Abstract
Background: Despite the fact that morbidity and mortality rates significantly increase with tricuspid regurgitation (TR) severity, limited treatment options are available for treating severe TR.
Aims: The single-arm, multicentre, prospective Confirmatory Clinical Study of Treating Tricuspid Regurgitation With K-Clip TM Transcatheter Annuloplasty System (TriStar) evaluated the 1-year outcomes of the novel transcatheter K-Clip annuloplasty system in treating secondary TR.
Methods: Between May 2022 and October 2022, patients with ≥severe secondary TR despite optimal medical therapy at 11 centres in China were deemed candidates for transcatheter tricuspid repair by the local Heart Team and a multidisciplinary screening committee. Echocardiographic parameters, clinical and quality-of-life measures, and major adverse events were collected at 1 year.
Results: Ninety-six patients were enrolled (mean age 72.6±7.0 years, 60.4% female, mean TRI-SCORE 5.4±2.1). The technical success rate was 97.9%. At 1 year, echocardiographic follow-up showed an average reduction in the annular septolateral diameter of 11.3% (41.9 mm vs 37.1 mm; p<0.01), compared with baseline, with marked right ventricular remodelling. A total of 82.5% of patients had ≤moderate TR, and 97.7% had a ≥1 grade reduction. Patients experienced significant clinical improvements in New York Heart Association Functional Class I/II (32.6% to 96.5%; p<0.001), the 6-minute walk distance increased by 31.9±71.8 m (p<0.001), and the overall Kansas City Cardiomyopathy Questionnaire score increased by 7.6±17.7 points (p<0.001). Neither cardiovascular death nor reintervention were recorded at the 30-day or 1-year follow-up, while severe bleeding requiring further treatment was noted in 5 patients at 1 year. The Kaplan-Meier estimates of survival and freedom from heart failure rehospitalisation were 97.8% and 95.1%, respectively, at 1 year.
Conclusions: The 1-year experience using the K-Clip tricuspid annuloplasty system demonstrated high survival and low rehospitalisation rates with durable TR reduction and clinical benefits in functional status and quality-of-life outcomes.
Although long considered benign, severe tricuspid regurgitation (TR) is nowadays being increasingly recognised as a significant public health threat associated with substantial morbidity, reduced functional capacity, and poor clinical prognosis if left untreated1. Of note, approximately 80% of TR is secondary TR (STR)2, in which tricuspid annular dilation caused by atrial fibrillation and increased pulmonary and right ventricular pressures is the dominant aetiology3. Since medical management outcomes are unsatisfactory, surgery is currently the only definitive treatment option for TR4. However, corrective surgery is rarely offered for patients with isolated severe TR due to the high operative risk and in-hospital mortality567. Hence, the identification of candidates in need of an alternative TR treatment has spawned rapid growth in transcatheter tricuspid valve (TV) interventions89.
The K-Clip system (Huihe Healthcare Technology Co., Ltd.) is a novel transcatheter tricuspid repair device that uses a clip-based annuloplasty approach. Our previous study on the compassionate use of the K-Clip system in 15 patients with severe TR indicated significant TR reduction with high procedural success10. The Confirmatory Clinical Study of Treating Tricuspid Regurgitation With K-Clip TM Transcatheter Annuloplasty System (TriStar) is the first multicentre study using the K-Clip tricuspid annuloplasty system to treat severe secondary TR patients that are symptomatic despite optimal medical therapy. Here, we report the study’s 1-year outcomes.
Methods
Study design and patient selection
This prospective, single-arm, multicentre study − TriStar − was registered at ClinicalTrials.gov: NCT05173233. The aim of this study was to evaluate the safety and efficacy of the K-Clip tricuspid annuloplasty system in treating patients with symptomatic severe STR. Between May 2022 and October 2022, 96 patients with ≥severe secondary TR were enrolled at 11 sites in China. The key exclusion criteria included improper anatomy for device implantation, history of TV surgery/intervention or pacemaker lead implantation, severe pulmonary hypertension (i.e., pulmonary artery systolic pressure >55 mmHg), severe left ventricular dysfunction (i.e., left ventricular ejection fraction [LVEF] <40%), indication for left-sided valve surgery, kidney dysfunction with an estimated glomerular filtration rate (eGFR) ≤25 mL/min/1.73 m², and chronic dialysis. The study protocol was approved by the institutional review board at each site, and written informed consent was obtained from all enrolled participants.
Device and procedure
The K-Clip is a percutaneous tricuspid annuloplasty system that uses a clip-based annular plication approach (Supplementary Figure 1)11. The K-Clip system is delivered exclusively through an 18 Fr adjustable delivery sheath. It is designed to deploy in the anterior and posterior regions of the tricuspid annulus in accordance with the derivation of individual regurgitant jets. Four clip arm diameters ranging from 12 mm to 18 mm (in 2 mm increments) are available. Its unique 3-point fixation technique confers rigidity and stabilisation to maintain tricuspid annular reconstruction (Central illustration).
Routine transthoracic echocardiography (TTE) and transoesophageal echocardiography (TOE) were conducted for anatomical evaluation. Preprocedural planning for K-Clip implantation was scheduled using gated cardiac computed tomography (CT). A cardiac CT scan was performed to determine the size of the tricuspid annulus and for procedural planning (mitigating the risk of injury to the right coronary artery [RCA] and facilitating the generation of planning views). The CT-derived tricuspid annulus circumference at the phase closest to the latest diastole was measured as the reference for selecting the device size.
The details of the K-Clip annuloplasty system used for implantation have been described elsewhere1213 (Figure 1A-Figure 1B-Figure 1C-Figure 1D). In brief, the procedure was performed under general anaesthesia, and surveillance was performed using TOE and digital subtraction angiography. The K-Clip system was delivered via the right transjugular access with a guidewire placed in the RCA to help identify the location. The tapping screw-shaped anchor was placed in the optimal position within the tricuspid annulus, close to the leaflet hinge point on the right atrial side. The clip arm was gradually closed, allowing the surrounding annular tissue to be clutched within the clip arms in a stepwise manner. Before the full deployment and release of the K-Clip system, the TV function and clutch status were assessed by echocardiography to ensure an optimal device position and to confirm annulus capture. RCA angiography was repeated to exclude coronary artery compression. Intravenous heparin was administered during implantation of the K-Clip to maintain an activated clotting time of 300-600 s.
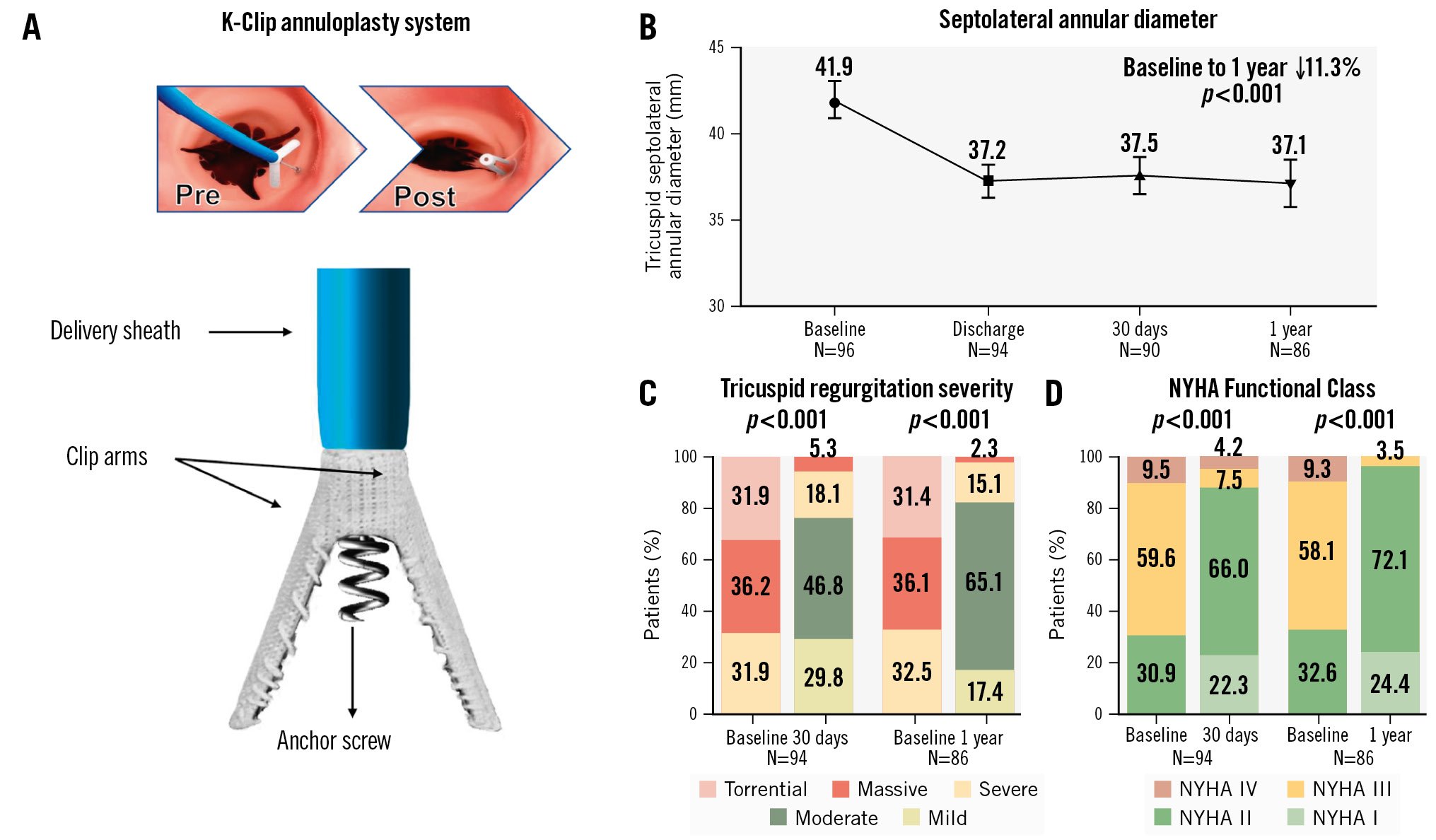
Central illustration. One-year outcomes of patients treated with the transcatheter K-Clip annuloplasty system for severe secondary tricuspid regurgitation. A) The K-Clip annuloplasty system contains two clip arms and one anchor screw to form a “clamp-annulus-screw sandwich structure” for tricuspid valve annuloplasty. B) End-diastolic septolateral diameters at baseline, discharge, 30 days and 1 year. Values are mean, with 95% confidence intervals. C) Paired analysis of core laboratory-adjudicated TR severity at baseline and during 30-day and 1-year follow-ups. D) Paired analysis of NYHA Functional Class at baseline and during 30-day and 1-year follow-ups. NYHA: New York Heart Association; TR: tricuspid regurgitation
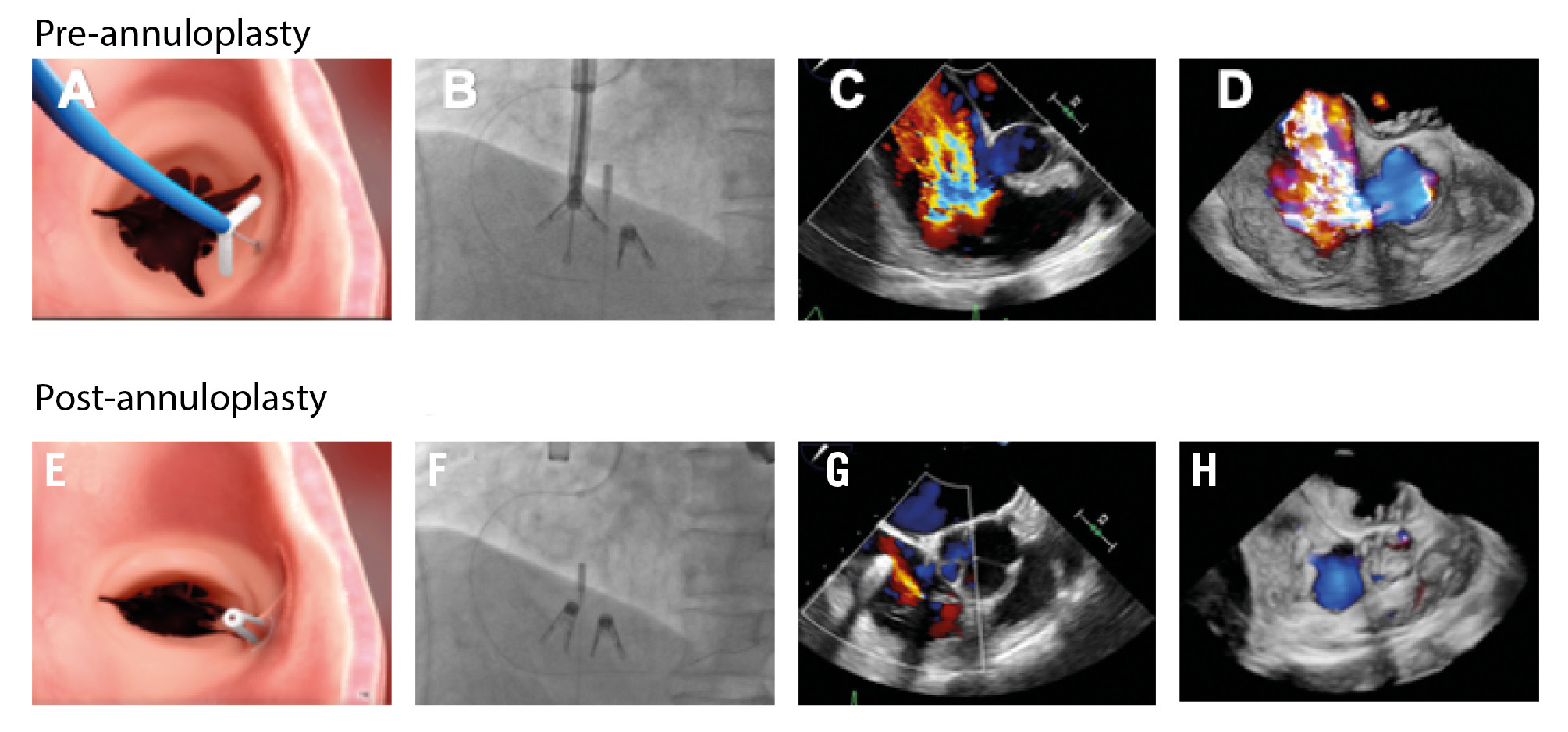
Figure 1. Pre- and post-annuloplasty images of the K-Clip tricuspid system. A,E) Tricuspid annulus capture, (B,F) fluoroscopy, (C,G) 2-dimensional TOE, and (D,H) 3-dimensional TOE en face. TOE: transoesophageal echocardiography
Trial endpoints
The safety endpoint was a composite of major adverse events (MAE) at 30 days, comprising cardiovascular mortality, non-elective TV reintervention, severe bleeding, major access site and vascular complications, major cardiac structural injury, conduction disturbances requiring permanent pacing, stroke, pulmonary embolism, deep vein thrombosis and acute kidney injury. An event adjudication committee comprising independent non-investigator physicians assessed adverse clinical events according to the updated Tricuspid Valve Academic Research Consortium (TVARC) definitions2.
The performance endpoints were technical, procedural and clinical success. Technical success was assessed on exit from the catheterisation laboratory. Specifically, it was a combination of (1) successful delivery and retrieval of the system, (2) correct implantation of at least one device deployed as intended, and (3) freedom from mortality and emergency surgery. Provided technical success was achieved, procedural success was assessed before discharge and determined as proper positioning of the device with at least one grade of TR reduction and without the need for surgical or percutaneous reintervention. Clinical success was defined as procedural success without MAE and was evaluated at the 30-day and 1-year follow-ups.
Functional status, exercise capacity, and improvement in health status were assessed for each patient at baseline, 30 days after the operation, and yearly thereafter by determining the New York Heart Association (NYHA) Functional Class, 6-minute walk distance (6MWD), and Kansas City Cardiomyopathy Questionnaire (KCCQ) scores, respectively. All-cause mortality, rehospitalisation for heart failure and non-elective TV reintervention were recorded, and the rates were analysed for the study period. Changes in diuretic requirements and biochemical outcomes (aspartate aminotransferase [AST], alanine transaminase [ALT], albumin, eGFR, and N-terminal pro-brain natriuretic peptide [NT-proBNP]) were also compared between baseline and follow-up.
Echocardiography core laboratory analysis
Echocardiographic data obtained at baseline and at the 30-day and 1-year follow-ups were analysed by an independent echocardiographic core laboratory following the American Society of Echocardiography standards14. Chamber size and cardiac function were evaluated using standard two-dimensional colour Doppler methods, whereas the TR effective regurgitant orifice area (EROA) and regurgitant volume were quantified using the proximal isovelocity surface area method. Individual TR severity was graded according to a prespecified 5-class scheme, as described previously15.
Medical therapy
Medical management was performed at the discretion of the investigator. The study guidelines recommended keeping patients on a preprocedural diuretic regimen for at least 3 months after implantation. For those successfully undergoing K-Clip implantation, patients were discharged with short-term dual antiplatelet therapy for a minimum of 1 month, while continuous oral anticoagulants were administered to patients with pre-existing atrial fibrillation or to those who were on anticoagulants before the intervention.
Statistical analysis
Continuous data are presented as the mean±standard deviation. Categorical variables are presented as n (%). The paired Student’s t-test was used to compare continuous variables, whereas categorical variables were compared using the Wilcoxon signed-rank test, between baseline and each follow-up timepoint. All data analyses were conducted using SPSS software 26.0 (IBM). Statistical significance was set at a 2-sided p-value<0.05.
Results
Baseline characteristics
A total of 112 patients were screened for the study, and 16 patients were ultimately excluded for the reasons shown in Supplementary Figure 2. The baseline demographics of our study population are listed in Table 1. The mean age of the 96 enrolled patients was 72.6±7.0 years, and 60.4% were women. Most patients (69.8%) were in NYHA Functional Classes III to IV, and the mean TRI-SCORE was calculated as 5.4±2.1. The aetiology of TR was secondary in all patients: 58 (60.4%) had ventricular STR, and 38 (39.6%) had atrial STR. Common comorbidities included hypertension (49.0%), atrial fibrillation/atrial flutter (AF/Af; 88.5%) and diabetes (14.6%). A total of 27 patients (28.1%) had a history of previous mitral and/or aortic valve surgery/intervention. All patients presented with ≥severe TR (massive in 35.4%, torrential in 31.3%), and the mean tricuspid annular perimeter and area were enlarged at 15.0±4.2 cm and 11.5±1.9 cm², respectively. The mean tethering distance of our study population was 5.6±2.7 mm.
Table 1. Baseline demographic and clinical characteristics of the study population.
| Variables | N=96 |
|---|---|
| Age, yrs | 72.6±7.0 |
| Female | 58 (60.4) |
| EuroSCORE II, % | 9.9±6.1 |
| TRI-SCORE, % | 5.4±2.1 |
| NYHA Functional Class ≥III | 67 (69.8) |
| Hypertension | 47 (49.0) |
| Diabetes mellitus | 14 (14.6) |
| AF/Af | 85 (88.5) |
| Stroke/TIA | 16 (16.7) |
| Peripheral vascular disease | 11 (11.5) |
| Chronic kidney disease | 11 (11.5) |
| Previous valvular surgery/intervention | |
| Aortic valve | 12 (12.5) |
| Mitral valve | 4 (4.2) |
| Aortic+mitral valve | 11 (11.5) |
| Tricuspid valve | 0 (0) |
| Laboratory examinations | |
| eGFR, mL/min/1.73 m² | 70.2±20.4 |
| ALT, U/L | 21.7±21.4 |
| AST, U/L | 27.9±14.7 |
| Γ-GGT, U/L | 71.5±68.1 |
| Bilirudin, μmol/L | 17.4±7.8 |
| ALP, U/L | 94.8±40.7 |
| Albumin, g/L | 40.8±5.0 |
| NT-proBNP, pg/mL | 1,089.5±824.7 |
| Aetiology | |
| Ventricular | 58 (60.4) |
| Atrial | 38 (39.6) |
| Tricuspid valve morphology | |
| I | 68 (70.8) |
| IIB | 2 (2.1) |
| IIIA | 3 (3.1) |
| IIIB | 22 (22.9) |
| IIIC | 1 (1.0) |
| Baseline medication | |
| Digoxin | 20 (20.8) |
| ACE inhibitor or angiotensin II blocker | 37 (38.5) |
| Beta blocker | 39 (40.6) |
| Calcium antagonist | 26 (27.1) |
| Diuretics agent | 96 (100) |
| Data are presented as mean±standard deviation or n (%). ACE: angiotensin-converting enzyme; AF/Af: atrial fibrillation/atrial flutter; ALP: alkaline phosphatase; ALT: alanine transaminase; AST: aspartate transaminase; eGFR: estimated glomerular filtration rate; EuroSCORE: European System for Cardiac Operative Risk Evaluation; GGT: gamma-glutamyl transferase; NT-proBNP: N-terminal pro-brain natriuretic peptide; NYHA: New York Heart Association; TIA: transient ischaemic attack | |
Procedural results
The procedural details of the study population are summarised in Table 2. Technical success was achieved in 94 of 96 patients. One patient experienced cardiac tamponade during steering of the delivery catheter inside the right atrium and was transferred for surgical repair. Right coronary perforation with mild to moderate pericardial effusion was reported in another patient after repeated attempts at screw anchoring; hence, device implantation was not performed thereafter. The mean procedure time was 157.0±61.2 min, and the duration from the insertion of the first implant to the release of the final implant was 73.7±34.4 min. The mean length of hospital stay was 5.2 days after implantation. Procedural success was assessed as 93.8% (90/96) without mortality, stroke, or new permanent pacemaker implantation.
Table 2. Procedural characteristics.
| Variables | N=96 |
|---|---|
| Technical success | 94 (97.9) |
| Procedural success | 90 (93.8) |
| Procedural duration, min | 157.0±61.2 |
| Device time (implant insertion to removal), min | 73.7±34.4 |
| Contrast volume, mL | 33.4±7.8 |
| Number of implants per patient | |
| 1 | 54 (56.3) |
| 2 | 38 (39.6) |
| 3 | 2 (2.1) |
| Device location | N=136 |
| A-S commissure | 0 (0) |
| A-P commisure | 43 (31.6) |
| P-S commissure | 68 (50.0) |
| Midpoint of A | 4 (2.9) |
| Midpoint of P | 21 (15.5) |
| Length of hospital stay, days | 5.2±3.9 |
| Data are presented as mean±standard deviation or n (%). A: anterior leaflet; P: posterior leaflet; S: septal leaflet | |
Clinical outcomes
The incidence of MAE at different follow-up timepoints is summarised in Table 3. The composite MAE rate was 6.3% at 30 days and 13.2% at 1 year. At 1 year, there were 5 deaths in implanted patients, all of which were adjudicated as unrelated to the study procedure by the clinical events committee (one due to septic shock, one due to severe pneumonia with major gastrorrhagia, one due to cerebral haemorrhage followed by multiple organ failure and two due to fatal gastrointestinal haemorrhage). There were no incidences of cardiovascular mortality, need for TV reintervention, stroke, embolism or renal complication requiring unplanned dialysis throughout the study period. Requirement of a new permanent pacemaker implantation was recorded in 3 patients due to AF/Af and sick sinus syndrome, all of which occurred after 30 days post-implantation. Severe bleeding events were reported in another patient with extensive retroperitoneal haematoma before discharge who needed a blood transfusion. Overall, the Kaplan-Meier estimates for survival and freedom from heart failure hospitalisation were 97.8% and 95.1%, respectively (Figure 2A-Figure 2B).
Table 3. Major adverse events.
| Complications | At 30 days (n=96) | At 1 year (n=96) |
|---|---|---|
| All-cause mortality | 0 (0) | 5 (5.2) |
| Cardiovascular mortality | 0 (0) | 0 (0) |
| Non-elective tricuspid valve reintervention | 0 (0) | 0 (0) |
| Severe bleeding | 1 (1.0) | 5 (5.2) |
| Major | 0 (0) | 1 (1.0) |
| Extensive | 1 (1.0) | 1 (1.0) |
| Life-threatening | 0 (0) | 1 (1.0) |
| Fatal | 0 (0) | 2 (2.1) |
| Major access site and vascular complications | 1 (1.0) | 1 (1.0) |
| Massive pericardial effusion or tamponade | 1 (1.0) | 1 (1.0) |
| Arrhythmia or conduction disturbance in need of a permanent pacemaker | 0 (0) | 3 (3.1) |
| Stroke | 0 (0) | 0 (0) |
| Pulmonary embolism or deep vein thrombosis | 0 (0) | 0 (0) |
| Renal complications requiring unplanned dialysis or renal replacement therapy | 0 (0) | 0 (0) |
| Data are presented as n (%). | ||
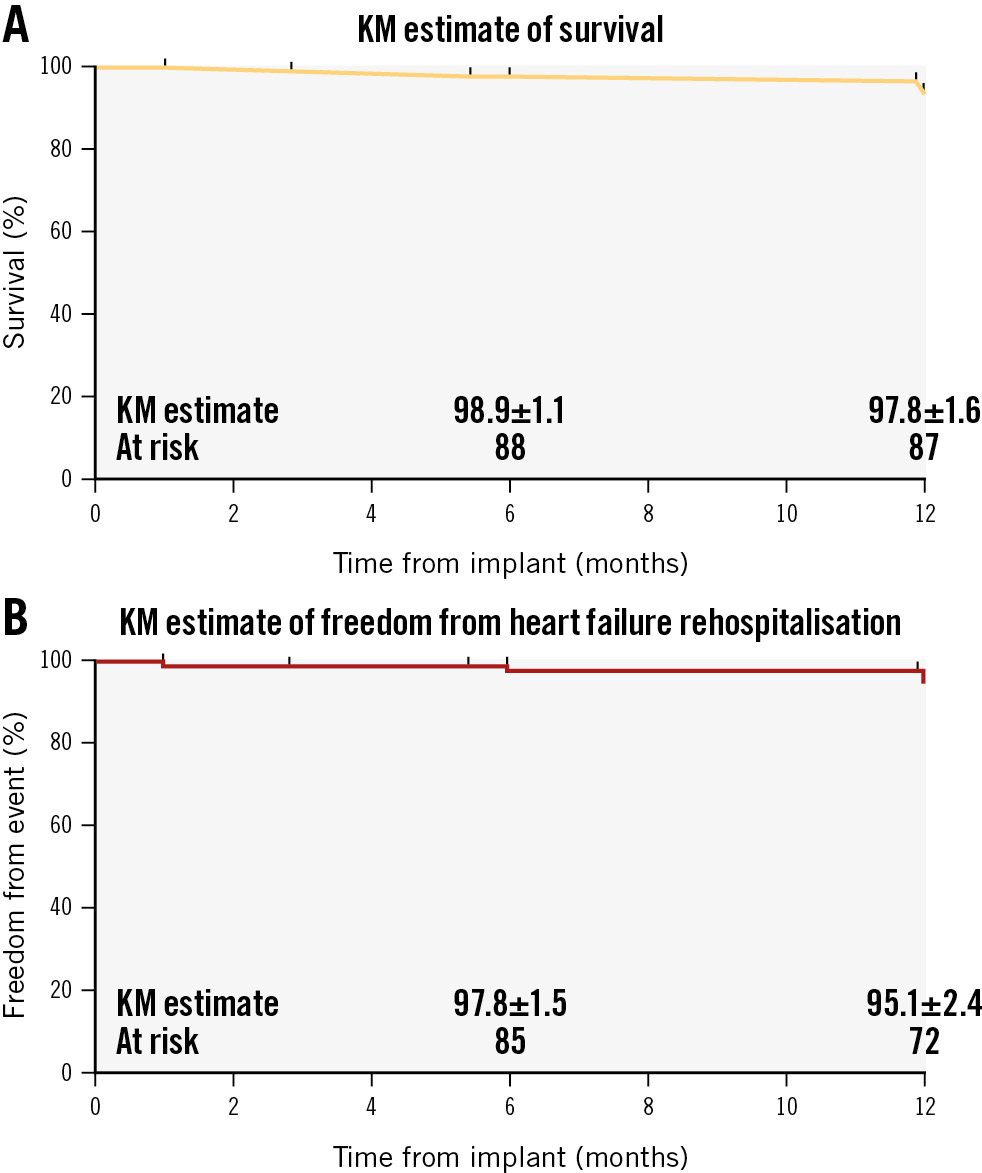
Figure 2. Kaplan-Meier curves. A) Time-to-event curve for the incidence of all-cause death. B) Time-to-event curve for the incidence of rehospitalisation for heart failure. KM: Kaplan-Meier
Echocardiographic outcomes
At 1 year, 86 patients were available for echocardiographic follow-up (Table 4). Patients experienced significant and sustained improvements in the end-diastolic septolateral tricuspid annular diameter, with an average reduction of 11.3% (41.9 mm vs 37.1 mm; p<0.001) at 1 year, compared with baseline (Central illustration). A marked reduction of TR severity at each timepoint up to 1 year was also observed in the paired cohorts (p<0.001). At 1 year, 82.5% of patients achieved ≤moderate TR (p<0.001) (Central illustration), with a ≥1-grade reduction in 97.7% and ≥2-grade reduction in 66.3%. Consistent with these findings, the vena contracta width significantly decreased from 11.8±4.1 mm to 5.6±2.3 mm (p<0.001), as did the EROA (0.9±1.0 cm² vs 0.3±0.2 cm²; p<0.001). Signs of right heart remodelling were also indicated by various echocardiographic indices, including the right atrial superior-to-inferior diameter (69.4±12.9 mm vs 64.1±16.8 mm), right atrial septolateral diameter (57.1±11.6 mm vs 50.8±12.0 mm), mid-right ventricular end-diastolic diameter (34.0±6.4 mm vs 29.2±8.5 mm), and inferior vena cava (22.3±5.4 mm vs 17.9±6.9 mm; all p<0.001). No significant differences were observed for tricuspid annular plane systolic excursion (TAPSE) or fractional area change. In the setting of a stable LVEF, significant increases in cardiac output (4.3±1.5 L/min vs 5.4±1.2 L/min; p<0.001) and stroke volume were observed (60.8±18.0 mL vs 67.6±19.8 mL; p=0.002).
Table 4. Comparison of echocardiographic parameters between baseline and follow-up.
| Baseline (n=96) | 30 days (n=94) | p-value* | 1 year (n=86) | p-value* | |
|---|---|---|---|---|---|
| RA superior-inferior diameter, mm | 69.4±12.9 | 62.0±14.2 | <0.001 | 64.1±16.8 | <0.001 |
| RA septolateral diameter, mm | 57.1±11.6 | 52.5±12.7 | <0.001 | 50.8±12.0 | <0.001 |
| Mid-RV end-diastolic diameter, mm | 34.0±6.4 | 31.4±6.2 | <0.001 | 29.2±8.5 | <0.001 |
| Base RV end-diastolic diameter, mm | 45.8±6.4 | 40.8±6.5 | <0.001 | 41.5±8.5 | <0.001 |
| Tricuspid anterior-posterior annular diameter, mm | 40.4±5.5 | 36.0±4.8 | <0.001 | 36.9±5.5 | <0.001 |
| Tricuspid septolateral annular diameter, mm | 41.9±5.3 | 37.5±5.3 | <0.001 | 37.1±6.4 | <0.001 |
| Mean vena contracta width, mm | 11.8±4.1 | 5.6±2.9 | <0.001 | 5.6±2.3 | <0.001 |
| PISA EROA, cm² | 0.9±1.0 | 0.3±0.2 | <0.001 | 0.3±0.2 | <0.001 |
| Regurgitation volume, mm³ | 69.1±47.3 | 27.0±18.0 | <0.001 | 28.3±16.1 | <0.001 |
| TAPSE, mm | 17.8±3.1 | 17.9±4.8 | 0.915 | 18.0±33.4 | 0.981 |
| FAC, % | 41.5±8.2 | 40.5±10.7 | 0.292 | 43.5±7.2 | 0.058 |
| IVC, mm | 22.3±5.4 | 18.2±6.0 | <0.001 | 17.9±6.9 | <0.001 |
| LVEF, % | 61.7±6.8 | 62.9±7.6 | 0.121 | 63.4±6.1 | 0.079 |
| LV stroke volume, mL | 60.8±18.0 | 66.6±24.2 | 0.007 | 67.6±19.8 | 0.002 |
| CO, L/min | 4.3±1.5 | 5.4±2.0 | <0.001 | 5.4±1.2 | <0.001 |
| PASP, mmHg | 42.3±7.5 | 36.7±8.0 | <0.001 | 36.9±7.9 | <0.001 |
| Mitral regurgitation | ≥0.999 | <0.001 | |||
| None | 17 (17.7) | 16 (17.0) | 29 (33.7) | ||
| Mild | 79 (82.3) | 78 (83.0) | 50 (58.1) | ||
| Moderate | 0 (0) | 0 (0) | 7 (8.2) | ||
| Severe | 0 (0) | 0 (0) | 0 (0) | ||
| E/e' ratio | 10.4±4.0 | 10.0±4.6 | 0.037 | 9.7±4.6 | <0.015 |
| Data are presented as mean±standard deviation or n (%). *Comparisons of echocardiographic parameters between baseline and follow-up in paired cohorts. CO: cardiac output; EROA: effective regurgitant orifice area; FAC: fractional area change; IVC: inferior vena cava; LV: left ventricular; LVEF: left ventricular ejection fraction; PASP: pulmonary artery systolic pressure; PISA: proximal isovelocity surface area; RA: right atrial; RV: right ventricular; TAPSE: tricuspid annular plane systolic excursion | |||||
Functional status, exercise capacity, and quality-of-life outcomes
Significant improvements in functional status, exercise capacity, and quality-of-life outcomes were observed between baseline and 1 year. Paired analysis showed that rates of patients assessed as NYHA Class I-II increased from 32.6% at baseline to 96.5% at 1-year follow-up (p<0.001) (Central illustration). The KCCQ score improved from 66.7±16.9 points to 74.4±13.5 points, reflecting a mean improvement of 7.6±17.7 points (p<0.001) (Figure 3A), while the mean 6MWD increased by 31.9±71.8 m from 304.3±81.2 m at baseline to 336.2±80.4 m at 1 year (p<0.001) (Figure 3B). At 1 year, 69.8% (60/86) of the patients required a lower dose of diuretics than at baseline, indicating less reliance on diuretic therapy in our study population. A significant improvement was observed in serum albumin level (40.8±5.0 g/L vs 44.1±2.8 g/L), while other biochemical variables including ALT, eGFR and NT-proBNP all trended towards improvement (Supplementary Table 1).
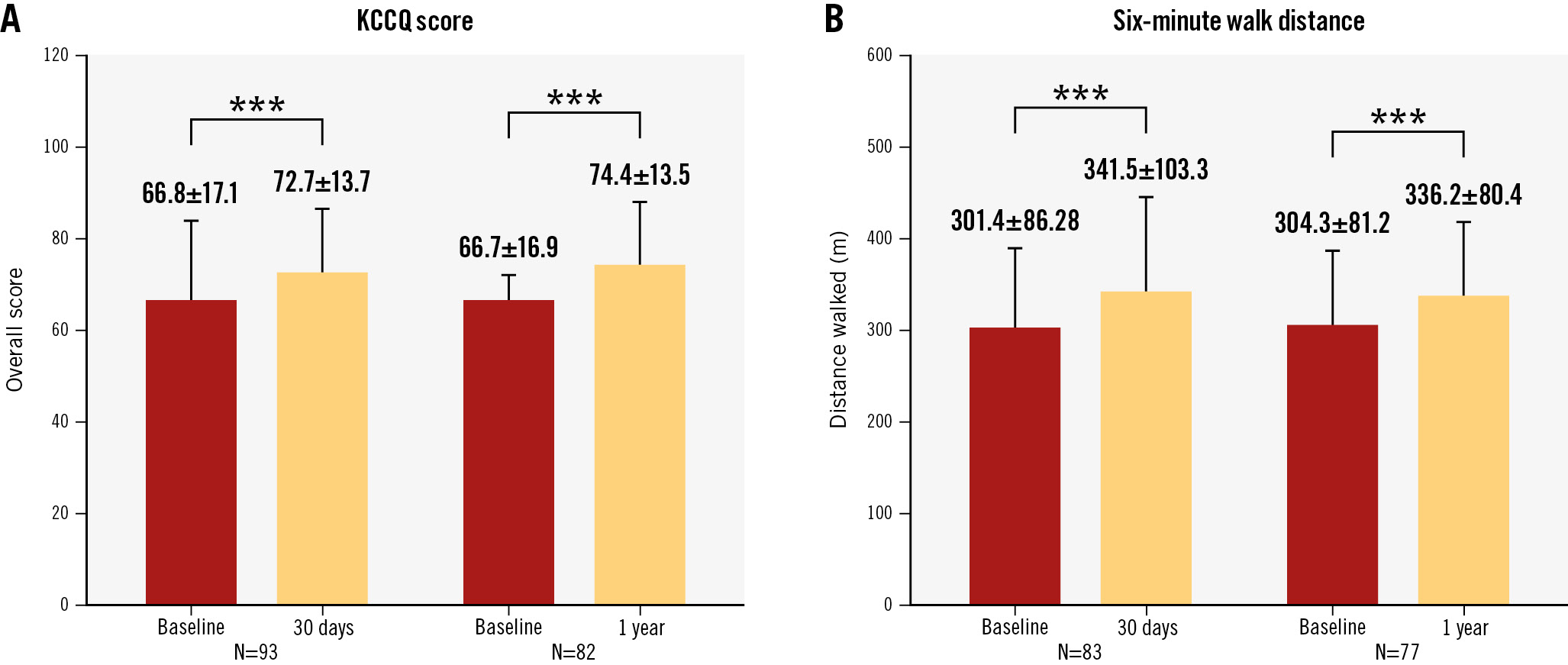
Figure 3. Functional and clinical analyses of the transcatheter K-Clip annuloplasty system. A) Kansas City Cardiomyopathy Questionnaire (KCCQ) score. B) Six-minute walk distance. ***p<0.001 as compared to baseline.
Discussion
In this paper, we reported the preliminary 1-year outcomes of the ongoing prospective multicentre TriStar study using the K-Clip tricuspid annuloplasty system. The key findings were as follows: (1) the technical and procedural success rates of the K-Clip system for severe secondary TR were 97.9% and 93.8%, respectively, without cardiovascular mortality events; (2) at 1 year, 82.5% of patients achieved ≤moderate TR with a substantial reduction of ≥1 grade in 97.7% and ≥2 grades in 66.3%; (3) marked improvement in the NYHA Functional Class was reported at 1-year follow-up, accompanied by a significantly increased 6MWD and KCCQ score.
Owing to the increased prevalence of TR burden and limited options in clinical practice, the rapid development of less invasive catheter-based therapies has advanced over the past decade16. Since the final common pathway for primary and secondary TR is progressive tricuspid annular dilation, tricuspid annuloplasty has emerged as a promising alternative17. The 1-year results with the Cardioband (Edwards Lifesciences), the first transcatheter device dedicated for tricuspid annular reconstruction, demonstrated significant and durable efficacy in TR reduction18. However, sequential annular anchoring is an extremely meticulous and time-consuming process which contributes to a considerable risk of RCA injury. Based on encouraging annuloplasty results, the K-Clip system was developed using a unique clip-based annular plication approach in an attempt to achieve tricuspid valve focal annuloplasty with improved manoeuvrability and flexibility.
In this study, immediate technical success and procedural success before discharge were achieved in 97.9% and 93.8% of patients, respectively. The mean procedural time was 157.0 min, and the time from the insertion of the first implant to the release of the final implant was 73.7 min. These are longer than the times reported for transcatheter edge-to-edge repair (TEER) systems1920, but, as a percutaneous annular reconstruction device, the K-Clip has significantly improved, easy-to-operate manoeuvrability. Folding and clamping of the tricuspid annulus through the anchor and two tooth clip arms led to an 11.3% reduction in the septolateral annular diameter, followed by a significant reduction in the EROA and mean vena contracta width. It should be noted that an improvement in TR severity was revealed in this study compared to our previous compassionate experience10. This might be due to the higher rate of secondary device implantation in this study. Based on the device’s unique clip-based, retrievable, and sutureless design, more than one clip can be located within the anterior or posterior annulus, thereby flexibly adapting to native individual TR jet origins and intraprocedural needs. The proportion of patients with ≤moderate TR significantly increased to 82.5% at 1 year, with a substantial reduction of ≥1 grade in 97.7% and ≥2 grades in 66.3%, similar to the results reported in previous studies182122.
In addition to the marked reduction of regurgitant volume, early signs of right heart remodelling were seen in our study population, as found in the TRI-REPAIR study23 and the TRILUMINATE study24. As an adaptation to improved right heart performance, cardiac output and stroke volume in our study population also increased significantly during follow-up. The significantly improved NYHA Functional Class, 6MWD, and KCCQ scores confirmed the clinical benefits of the K-Clip annuloplasty system on health status, but its long-term impact on mortality remains to be determined.
Above all, the novel K-Clip tricuspid annuloplasty system has emerged as a promising alternative for reducing TR with a considerable safety profile and easy manoeuvrability. This could cater to the unmet needs of severe secondary TR patients with large coaptation defects, non-central or very eccentric jets, or jets originating from the anteroposterior commissure, who are currently considered unsuitable for TEER25. Surgical annuloplasty with additional edge-to-edge plications and tricuspid valve-in-ring replacement have been reported to be efficacious in preventing recurrent TR with satisfactory midterm clinical outcomes26. Whether percutaneous tricuspid annuloplasty with leaflet edge-to-edge repair or transcatheter tricuspid valve implantation could improve severe TR remains to be explored.
Several issues with the present study should be addressed. First, a CT scan, an optional imaging choice for TEER, is crucial for K-Clip procedural planning. Hence, a comprehensive anatomical evaluation of the tricuspid valve, right ventricle, vena cava and its spatial relationships with the RCA according to CT analysis must be cautiously discussed in the preprocedural multidisciplinary team meeting. Second, the reduction in the septolateral diameter after K-Clip implantation appears to be lower than that documented in previous studies using direct ring-based annuloplasty27. The long-term stability and durability of this clip-based sutureless system has yet to be proven. Third, although the K-Clip annuloplasty system seems to be more versatile in complex TV anatomies, including extreme annulus dilation and multiple regurgitant jets, leaflet malcoaptation could still exist. Hence, its indications should not be easily generalised, and combined therapies are promising. In addition, surrounding cardiac structure injury remains a concern for K-Clip implantation. Aside from the case of RCA perforation documented in our study, three other patients experienced transient hypotension and bradycardia during plication. A downsized clip was substituted to mitigate the risk of RCA compression. Hence, further exploration of the impact of annular reduction with the K-Clip system on RCA distribution should be of utmost clinical significance.
Limitations
This was a single-arm observational study with a limited number of patients and a midterm follow-up. The absence of a lead-to-lead comparison with the gold standard for tricuspid treatment limits conclusions about feasibility. Additionally, most patients in this study presented with a still rather preserved ventricular function at baseline, indicating a considerable proportion of patients with heart failure and preserved ejection fraction within the cohort. Hence, despite these encouraging findings, further exploration of their long-term outcomes as well as studies enrolling patients with heart failure and reduced ejection fraction are warranted to make a comprehensive assessment on the haemodynamic and clinical benefits of K-Clip transcatheter annuloplasty system in treating secondary TR.
Conclusions
The K-Clip annuloplasty system demonstrated high device safety with a remarkable reduction in TR severity, accompanied by early evidence of right heart remodelling and significant improvements in cardiac function and health status at 1 year. Follow-up is ongoing to determine its durability and benefits for long-term clinical prognosis.
Impact on daily practice
The 1-year results of the TriStar study demonstrated that the K-Clip transcatheter reconstruction system substantially reduced the severity of tricuspid regurgitation (TR), with an acceptable safety profile, followed by improvements in functional status, exercise capacity and quality of life. Longer-term follow-up data and further prospective randomised trials are warranted to confirm the safety, efficacy and durability of this interventional technique in reducing TR and improving functional and clinical outcomes.
Funding
This work was funded by Shanghai Huihe Healthcare Technology Co., Ltd.
Conflict of interest statement
The authors have no conflicts of interest relevant to the contents of this paper to declare.
Supplementary data
To read the full content of this article, please download the PDF.

