Abstract
Aims: Poor left ventricular function is considered a high risk condition for performing either percutaneous (PCI) or surgical revascularisation. The aim of this study was to evaluate immediate and long term results of PCI in patients with coronary artery disease (CAD) and severe left ventricular dysfunction (EF < 0.30).
Methods and results: Seventy-eight consecutive patients with CAD and severe left ventricular dysfunction (EF < 30%) were selected. The majority of these patients (87%) had multivessel disease. Coronary angioplasty procedure was mainly motivated by angina associated with clinical manifestation of heart failure (54%). Total number of treated vessels was 181, and a total of 203 stents were implanted (2.6 stent/patient). Procedural success was achieved in 77 patients (97.8%). The total procedural and in-hospital adverse event rate was 7.8%. Mean follow-up period (FU) was 25±6 months. Event-free survival rate at the end of FU was 55%; repeat revascularisation was performed in 21 patients (27.6%). Female gender, diabetes, new acute myocardial infarction and number of treated vessels were independent predictors for death and combined mayor adverse cardiac events (MACE) during the follow-up.
Conclusions: In symptomatic patients with CAD and severe left ventricular dysfunction, PCI can be performed with excellent procedural outcome and acceptable long-term morbidity and mortality.
Introduction
Severe left ventricular dysfunction in patients with coronary artery disease is often, but not always, the consequence of an acute myocardial infarction or an ischaemic cardiomyopathy1. It is estimated that most patients with severe left ventricular dysfunction, undergoing heart transplantation, might have areas of hibernated myocardium and could benefit from myocardial revascularisation2. Even symptomatic patients are often denied appropriate interventional treatment due to fear of procedural failure and are maintained on heavy medical therapy, the fortunate few receiving cardiac transplantation. Such patients have very limited survival when treated medically3-4 and usually die of cardiac causes related to recurrent ischaemia or infarction, cardiac heart failure or ventricular arrhythmias.
In patients with ischaemic dilated cardiomyopathy, myocardial revascularisation is highly indicated when myocardial viability is documented by non-invasive tests (dobutamine stress echocardiography, thallium 201 scintigraphy, PET). However, it must be also underlined that poor left ventricular function, per se, is considered a high risk condition for either percutaneous or surgical coronary revascularisation5. The clinical benefit of percutaneous coronary interventions (PCI) in such patients is not well established.
The aim of the present study was to evaluate immediate and long-term procedural and clinical outcome of PCI in patients with obstructive coronary disease (mostly multivessel) and severe left ventricular dysfunction (EF <30%).
Methods
Study group
Between January 2000 and December 2001, from the patients undergoing percutaneous coronary interventions in our centre after having performed myocardial viability studies, we selected 78 consecutive patients with documented coronary artery disease, severe left ventricular dysfunction (EF< 30%) and evidence of viable myocardium. Indication for coronary angiography were made by the referring physician on the basis of chest pain and/or cardiac failure manifestations. Indications for PCI were made by the attending physician after having reviewed the coronary angiography and the results of myocardial viability studies. Myocardial viability was documented in all patients by SPECT and/or by dobutamine stress echocardiography. Left ventricular ejection fraction was assessed by transthoracic echocardiogram (using a planimetric method) and by left ventriculography at the time of diagnostic angiography.
Cardiac catheterisation
Diagnostic coronary angiography was performed in all patients to assess left ventricular function and the presence and extent of coronary artery disease. The left ventricular ejection fraction was estimated by a single-plane 35°RAO ventriculogram. PCI was performed in all patients. Intention to treat was for complete revascularisation by PCI in all patients. Procedural and in-hospital events were monitored in all.
Long-term clinical follow-up
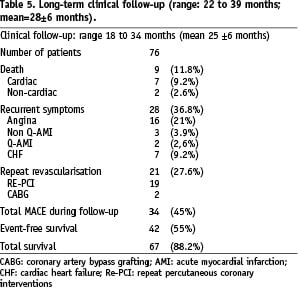
Patients were followed with out-patient visits after one, three and every six months or by phone interviews. At each visit, health status was assessed by a complete cardiac evaluation, including echocardiogram, when needed. An echocardiogram for the assessment of regional and global left ventricular function was performed routinely at discharge and at six months after PCI. Non-fatal and fatal cardiac events were documented either by hospital charts or by coroner’s reports. Mean follow-up period was 28±6 months (range: 22 to 39 months) and was completed in 92.3% (72/78 patients). The following events were recorded during follow-up: 1) cardiac and non-cardiac death, 2) recurrent symptoms, 3) recurrent ischaemic events (myocardial infarction non-Q and Q-AMI), 4) congestive heart failure, 5) need for revascularisation (re-PCI or CABG). EF on transthoracic echocardiography was also evaluated.
Statistical analysis
Data are expressed as mean±SD or as percentage where appropriate. Multivariate analysis was performed using a Cox proportional hazard model to assess predictor clinical factors of long term outcome. A p value of less than 0.05 was considered significant. The following variables were entered in the multivariate analysis: age, sex, coronary risk factors, extent of coronary artery disease, left ventricular function, complete/incomplete revascularisation, peri-procedural cardiac enzyme release previous myocardial infarction, previous CABG, previous PCI.
Results
Demographic and clinical characteristics of study patients are shown in Table 1.
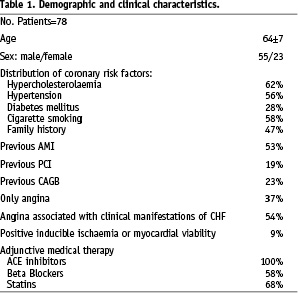
The majority of patients (71%) were males, mean age was 64±7 years with 27 patients having more than 65 and nine more than 75 years. Hypertension, diabetes, hypercholesterolaemia, a history of smoking and a family history of coronary artery disease were common, 36 patients (53%) had experienced a previous Q wave or non-Q myocardial infarction. Thirty-three patients (42%) had undergone a previous myocardial revascularisation: 15 (19%) by PCI and 18 (23%) by coronary artery bypass surgery (CABG). All patients were on medical therapy with ACE-inhibitors. 58% were also receiving beta blockers and 68% of patients were on statins. Double anti-platelet therapy (ASA + clopidogrel or ticlopidine) was continued for at least one month following the procedure in all patients.
Clinical indications for PCI procedure were mostly represented by angina and/or heart failure manifestations: 29 patients had only angina and 42 patients had angina associated with congestive heart failure manifestation, in seven patients PCI procedure was performed on the basis of the worsening of left ventricular function associated with a positive non-invasive test for inducible ischaemia or myocardial viability. Left ventricular EF was 25±2% ranging from 23 to 28%.
Angiographic characteristics are summarised in Table 2.

Ten patients (13%) had single vessel disease and 68 (87%) had multivessel disease. All 18 patients with previous CABG had multivessel disease with at least one venous graft disease.
Procedural and in-hospital outcomes
All patients received IIb/IIIa antagonists prior to procedure. In 36 patients (47%) intra-aortic balloon contra-pulsation was used during the PCI procedure. Complete revascularisation by PCI was attempted in all patients with 181 vessels treated (2.3 vessel/ patient), 23% of treated vessel were left anterior descending arteries, 8% were left main, 9% were diagonal branches, 17% were left circumflex arteries, 10% were marginal branches, 22% were right coronary arteries, 10% were posterior descending or postero-lateral branches and 6% were saphenous vein grafts (Table 2). Multivessel PCI was performed in 88% of patients. Multiple stent implantation was used in 76% of patients. Debulking before stent implantation by directional atherectomy was performed in 4% of patients. In two patients, rotational ablation atherectomy was needed because of heavily calcified lesions. A total of 203 stents were implanted (2.6 stent /patient, 1.1 stent / vessel) .
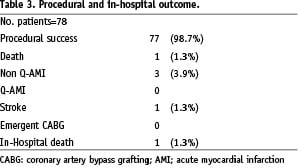
Technical procedural success was obtained in 98.7% of patients. Procedural death occurred in one patient (1.3%), non Q-AMI in three patients (3.9%), stroke in one patient (1.3%), emergency CABG was not needed in any case. One death occurred two weeks following the procedure for acute renal failure. No other complication were observed. Thus total procedural and in-hospital major adverse cardiac events were 7.7% (Table 3).
Clinical follow-up
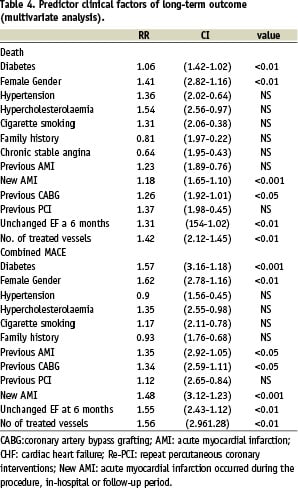
During a mean follow up of 25±6 months 42 patients (55%) had event-free survival. Nine patients (11.8%) died: seven (9.2%) cardiac, and two (2.6%) non-cardiac. Cardiac death was caused by left heart failure in three patients, ischaemic events in two patients and ventricular fibrillation in two patients. Sixteen patients (21%) had recurrence of angina, while four patients (5.3%) had new MI (three non-Q, and one Q-AMI) and five patients (6.6%) were hospitalised because of congestive heart failure. All 20 patients (26.3%) with recurrent angina or AMI required a repeat PCI. Thus, total major adverse cardiac events during the follow up were 44.7%, mainly represented by the need for repeat revascularisation. In 39 patients, left ventricular function assessed by echocardiography improved at six months from 25±2% to 39±5%, p< 0.001. In the remaining 37 patients, left ventricular ejection fraction was unchanged (basal conditions 26±4 vs 6-month follow-up 28±5, ns).
Female gender, the presence of diabetes mellitus, previous CABG, new AMI, unchanged EF at six months following the procedure and the number of treated vessels were significantly predictors of death during follow-up.The same parameters were also significant predictors for combined major adverse cardiac events during follow-up (Table 4).
Discussion
Immediate and long-term prognosis of patients with ischaemic cardiomyopathy following myocardial revascularisation (both mechanical or surgical) have been investigated in few studies, and among these the majority were undertaken many years ago when technological and medical treatments were different from the ones available at the present time (Table 5)5-11.
Technical advances in the field of interventional cardiology with the introduction of coronary stents and potent antiplatelet therapies have resulted in a marked increase in the complexity of procedures and expanded indications, even in patients considered as high-risk, like those included in the present study. One of the striking findings of this study is the safety of PCI as well as the favourable long term outcome in patients with severe left ventricular dysfunction due to coronary artery disease. This is in contrast with the data recently reported by Keelan at al from the DYNAMIC Registry12 where there was an observed increased in-hospital MACE (including mortality) in patients with left ventricular ejection fraction < 40 undergoing PCI. This discrepancy might be due to several factors: the Registry included patients recruited in 10 different sites, GP IIb/IIIa inhibitors were used in only 24% of patients and it was not mentioned if intra-aortic balloon contra-pulsation was used while in the present study IIb/IIIa inhibitors were used in all patients and in almost half of patients intra-aortic balloon contra-pulsation was employed during the procedure.
Medical treatment for patients with severe left ventricular function and coronary artery disease is associated with poor prognosis. Luciani and colleagues reported 28%, five year survival in 72 patients with an average EF of 21%4. However, medical treatment have also been markedly improved over the last decade. Although many questions remain in attempting to optimise the management for these patients, myocardial revascularisation seems to offer some advantages.
The BARI study showed a seven year survival after PTCA in patients with reduced left ventricular function (EF<50%) and two vessel disease of 78%13. The same study provided evidence that there was no significant differences in seven year survival in an initial strategy of PTCA or CABG among non-diabetic patients with three-vessel disease, and among all patients with two-vessel disease involving the proximal left anterior descendent artery. The authors suggested that PTCA may be offered to such patients as an initial revascularisation without compromising the seven year survival.
Hasdai and colleagues, reported among 611 patients undergoing PCI a higher 10 year mortality in patients with left ventricular dysfunction (EF<40%) compared to those with normal systolic function (47% vs 20%; p<0,0001)14.
In a recent study by Li et al15, 74 patients with ejection fraction < 40% underwent percutaneous coronary angioplasty, including 61 with stent back-up. During long-term follow up (29±23 months), 58 (87.9%) of 66 patients with clinical success were alive, including 44 (68.6%) free from cardiac events. In the present study, total survival at 25±6 months was 88.2% (with an event-free survival 55%), similar to that reported in the previous studies, despite a more severe left ventricular dysfunction (EF < 30%) than those reported in those studies13-15.
Recent reports suggest that approximately 50% of patients with ischaemic cardiomyopathy have viable myocardium, and 25% of these have sufficient amounts of viable myocardium resulting in a functional improvement (increased left ventricular ejection fraction, increased exercise capacity) following myocardial revascularisation16-18. Improved functional status and symptoms after revascularisation, even with limited preoperative assessment of myocardial viability, has been reported by Christian et al17.
All patients included in the present study had documented viable myocardium, but only 39 patients showed a significant improvement of ejection fraction at six months following the procedure. Improvement in left ventricular function after PCI is an important predictor of long-term outcome. Multivariate analysis showed that unchanged EF at follow-up was significantly associated with increased cardiac death and MACE. Worsening of left ventricular function related to new myocardial damage, either by intraprocedural AMI or occurring during follow-up, were also significant predictors for death and MACE. Multivessel PCI as well as diffuse disease are often associated with slight increases of cardiac enzymes, and this may be the cause of greater occurrence of cardiac events in diabetics and in patients with complex lesions and side-branch involvement. The loss of even small amounts of viable myocardium in these patients adds a further myocardial damage and might negatively influence long-term clinical outcome as shown by multivariate analysis.
Taking into account these considerations, long-term clinical outcome is acceptable considering the natural history in this patient subset. Total survival at > 2 years is excellent, although only 50% may benefit from PCI procedures with event-free survival. The key for a favourable long-term clinical outcome seems to be the recovery in left ventricular function, but unfortunately this cannot always be predicted by myocardial viability tests. All patients included in the present study had positive test, but only 53% had improvement of EF. On the other hand, it has been reported that myocardial revascularisation might improve functional capacity in patients with left ventricular dysfunction and negative myocardial viability test19,20. Incomplete revascularisation was not associated with unfavourable outcome. In this series, revascularisation was limited to vessels supplying areas with documented viable myocardium, thereby suggesting that in most cases an incomplete revascularisation may benefit patients with left ventricular dysfunction if PCI is performed on the vessel targeted by the viability study. Caution should be made in patients selection; those with diffuse and complex coronary lesions are at higher risk for procedural complications, especially biomarker elevations following procedure must be carefully evaluated. Complex procedures associated with biomarker elevations might even worsen long-term clinical outcome in these patients.
Another important finding of this study is that long-term mortality remains high, even after successful coronary intervention. We found an overall mortality of 11% at 24 month follow-up, compared to 6% in the study by Di Sciascio21. However, patients in our study had more frequent diabetes (28% versus 14%), more prior CABG (23% versus 6%), and more multivessel coronary disease (87% versus 54%). The mortality of 11% at 24-month follow-up in our study compares favourably to the 15-25% mortality rate at two- to three-year follow-up reported in the angioplasty era.
Study limitations
This is a non-randomised, retrospective, observational study with the all the inherent limitations of such a trial. There is no control group, however, it is well documented that prognosis in this subgroup of patients (EF<0.30) is very poor, even over a short time period. Event-free survival of 55% after a time period of 25±8 months could be considered poor, but the main component of major cardiac events during the follow up period was recurrence of angina and repeat revascularisation. It could be speculated that better results might be obtained with the use of drug-eluting stents. Finally, all patients included in this study were on treatment with ACE-inhibitors, and the majority were also treated with statins (68%) and beta blockers (58%), and this might influence clinical outcome.
In conclusion, PCI in patients with severe left ventricular dysfunction and documented viability can be performed with excellent procedural and in-hospital outcome in those patients with severe ischaemic left ventricular dysfunction and evidence of viable myocardium. Predictors for unfavourable long-term outcome are represented by female gender, the presence of diabetes, extent of coronary disease, lack of recovery in left ventricular function and new episodes of myocardial infarction. In some of these patients, PCI and repeat procedures represent the only therapeutic option. In addition, in carefully selected subgroups of patients with severe left ventricular dysfunction and coronary artery disease, PCI may be considered as an alternative or a “bridge” approach for cardiac transplantation.

