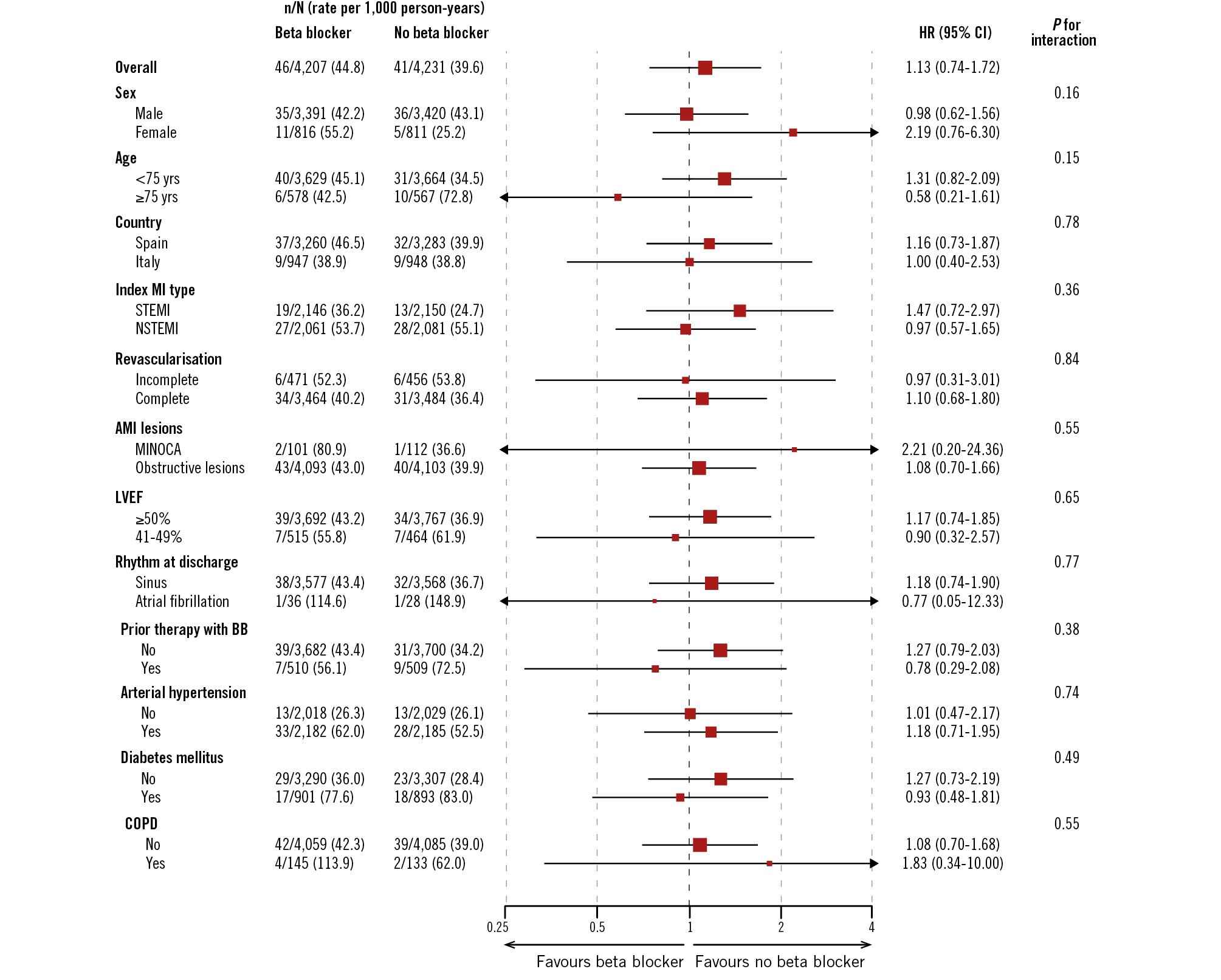
Figure 1. Subgroup analysis of the ischaemic endpoint. Prespecified subgroups of the REBOOT trial. The composite outcome includes cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation. AMI: acute myocardial infarction; BB: beta blocker; CI: confidence interval; COPD: chronic obstructive pulmonary disease; HR: hazard ratio; MI: myocardial infarction; MINOCA: myocardial infarction without non-obstructive coronary arteries; NSTEMI: non-ST-segment elevation myocardial infarction; STEMI: ST-segment elevation myocardial infarction; VF: ventricular fibrillation; VT: ventricular tachycardia
Table 3. Three-month rate of ischaemic endpoints according to patients having received beta blocker therapy before randomisation or not.
| Prior beta blocker | Without prior beta blocker | p-value for interaction | |||||
|---|---|---|---|---|---|---|---|
| N (rate per 1,000 patient-years) | HR (95% CI) | N (rate per 1,000 patient-years) | HR (95% CI) | ||||
| Beta blocker | No beta blocker | Beta blocker | No beta blocker | ||||
| All-cause death | 3 (23.9) | 2 (16.0) | 1.50 (0.25-8.96) | 10 (11.1) | 13 (14.3) | 0.77 (0.34-1.76) | 0.51 |
| Cardiac death | 1 (8.0) | 1 (8.0) | 1.00 (0.06-15.96) | 7 (7.7) | 9 (9.9) | 0.78 (0.29-2.10) | 0.87 |
| Non-fatal reinfarction | 3 (24.0) | 5 (40.1) | 0.60 (0.14-2.50) | 19 (21.1) | 16 (17.7) | 1.19 (0.61-2.32) | 0.38 |
| Unplanned revascularisation | 5 (40.1) | 4 (32.1) | 1.25 (0.34-4.66) | 16 (17.7) | 12 (13.2) | 1.34 (0.63-2.83) | 0.93 |
| Admission for stroke | 0 (0) | 1 (8.0) | - | 5 (5.5) | 2 (2.2) | 2.51 (0.49-12.95) | - |
| Sustained VT/VF or resuscitated cardiac arrest | 0 (0) | 0 (0) | - | 2 (2.2) | 0 (0) | - | - |
| Cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation | 7 (56.1) | 9 (72.5) | 0.78 (0.29-2.08) | 39 (43.4) | 31 (34.2) | 1.27 (0.79-2.03) | 0.38 |
| Hazard ratios were estimated using Cox proportional hazard models with an interaction between prior beta blocker use and treatment arm; the models were used to estimate the effect of taking a beta blocker versus no beta blocker. CI: confidence interval; HR: hazard ratio; VF: ventricular fibrillation; VT: ventricular tachycardia | |||||||
Table 4. Three-month rate of ischaemic endpoints by beta blocker type and dose.
| No. of patients | No. of events (rate per 1,000 person-years) |
HR (95% CI) | p-value | |
|---|---|---|---|---|
| Beta blocker type | ||||
| No beta blocker | 4,231 | 41 (39.6) | Reference group | 0.82 |
| Bisoprolol | 3,549 | 40 (46.1) | 1.16 (0.75-1.80) | |
| Metoprolol | 309 | 2 (26.5) | 0.67 (0.16-2.76) | |
| Other beta blocker | 273 | 3 (44.8) | 1.13 (0.35-3.65) | |
| Beta blocker dose | ||||
| No beta blocker | 4,231 | 41 (39.6) | Reference group | 0.81 |
| Beta blocker dose ≤median | 3,540 | 38 (43.9) | 1.11 (0.71-1.72) | |
| Beta blocker dose >median | 573 | 7 (50.1) | 1.26 (0.57-2.81) | |
| Hazard ratios were estimated using a Cox proportional hazard model; the models were used to estimate the effect of taking each type or dose of beta blocker versus no beta blocker. Median doses: atenolol 50 mg, bisoprolol 2.5 mg, carvedilol 12.5 mg, metoprolol 50 mg, nebivolol 2.5 mg, other 40 mg. CI: confidence interval; HR: hazard ratio | ||||
Effect of beta blocker therapy on repetitive ischaemic events across the trial follow-up
During a median follow-up of 3.7 years, 291 and 282 patients in the beta blocker and control groups, respectively, developed the composite ischaemic endpoint of cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation. The total number of events including repetitive episodes were 319 (22.2/1,000 patient-years) and 330 (22.8/1,000 patient-years) in patients randomised to beta blocker or control, respectively (HR 0.98, 95% CI: 0.82-1.16; p=0.78).
The number of total recurrent ischaemic events (composite outcome) according to the type of MI was 149 (20.3/1,000 patient-years) versus 133 (17.9/1,000 patient-years) in STEMI patients (HR 1.13, 95% CI: 0.87-1.47) and 170 (24.2/1,000 patient-years) versus 197 (27.8/1,000 patient-years) in NSTEMI patients (HR 0.87, 95% CI: 0.69-1.09). The p-value for interaction was 0.13.
Recurrent events (the composite endpoint and its individual components) in patients with or without previous beta blocker treatment are shown in Table 5. Relative to those not on beta blockers before the index MI, patients on previous beta blocker therapy had more ischaemic events. However, there was no heterogeneity in treatment effect in any of the ischaemic events between those with and without beta blockers before the index MI – i.e., no significant interactions were found between prior treatment with beta blockers and randomisation to beta blockers for any of the ischaemic endpoints (Table 5).
As shown in Figure 2, most patients had only one ischaemic event during follow-up. Beta blocker treatment had no impact on the prevention of ischaemic events in patients developing only one event. Conversely, there were numerically fewer patients developing recurrent ischaemic events (≥2) in the beta blocker group than in the control group. The small number of recurrent events limits the power of this analysis.
Figure 3 shows the cumulative incidence of recurrent composite ischaemic endpoint events.
Table 5. Recurrent ischaemic events across the trial according to patients having received beta blocker therapy before randomisation or not.
| Prior beta blocker | Without prior beta blocker | p-value for interaction | |||||
|---|---|---|---|---|---|---|---|
| N (rate per 1,000 patient-years) | HR (95% CI) | N (rate per 1,000 patient-years) | HR (95% CI) | ||||
| Beta blocker | No beta blocker | Beta blocker | No beta blocker | ||||
| Reinfarction | 40 (22.0) | 38 (20.9) | 1.05 (0.65-1.72) | 110 (8.8) | 121 (9.6) | 0.92 (0.70-1.21) | 0.63 |
| Unplanned revascularisation | 41 (22.5) | 37 (20.3) | 1.11 (0.70-1.77) | 145 (11.6) | 143 (11.3) | 1.02 (0.80-1.31) | 0.76 |
| Admission for stroke | 6 (3.3) | 4 (2.2) | 1.50 (0.36-6.32) | 31 (2.5) | 21 (1.7) | 1.49 (0.86-2.59) | 0.99 |
| Sustained VT/VF or resuscitated cardiac arrest | 1 (0.5) | 0 (0.0) | -- | 9 (0.7) | 11 (0.9) | 0.83 (0.33-2.06) | -- |
| Cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation | 65 (35.7) | 70 (38.4) | 0.93 (0.64-1.34) | 254 (20.3) | 256 (20.3) | 1.00 (0.83-1.21) | 0.73 |
| Hazard ratios were estimated using Lin Wei Yang Ying models for recurrent events with an interaction between prior beta blocker use and treatment arm; the models were used to estimate the effect of taking a beta blocker versus no beta blocker. CI: confidence interval; HR: hazard ratio; VF: ventricular fibrillation; VT: ventricular tachycardia | |||||||
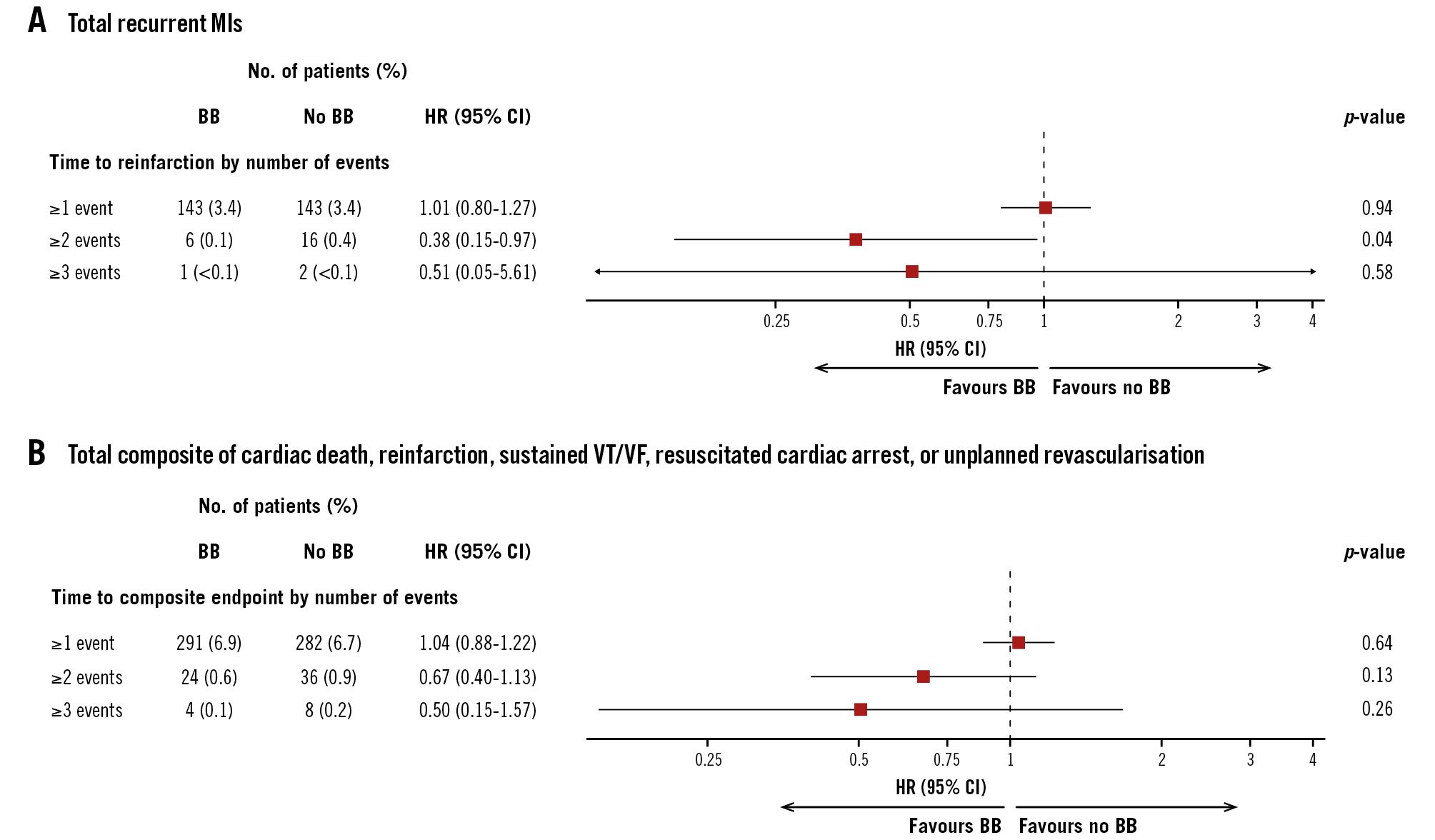
Figure 2. The effect of beta blockers according to the number of recurrent repetitive ischaemic events. Forest plots for patients developing 1 event during follow-up, 2 or more events, and 3 or more events. A hazard ratio (HR) below 1 shows a benefit of beta blocker therapy and above 1 shows a benefit of control. Forest plots are for the reinfarction endpoint during follow-up (A) and for the composite ischaemic endpoint of cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation (B). BB: beta blocker; CI: confidence interval; MI: myocardial infarction; VF: ventricular fibrillation; VT: ventricular tachycardia
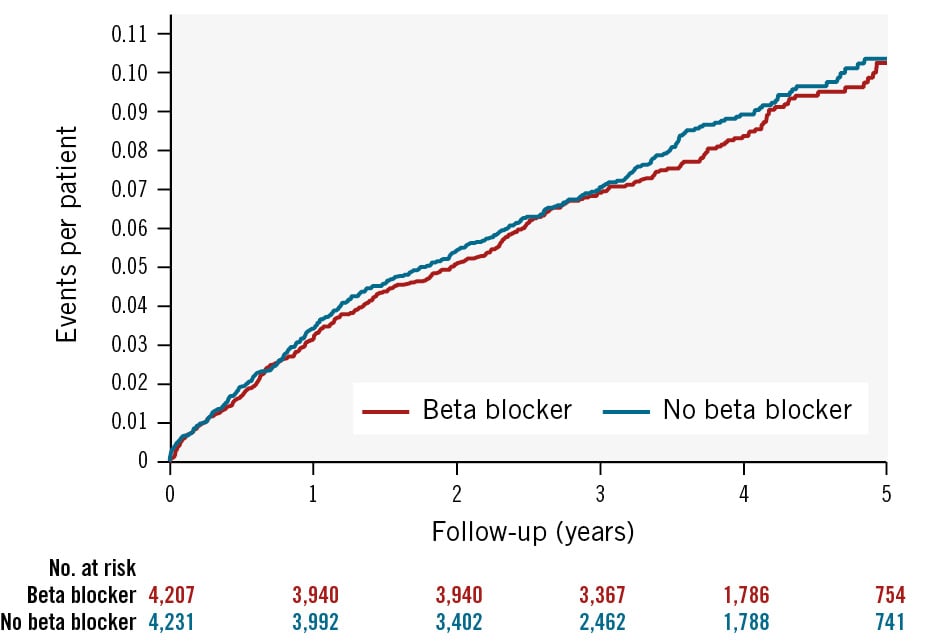
Figure 3. Cumulative incidence of the recurrent ischaemic endpoint. Kaplan-Meier plot of the cumulative incidence of the ischaemic endpoint (composite of cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned revascularisation) across the study follow-up. VF: ventricular fibrillation; VT: ventricular tachycardia
Discussion
In this post hoc analysis of the REBOOT trial of patients with MI without reduced LVEF randomised to beta blocker or no beta blocker therapy, we assessed the effect of the intervention on short-term ischaemic events and recurrent ischaemic episodes throughout follow-up in the overall population as well as in a population stratified according to whether they had been on chronic beta blocker therapy before their index hospitalisation. The main results are as follows: (1) patients on beta blocker therapy before index admission were older, more frequently female, had a higher burden of cardiovascular comorbidities, and presented more frequently with NSTEMI than those not on beta blockers previously. This worse cardiovascular profile translated into a high rate of ischaemic events throughout the trial. 2) Withholding beta blockers upon discharge from an uncomplicated MI without reduced LVEF (i.e., no prescription at discharge) or withdrawing them (i.e., abrupt discontinuation at discharge in those taking them before the index episode) was not associated with any increase in short-term (3-month) ischaemic endpoints (Central illustration).
Current 2025 ACC/AHA/ACEP/NAEMSP/SCAI ACS guidelines continue to recommend the use of beta blockers in all patients following MI, irrespective of LVEF, with the stated goal of reducing the risks of reinfarction and ventricular arrhythmias (Class I recommendation)2. However, the recent REDUCE-AMI trial, which enrolled 5,050 patients with preserved LVEF (≥50%), failed to demonstrate a reduction in the composite outcome of death or recurrent MI with beta blocker therapy5. Similarly, the REBOOT trial − the largest randomised trial to date in this setting (n=8,505) − reported no benefit of beta blocker therapy in patients with LVEF >40%, including no reduction in MI incidence4. In both trials, the lack of benefit extended to the individual endpoint of reinfarction. In the present analysis, we extended this investigation by evaluating a broader ischaemic composite outcome – including cardiac death, reinfarction, sustained VT/VF, resuscitated cardiac arrest, or unplanned coronary revascularisation – to capture all relevant ischaemic events. We show that beta blocker therapy has no impact on this broader ischaemic endpoint. This contrasts with findings from historical trials and meta-analyses conducted prior to the reperfusion era, which reported substantial reductions in MI with beta blocker therapy10111213. However, those studies were conducted before the routine use of timely reperfusion, complete revascularisation, potent DAPT, and high-intensity statins; they did not exclude patients with heart failure; and LVEF was not reported, suggesting that some benefit of beta blockers might have been derived from subgroups with heart failure. Notably, the incidences of reinfarction in REBOOT and REDUCE-AMI were markedly lower than in these older studies. For example, the 1-year reinfarction rate was slightly above 1% in REBOOT, compared with rates ranging from 5% to 10% within the first year in historical trials1214. Similarly, the rate of unplanned revascularisation in REBOOT was approximately 1.2% per year, a stark contrast to the 5% reported in older registries and trials15.
The inclusion of sustained VT/VF and resuscitated cardiac arrest in the broader composite endpoint is relevant, as one of the mechanisms associating beta blockers with reduced post-MI mortality was their antiarrhythmic abilities. Yet, the rate of malignant ventricular arrhythmias has also dramatically declined. In REBOOT, sustained VT/VF or cardiac arrest occurred in only 0.25% of patients. In contrast, older trials such as BHAT and ISIS-1 reported rates exceeding 3% within the first weeks post-MI1316. This substantial shift likely reflects improved myocardial salvage, shorter ischaemic times, and widespread use of evidence-based therapies. Mortality in contemporary trials is also markedly lower (e.g., 3.7% all-cause death in REBOOT over 3.7 years), indicating a profound change in the natural history of MI. Mortality in historical beta blocker trials was 10-15%1214.
The traditional antiarrhythmic and anti-ischaemic properties of beta blockers were highly effective in a setting of high arrhythmogenic burden, residual coronary artery disease, and extensive myocardial necrosis. However, this is no longer representative of the clinical profile of most patients with MI without reduced LVEF managed with modern therapies. The current data, therefore, question the universal role of beta blockers in this population.
Concerns regarding a possible rebound phenomenon upon abrupt beta blocker withdrawal have historically influenced clinical practice. Early studies in the 1970s and 1980s reported increased incidences of angina, MI, and arrhythmias following sudden discontinuation, leading to warnings in the FDA label617. In REBOOT, a short-term clinical follow-up visit was purposely scheduled at 3 months to identify any early hazard, especially in patients previously on chronic beta blocker therapy. This strategy was reviewed by the independent Data and Safety Monitoring Board, which monitored early event rates. As shown in the current analysis, there was no increase in ischaemic events at 3 months among patients who had beta blockers withdrawn at discharge: in those on beta blocker therapy before index MI, the composite ischaemic endpoint occurred in 56.1 and 72.5 per 1,000 person-years in those randomised to beta blocker and control, respectively (HR 0.7, 95% CI: 0.29-2.08). These findings suggest that abrupt beta blocker discontinuation is safe in the contemporary post-MI setting.
Patients receiving beta blockers before randomisation most likely did so for clinical reasons. In fact, in our study, those with prior beta blocker therapy had a higher prevalence of hypertension and atrial fibrillation before admission. Discontinuing treatment in such cases may have removed ancillary benefits, such as blood pressure reduction or rate control. In this regard, the Assessment of βeta Blocker Interruption After Uncomplicated mYocardial Infarction on Safety and Symptomatic Cardiac Events Requiring Hospitalization (AβYSS) trial, which randomised patients to beta blocker withdrawal or continuation ~3 years after the index MI18, reported that patients who were randomised to withdrawal had significant increases in blood pressure and heart rate19. Whether a similar effect occurred in the REBOOT trial is unknown because of the absence of data on prior treatments other than beta blockers, as well as the lack of post-discharge blood pressure and heart rate monitoring. In REBOOT, patients who had a prior treatment with beta blockers were more frequently discharged on ACE inhibitors/angiotensin II receptor blockers and diuretics, possibly to compensate for the loss of the blood pressure-lowering effect of beta blockers.
The presumed rebound effect in older studies was likely due to unrecognised severe residual coronary disease. In REBOOT, 82% of patients who were on chronic beta blocker therapy before their index MI underwent complete revascularisation, minimising this potential mechanism of harm. Furthermore, the subgroup of patients with prior hypertension – historically considered at increased risk for rebound events – was overrepresented among those with chronic beta blocker use prior to MI (72.8% vs 49.0%), yet no excess risk was observed after therapy discontinuation.
An intriguing observation of our study is the higher proportion of NSTEMI presentations among patients who were on chronic beta blocker therapy before the index MI (62.4% vs 47.2%). While speculative, these findings may reflect a biological effect of beta blockers in modifying thrombus propagation, potentially attenuating the severity of ischaemic occlusion. Beta blockers have been shown to interfere with platelet-neutrophil aggregates, which play a role in coronary thrombosis and microvascular obstruction2021. Moreover, previous data support a protective effect of early intravenous beta blocker administration on reperfusion injury and infarct size, as demonstrated in the METOCARD-CNIC trial2223. Although infarct size was not assessed in REBOOT, the increased rate of NSTEMI among chronically treated patients could hypothetically reflect smaller infarcts or less transmural involvement, warranting further mechanistic investigation.
Limitations
First, the ample composite ischaemic endpoint was not defined in the protocol. We decided to use this endpoint to capture any possible effect across the entire spectrum of ischaemic presentations. Second, although the 3-month time period was set in the protocol to rule out any harmful effect in the most vulnerable period after ACS, the present short-term analysis was planned after closing the statistical analysis plan. While there does not appear to be a meaningful difference in treatment effect between patients with and without prior beta blocker therapy at 3 months, even if such a difference was present, this analysis would unlikely be able to detect it because of the reduced number of events at this timepoint. Third, our findings were based on the original randomisation (e.g., whether patients were on beta blockers at baseline), disregarding whether patients remained on the same treatment over follow-up. However, the crossover rate at 3 months was low, as reported in the primary publication of the trial4: 5% and 9% in patients randomised to beta blocker and no beta blocker therapy, respectively. Fourth, while treatment or no treatment with beta blockers before the index MI was a prespecified subgroup analysis, this was defined for the primary outcome of the trial (death, MI, or heart failure). Fifth, the absence of placebo in the control group represents a limitation, particularly in this post hoc analysis. Without blinding, treatment allocation may have influenced patient or physician behaviour, potentially affecting outcomes. While randomisation in the main trial mitigates some bias, the non-prespecified nature of the present analyses reduces the protection afforded by randomisation, making residual confounding and expectation effects more likely and warranting cautious interpretation of these findings. Finally, no adjustment for multiplicity was performed24. However, this might have little impact on our main conclusions given that the alternative hypothesis has been largely rejected in most of our comparisons.
Conclusions
In this post hoc analysis of the REBOOT trial, withholding or withdrawing beta blockers at discharge in patients following MI with an LVEF >40% was not associated with an increased incidence of short-term ischaemic events. Despite a higher baseline cardiovascular risk and greater ischaemic event burden among patients previously on chronic beta blocker therapy, discontinuation of treatment did not result in early harm, supporting the safety of beta blocker withdrawal in this contemporary post-MI population.
Impact on daily practice
In patients with myocardial infarction (MI) and left ventricular ejection fraction (LVEF) >40%, beta blocker withholding or withdrawal at discharge was not associated with increased short-term or recurrent ischaemic events. Not prescribing beta blockers after an uncomplicated MI is not associated with any acute ischaemic safety concern. Withdrawal of beta blockers after MI in patients who were on these agents chronically is not associated with any ischaemic rebound effect. These findings in post-MI patients without heart failure and with LVEF >40% may challenge the current European Society of Cardiology recommendation of using beta blockers to reduce the risk of reinfarction and ventricular arrhythmias.
Conflict of interest statement
The authors have no conflicts of interest to declare related to this work.

