Abstract
The management of patients with coronary artery disease requiring percutaneous coronary revascularisation has undergone a significant transformation over the past several decades. With the increasing use of early invasive strategies, the role of pretreatment with oral antiplatelet agents has evolved. Additionally, the introduction of novel agents such as cangrelor has had a significant impact on the management of thrombotic risk. In this review, we summarise the current evidence on the role of pretreatment with antiplatelet agents across the spectrum of coronary artery disease, from non-ST-segment elevation acute coronary syndromes and ST-segment elevation myocardial infarction to stable coronary artery disease. We also highlight data on novel parenteral agents, both available and in development.
Dual antiplatelet therapy, with aspirin and a P2Y12 inhibitor, is an established treatment regimen for patients with coronary artery disease (CAD), including acute coronary syndrome (ACS). However, the timing of oral P2Y12 inhibitor administration remains a topic of debate. In patients undergoing coronary revascularisation, the term “pretreatment” refers to the administration of a P2Y12 inhibitor before the coronary anatomy is defined via coronary angiography1. The administration may occur at a variable timepoint – en route to the hospital, in the emergency department, or immediately prior to the procedure.
Advantages that have been cited in favour of pretreatment include achieving greater platelet inhibition, which can potentially provide ischaemic protection and reduce periprocedural thrombotic complications123 (Figure 1). However, the disadvantages of pretreatment include excess bleeding risk in patients who require percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG)12. In this review, we summarise the current evidence for pretreatment with oral antiplatelet agents in patients with non-ST-segment elevation acute coronary syndromes (NSTE-ACS), ST-segment elevation myocardial infarction (STEMI), and stable CAD undergoing PCI, along with highlighting data for the intravenous antiplatelet agent, cangrelor, and newer parenteral agents on the horizon.

Figure 1. Potential advantages and disadvantages of pretreatment with oral antiplatelet agents. CABG: coronary artery bypass grafting; ED: emergency department; GP: glycoprotein
Treatment with oral antiplatelet agents in non-ST-segment elevation acute coronary syndromes
Adequate platelet inhibition is an essential component of treatment in NSTE-ACS, but the timing of oral antiplatelet agent administration is debated. Both the American and European guidelines have significantly shifted over time with regard to recommendations on pretreatment (Table 1). The 2018 European Society of Cardiology (ESC) Guidelines on myocardial revascularization have a Class IIa recommendation for pretreatment with ticagrelor or clopidogrel in NSTE-ACS4. The 2023 ESC Guidelines for the management of ACS have a Class III recommendation against routine pretreatment with a P2Y12 inhibitor in NSTE-ACS patients in whom the coronary anatomy is not known and early invasive management (<24 hours) is planned. Furthermore, there is a Class IIb recommendation that pretreatment may be considered in patients undergoing a primary PCI strategy or in those who are not expected to undergo an early invasive strategy and do not have a high bleeding risk, which is similar to the recommendation in the 2025 ACC/AHA ACS guidelines56. Conversely, in the most recent 2021 ACC/AHA/Society for Cardiovascular Angiography and Interventions (SCAI) Guideline for Coronary Artery Revascularization, there are no specific recommendations with regard to pretreatment7.
Table 1. Summary of the latest American and European guidelines.
| Guidelines | Recommendations | Class of Recommendation | Level of Evidence |
|---|---|---|---|
| Oral pretreatment in NSTE-ACS | |||
| 2018 ESC Guidelines on myocardial revascularization4 | For pretreatment in patients with NSTE-ACS undergoing invasive management, ticagrelor administration (180 mg loading dose, 90 mg bid), or clopidogrel (600 mg loading dose, 75 mg daily dose) if ticagrelor is not an option, should be considered as soon as the diagnosis is established. | IIa | C |
| 2023 ESC Guidelines for the management of acute coronary syndromes5 | Pretreatment with a P2Y12 receptor inhibitor may be considered in NSTE-ACS patients who are not expected to undergo an early invasive strategy. | IIb | C |
| Pretreatment with a P2Y12 receptor inhibitor may be considered in patients undergoing a primary PCI strategy. | IIb | B | |
| Routine pretreatment with a P2Y12 receptor inhibitor is not recommended in NSTE-ACS patients in whom the coronary anatomy is not known and early invasive management is planned. | III | A | |
| 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization7 | In patients with ACS undergoing PCI, a loading dose of P2Y12 inhibitor, followed by daily dosing, is recommended to reduce ischaemic events. | I | B-R |
| 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes6 | In patients with NSTE-ACS planned for an invasive strategy with the timing of angiography anticipated to be >24 hours, upstream treatment with clopidogrel or ticagrelor may be considered to reduce MACE. | IIb | B-NR |
| Oral pretreatment in STEMI | |||
| 2018 ESC Guidelines on myocardial revascularization4 | A potent P2Y12 inhibitor (prasugrel or ticagrelor), or clopidogrel if these are not available or are contraindicated, is recommended before (or at the latest at the time of) PCI and maintained over 12 months, unless there are contraindications such as excessive risk of bleeding. | I | A |
| Administration of prasugrel in patients in whom the coronary anatomy is not known is not recommended. | III | B | |
| 2023 ESC Guidelines for the management of acute coronary syndromes5 | Pretreatment with a P2Y12 receptor inhibitor may be considered in patients undergoing a primary PCI strategy. | IIb | B |
| 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization7 | In patients with ACS undergoing PCI, a loading dose of P2Y12 inhibitor, followed by daily dosing, is recommended to reduce ischaemic events. | I | B-R |
| Oral pretreatment in stable CAD | |||
| 2018 ESC Guidelines on myocardial revascularization4 | Treatment with 600 mg clopidogrel is recommended in elective PCI patients once the coronary anatomy is known and a decision is made to proceed with PCI. | I | A |
| Pretreatment with clopidogrel may be considered if the probability of PCI is high. | IIb | C | |
| 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization7 | In patients with SIHD undergoing PCI, a loading dose of clopidogrel, followed by daily dosing, is recommended to reduce ischaemic events. | I | C-LD |
| Cangrelor | |||
| 2018 ESC Guidelines on myocardial revascularization4 | Cangrelor may be considered in P2Y12 inhibitor-naïve patients undergoing PCI. | IIb | A |
| 2023 ESC Guidelines for the management of acute coronary syndromes5 | In P2Y12 receptor inhibitor-naïve patients undergoing PCI, cangrelor may be considered. | IIb | A |
| 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization7 | In patients undergoing PCI who are P2Y12 inhibitor naïve, intravenous cangrelor may be reasonable to reduce periprocedural ischaemic events. | IIb | B-R |
| 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes6 | Among patients with ACS undergoing PCI who have not received a P2Y12 inhibitor, intravenous cangrelor may be reasonable to reduce periprocedural ischaemic events. | IIb | B-R |
| ACC: American College of Cardiology; ACEP: American College of Emergency Physicians; ACS: acute coronary syndrome; AHA: American Heart Association; CAD: coronary artery disease; ESC: European Society of Cardiology; LD: limited data; MACE: major adverse cardiovascular events; NAEMSP: National Association of EMS Physicians; NR: non-randomised; NSTE-ACS: non-ST-segment elevation acute coronary syndrome; PCI: percutaneous coronary intervention; R: randomised; SCAI: Society for Cardiovascular Angiography and Interventions; SIHD: stable ischaemic heart disease; STEMI: ST-segment elevation myocardial infarction | |||
Clopidogrel
Clopidogrel is the most widely studied P2Y12 inhibitor, and older guidelines that recommended pretreatment were based on the PCI-Clopidogrel in Unstable angina to prevent Recurrent Events (PCI-CURE)8 and Clopidogrel for the Reduction of Events During Observation (CREDO) trials9. In the 2001 PCI-CURE trial, pretreatment with aspirin and clopidogrel led to a 31% reduction in cardiovascular death or myocardial infarction (MI) along with lower use of glycoprotein (GP) IIb/IIIa inhibitors8. However, the median time from randomisation to PCI was 6 days in the clopidogrel arm8, and therefore, the results are not directly translatable to the current era in which times to coronary angiography are shorter. The CREDO trial investigated treatment with a P2Y12 inhibitor at a shorter interval before PCI (between 3 and 24 hours) and found it did not significantly reduce the combined risk of death, MI, or urgent target vessel revascularisation. However, there was a 38.6% relative risk reduction in patients who received clopidogrel more than 6 hours before PCI9. A post hoc analysis of CREDO demonstrated a strong relationship between the duration of pretreatment and the occurrence of the primary endpoint, with statistical significance seen after 15 hours of clopidogrel pretreatment10.
While the PCI-CURE and CREDO trials compared clopidogrel pretreatment to placebo, the Antiplatelet therapy for Reduction of MYocardial Damage during Angioplasty (ARMYDA)-5 trial compared the administration of clopidogrel 4 to 8 hours before PCI with the administration of clopidogrel in the catheterisation lab after coronary angiography but before PCI. In a population in which 39% of patients presented with NSTE-ACS, the trial showed no differences in the primary ischaemic endpoint or risk of bleeding with pretreatment, highlighting that pretreatment may be unnecessary11. Further evidence that in-lab clopidogrel loading after defining coronary anatomy leads to similar outcomes as clopidogrel pretreatment in the ACS population was seen in the Cornell Angioplasty Registry12. Overall, the existing evidence appears to suggest that there is no clear clinical benefit of routine pretreatment with clopidogrel; however, there may be benefit in patients with a prolonged delay to angiography.
Ticagrelor
Older evidence in support of using ticagrelor for pretreatment came from the PLATelet inhibition and patient Outcomes (PLATO) trial in which ticagrelor was associated with a reduction in the risk of ischaemic events in ACS patients compared with clopidogrel13. However, patients in both arms of the trial received the P2Y12 inhibitor before coronary angiography, so the trial was not specifically designed to evaluate a pretreatment strategy13.
A trial by Bonello et al specifically evaluated the timing of P2Y12 inhibitor treatment by randomising patients to either ticagrelor at least 4 hours before PCI or prasugrel given at the time of PCI; it demonstrated that earlier treatment with ticagrelor significantly reduced the occurrence of periprocedural myonecrosis in patients with NSTE-ACS who underwent PCI14. However, the rates of major adverse cardiovascular events (MACE) at 1-month follow-up were similar between the groups14. This was a not a true pretreatment study as it only included patients with a PCI indication in whom use of a P2Y12 inhibitor was indicated. The more contemporary Intracoronary Stenting and Antithrombotic Regimen: Rapid Early Action for Coronary Treatment (ISAR-REACT) 5 Trial allowed for a comparison between P2Y12 inhibitor pretreatment and P2Y12 inhibitor administration after ascertaining coronary anatomy in patients with ACS, and found a prasugrel-based strategy with deferred loading to be superior to ticagrelor pretreatment in regard to the incidence of death, MI, or stroke15. This benefit of a prasugrel-based strategy was also seen in the subset of patients with NSTE-ACS in a post hoc analysis of outcomes at 1 year16. This lack of advantage with ticagrelor pretreatment was also demonstrated in the Downstream Versus Upstream Strategy for the Administration of P2Y12 Receptor Blockers In Non-ST Elevated acUte Coronary Syndromes With Initial Invasive Indication (DUBIUS) trial in which there was no difference in the composite of death due to vascular causes17. The mean time between P2Y12 administration and coronary angiography was only 61 minutes in ISAR-REACT 5 and 24 hours in DUBIUS, so the lack of benefit with pretreatment cannot be extrapolated to patients not undergoing an early invasive strategy.
Prasugrel
The Comparison of Prasugrel at the Time of Percutaneous Coronary Intervention Or as Pretreatment At the Time of Diagnosis in Patients With Non-ST-Elevation Myocardial Infarction (ACCOAST) trial evaluated pretreatment with prasugrel in NSTE-ACS patients scheduled for PCI within 48 hours18. The primary efficacy endpoint was similar between prasugrel at the time of PCI versus pretreatment; however, patients with pretreatment had double the risk of major bleeding complications18, which was consistent across varying durations of pretreatment19. Furthermore, the ACCOAST Study found that there was no additional benefit with pretreatment in terms of ischaemic endpoints, thrombus burden, stent thrombosis, rates of urgent revascularisation, or the use of GPIIb/IIIa inhibitors (GPIs)20.
Overall
Observational data on pretreatment in patients with NSTE-ACS also argue against routine pretreatment with P2Y12 inhibitors. The Melbourne Interventional Group registry showed no difference in 30-day mortality, MACE, or bleeding complications with pretreatment in a group of 6,817 patients21. Similarly, pretreatment with any P2Y12 inhibitor in 64,857 patients from the Swedish Coronary Angiography and Angioplasty Registry did not improve survival or stent thrombosis, but it did lead to an increased risk of in-hospital bleeding22. Increased risk of bleeding with pretreatment was also seen in all patients as well as, specifically, patients undergoing PCI in a meta-analysis of 32,383 NSTE-ACS patients from 7 studies23. Excess bleeding risk is particularly concerning in patients who require future CABG, with a retrospective analysis of patients from the Acute Coronary Treatment and Intervention Outcomes Network Registry demonstrating longer times from catheterisation to CABG, along with a higher risk of post-CABG major bleeding in patients receiving pretreatment24.
Overall, data from randomised trials (Table 2) and observational studies suggest that routine pretreatment should not be pursued in patients with NSTE-ACS given the lack of benefit and association with harms such as the risk of bleeding. This is also supported by a contemporary meta-analysis of 7 randomised controlled trials (RCTs) in which pretreatment did not lower the risk of short-term MACE, MI, or cardiovascular death, with no difference in findings when stratified by P2Y12 inhibitor type, revascularisation strategy, or arterial access site. However, most trials included in this analysis had a median time to angiography of 24 hours, and therefore, the results are not applicable to patients who experience a prolonged delay to angiography25. Furthermore, if a conservative strategy without angiography is pursued for NSTE-ACS management, then prompt treatment with a P2Y12 inhibitor is indicated.
While guidelines and results from trials do not support routine pretreatment in patients with NSTE-ACS, real-world registry data utilising a survey to assess clinical practices in Europe demonstrated heterogeneity in the proportion of patients who were pretreated with an oral P2Y12 inhibitor, which was most commonly initiated in the emergency room26.
Table 2. Summary of randomised clinical trials of pretreatment with oral P2Y12 inhibitors in patients with NSTE-ACS.
| Trial (year) | # of patients | Patient population | Intervention | Duration of pretreatment | Primary endpoint | Results |
|---|---|---|---|---|---|---|
| PCI-CURE (2001)8 | 2,658 | NSTE-ACS undergoing PCI | 300 mg clopidogrel loading dose or placebo | Median of 6 days before PCI & median of 10 days overall | A composite of cardiovascular death, MI, or urgent target vessel revascularisation within 30 days of PCI | 4.5% clopidogrel versus 6.4% placebo RR 0.7, 95% CI: 0.50-0.97; p=0.03 |
| CREDO (2002)9 | 2,116 | Referred for planned PCI or coronary angiography (13.7% recent MI, 52.8% unstable angina, 32.8% stable angina) | 300 mg clopidogrel loading dose or placebo, 3 to 24 hours before PCI | Prespecified time-to-treatment intervals of 3-6 hours, 6-12 hours, and 12-24 hours prior to PCI | 28-day incidence of a composite of death, MI, or urgent target vessel revascularisation | No significant reduction 18.5% clopidogrel, 95% CI: –4.2% to 41.8%; p=0.23 |
| ARMYDA-5 (2010)11 | 409 | Stable angina or NSTE-ACS undergoing coronary angiography (39% NSTE-ACS) | 600 mg clopidogrel loading dose before PCI or 600 mg loading dose given in the catheterisation lab after coronary angiography but prior to PCI | 4-8 hours before PCI | 30-day incidence of major adverse cardiac events: cardiac death, MI, or unplanned target vessel revascularisation | No significant difference 10.3% preload versus 8.8% in-lab; p=0.72 |
| Bonello et al (2015)14 | 213 | NSTE-ACS | 180 mg ticagrelor loading dose as soon as possible after admission and before PCI or 60 mg prasugrel loading dose at the time of PCI | Mean of 13.4 hours | Periprocedural myonecrosis defined by an increase of >5 times the 99th percentile in troponin-negative patients or 20% in troponin-positive patients | 19.8% ticagrelor versus 38.3% prasugrel; p=0.03 |
| ISAR-REACT 5 (2019)15 | 4,018 | ACS and for whom invasive evaluation was planned (46.2% NSTEMI, 12.7% unstable angina) | 180 mg ticagrelor loading dose as soon as possible or 60 mg prasugrel loading dose (as soon as possible after randomisation for STEMI, but after the coronary anatomy is known for NSTE-ACS) | Mean of 61 minutes | A composite of death, MI, or stroke at 1 year | 9.3% ticagrelor versus 6.9% prasugrel HR 1.36, 95% CI: 1.09-1.70; p=0.006 |
| DUBIUS (2020)17 | 1,449 | NSTE-ACS undergoing invasive treatment | Pretreatment with ticagrelor before angiography versus no pretreatment | Median of 24 hours | A composite of death due to vascular causes, non-fatal MI, non-fatal stroke, and BARC Type 3, 4, or 5 bleeding | No significant reduction Percentage absolute risk reduction: –0.46, 95% CI: –2.90 to 1.90 |
| ACCOAST (2013)18 | 4,033 | NSTE-ACS scheduled to undergo angiography within 2 to 48 hours | 30 mg prasugrel loading dose before angiography or placebo | Median of 4.3 hours | A composite of death from cardiovascular causes, MI, stroke, urgent revascularisation, or GPIIb/IIIa inhibitor rescue therapy up until day 7 | No significant difference HR 1.02, 95% CI: 0.84-1.25; p=0.81 |
| ACS: acute coronary syndrome; BARC: Bleeding Academic Research Consortium; CAD: coronary artery disease; CI: confidence interval; GP: glycoprotein; HR: hazard ratio; MACE: major adverse cardiovascular events; MI: myocardial infarction; NSTEMI: non-ST-segment elevation myocardial infarction; NSTE-ACS: non-ST-segment elevation acute coronary syndrome; PCI: percutaneous coronary intervention; RR: relative ratio; STEMI: ST-segment elevation myocardial infarction | ||||||
Treatment with oral antiplatelet agents in ST-segment elevation myocardial infarction
Patients with STEMI represent a group at the highest risk of early recurrent ischaemic events. Both the American and European revascularisation guidelines have a Class I recommendation for a loading dose of P2Y12 inhibitor in patients with STEMI; however, they do not specify a certain timing of administration47 (Table 1) as the data from randomised clinical trials remain unclear (Table 3).
Table 3. Summary of randomised clinical trials of pretreatment with oral P2Y12 inhibitors in patients with STEMI.
| Trial (year) | # of patients | Patient population | Intervention | Duration of pretreatment | Primary endpoint | Results |
|---|---|---|---|---|---|---|
| PCI-CLARITY (2005)27 | 1,863 | Receiving fibrinolytics for STEMI | 300 mg clopidogrel at time of fibrinolysis and given until angiography or placebo | Median of 3 days | Composite of cardiovascular death, recurrent MI, or stroke from PCI to 30 days | 3.6% pretreatment versus 6.2% placebo Adjusted OR 0.54, 95% CI: 0.35-0.85; p=0.008 |
| CIPAMI (2012)28 | 337 | STEMI scheduled for primary PCI | Prehospital 600 mg clopidogrel loading dose+standard therapy or standard therapy alone | 47 minutes | TIMI 2/3 patency of infarct-related artery in diagnostic angiography immediately prior to PCI | No difference 49.3% prehospital vs 45.1% standard; p=0.5 |
| Load&Go (2013)29 | 168 | STEMI referred for PCI | 600 mg or 900 mg clopidogrel loading dose at first medical contact or no pretreatment | 65 minutes | Rate of TIMI grade 3 flow | No difference 64.9% pretreatment versus 66.1% no pretreatment; p=0.88 |
| ATLANTIC (2014)36 | 1,862 | Ongoing STEMI of less than 6 hours duration | 180 mg ticagrelor loading dose prehospital or placebo in hospital | 48 minutes | Coprimary: (1) proportion of patients who did not have a 70% or greater resolution of ST-segment elevation before PCI and (2) proportion of patients who did not have TIMI flow grade 3 in the infarct-related artery | No difference (1) OR 0.93, 95% CI: 0.69-1.25; p=0.63 (2) OR 0.97, 95% CI: 0.75-1.25; p=0.82 |
| CI: confidence interval; MI: myocardial infarction; OR: odds ratio; PCI: percutaneous coronary intervention; STEMI: ST-segment elevation myocardial infarction; TIMI: Thrombolysis in Myocardial Infarction | ||||||
Prasugrel
There is no clear evidence of the value of pretreatment with prasugrel in the STEMI population. The TRial to assess Improvement in Therapeutic Outcomes by optimising platelet inhibitioN with prasugrel (TRITON)-TIMI 38 trial was a randomised controlled trial comparing prasugrel with clopidogrel. In the cohort of patients presenting with STEMI, prasugrel led to a reduction in the primary endpoint of cardiovascular death, non-fatal MI, or non-fatal stroke compared with clopidogrel, with no difference in TIMI major bleeding. While 27% of patients in this trial received the study drug before PCI, the trial was not designed to investigate the effects of timing of treatment before PCI38.
An observational study of 3,497 patients with STEMI who underwent PCI showed that prehospital loading with prasugrel or ticagrelor did not significantly improve coronary reperfusion, assessed via TIMI flow grade 3 in the infarct-related artery, when compared with prehospital loading with clopidogrel39. Despite the longer time interval between P2Y12 inhibitor treatment and angiography (65-75 minutes), which was longer than in the ATLANTIC study described above, there was no improvement in coronary reperfusion with ticagrelor or prasugrel pretreatment compared with clopidogrel.
Overall
While the evidence points towards a limited benefit of early P2Y12 inhibition in NSTE-ACS patients and associated harms, the data for pretreatment in STEMI patients do show a signal towards potential benefit. In a retrospective observational study of patients in the ARIAM-Andalucia Registry, in which 69% of patients received upstream pretreatment with clopidogrel, adjusted multivariable analyses demonstrated that pretreatment was associated with a significant reduction in MACE, but only in the STEMI group32. The benefit in STEMI patients may be driven by the higher thrombotic burden in this population, as well as the higher likelihood of stenting, and the fact that few go on to surgery or receive medical therapy alone. Ongoing trials of novel parenteral agents will help clarify the role of early platelet inhibition in patients with STEMI, as discussed below.
Clopidogrel
Evidence supporting pretreatment with clopidogrel for patients with STEMI originated from the PCI-Clopidogrel as Adjunctive Reperfusion Therapy (CLARITY) - Thrombolysis in Myocardial Infarction (TIMI) 28 trial, in which post-fibrinolysis patients underwent PCI an average of 3 days after fibrinolytic therapy; pretreatment with clopidogrel significantly reduced the odds of cardiovascular death, recurrent MI, or stroke within 30 days following PCI, by 46%27. However, two smaller trials did not show significant benefit with clopidogrel pretreatment when analysing surrogate endpoints such as reperfusion, angiographic indices, and patency of the culprit vessel2829. The Clopidogrel Administered Prehospital to Improve Primary PCI in Patients With Acute Myocardial Infarction (CIPAMI) Study randomised 337 patients to prehospital clopidogrel loading versus standard therapy and did not demonstrate any differences in pre-PCI patency of the infarct vessel; however, the time between clopidogrel loading and angiography was only 47 minutes, which is likely not enough time for the prodrug clopidogrel to be activated28.
Several registries and smaller studies also support the early use of clopidogrel in patients with STEMI3031. The Analysis of Delay in Acute Myocardial Infarction-Andalucia Registry showed reductions in the occurrence of death and thrombotic outcomes exclusively in the subgroup of ACS patients with STEMI, though there was an increase in minor bleeding32. Similarly, a registry of patients undergoing PCI for STEMI in Austria between 2005 and 2009 showed a reduction in in-hospital mortality with clopidogrel pretreatment33. In another analysis of 2,014 consecutive STEMI patients who were pretreated with clopidogrel, patients with longer pretreatment duration had reduced reinfarction/reischaemia rates at 30 days with no increase in major bleeding34. Larger-scale data from a meta-analysis of 37,000 patients also support the use of clopidogrel pretreatment in patients with STEMI; pretreatment was not associated with a lower risk of mortality but carried a lower risk of MACE. The higher-risk STEMI population had the most benefit with pretreatment, unlike patients undergoing elective PCI35.
Ticagrelor
Ticagrelor, compared with clopidogrel, has a more rapid antiplatelet effect. The Administration of Ticagrelor in the cath Lab or in the Ambulance for New ST elevation myocardial Infarction to open the Coronary artery (ATLANTIC) study randomised patients to prehospital or in-hospital treatment with ticagrelor. In this study, with a mean time to angiography of 48 minutes, there were no significant differences in the two coprimary endpoints (the proportion of patients who did not have 70% or greater resolution of ST-segment elevation before PCI and the proportion of patients who did not have TIMI flow grade 3 in the infarct-related artery) or in the rates of major bleeding; however, the rates of stent thrombosis at 30 days were lower in the prehospital treatment group36. The short interval to PCI may explain the absence of detectable benefit as it may not be entirely reflective of routine clinical practice. Furthermore, there was a significant interaction between morphine administration and one of the endpoints, which led the investigators to postulate that prehospital morphine may have diminished the potential benefits of upstream ticagrelor36. The ATLANTIC-H24 study, a post hoc exploratory analysis, demonstrated that the largest difference in platelet aggregation between prehospital or in-hospital treatment with ticagrelor occurred immediately after PCI37. The composite myocardial ischaemic endpoint of death, MI, urgent revascularisation, stent thrombosis, and downstream glycoprotein IIb/IIIa inhibitor use was lower at 24 hours in the prehospital treatment group37.
Treatment with oral antiplatelet agents in stable coronary artery disease
In patients with stable CAD, the American guidelines7 do not specifically comment on pretreatment, while the European guidelines have a Class IIb recommendation that pretreatment with clopidogrel may be considered if the probability of PCI is high4 (Table 1).
The CREDO trial, described above in the section on NSTE-ACS, also included stable CAD patients undergoing elective PCI and did not demonstrate benefit with pretreatment, though there was benefit seen in patients who received clopidogrel at least 6 hours before PCI9. Other prior studies specifically within the stable CAD population do not support pretreatment with clopidogrel, which may be related to the differences in platelet activity and risk of ischaemic events in this patient population as compared with ACS. The PRimary Angioplasty in patients transferred from General community hospitals to specialized percutaneous transluminal coronary angioplasty (PTCA) Units with or without Emergency thrombolysis-8 (PRAGUE-8) Trial of 1,028 patients with stable angina randomised to clopidogrel more than 6 hours before angiography or to clopidogrel given in the catheterisation lab showed no benefit on periprocedural infarction, but there was an increase in minor bleeding complications40. This was consistent with the ISAR-REACT trial which revealed no benefit with clopidogrel pretreatment in patients undergoing elective PCI41.
Alternative to oral antiplatelet pretreatment – utilising intravenous antiplatelet agents
Cangrelor
An alternative to pretreating with oral antiplatelets is utilising a potent, fast-acting, and rapidly reversible intravenous (IV) antiplatelet agent, such as cangrelor, during PCI. The use of cangrelor is supported by guidelines with a Class IIb recommendation for use in P2Y12 inhibitor-naïve patients undergoing PCI4567 (Table 1).
The Cangrelor versus standard therapy to acHieve optimal Management of Platelet InhibitiON (CHAMPION) PCI Trial showed that cangrelor was not superior to clopidogrel in patients undergoing PCI (85% ACS and 15% stable CAD) for the composite of death, MI, or ischaemia-driven revascularisation; there was also no significant difference in severe or major bleeding42. In addition, the CHAMPION PLATFORM Trial of cangrelor versus placebo in patients with NSTE-ACS or PCI for stable angina also indicated no difference in the primary composite outcome of death, MI, or ischaemia-driven revascularisation at 48 hours; however, there were reductions in the prespecified secondary endpoints of stent thrombosis and the rate of death from any cause at 48 hours43. Limitations of the first two CHAMPION trials included the lack of clear discrimination between MI prior to randomisation and periprocedural MI. The CHAMPION PHOENIX Trial randomised patients undergoing PCI (56% stable angina, 26% NSTE-ACS, and 18% STEMI) to cangrelor versus clopidogrel and did demonstrate a significant reduction in the primary composite endpoint of death from any cause, MI, ischaemia-driven revascularisation, or stent thrombosis at 48 hours with cangrelor, mainly driven by a reduction in periprocedural MI44. A post hoc analysis of this trial showed that periprocedural MACE after PCI is strongly associated with the number of high-risk lesion features and, although cangrelor reduced ischaemic events regardless of baseline lesion complexity, the absolute benefit-risk profile of cangrelor is likely greatest in patients undergoing PCI for complex coronary anatomy45. Finally, a pooled analysis of all three CHAMPION trials showed a 20% reduction in the primary endpoint (death, MI, ischaemia-driven revascularisation, stent thrombosis) with cangrelor at 48 hours, with no increase in severe or life-threatening bleeding46.
Intravenous cangrelor offers a compelling therapeutic option also for patients presenting with acute myocardial infarction and/or cardiogenic shock, a population in whom the effect of oral P2Y12 inhibitors may be unreliable because of impaired drug absorption, delayed gastric emptying, gut hypoperfusion, and altered metabolism4748. Unlike oral agents, cangrelor provides a potent P2Y12 inhibition effect with an onset within 2 minutes and a half-life of 3-6 minutes, making it particularly advantageous in patients who are intubated, sedated, or receiving vasopressors or mechanical circulatory support4950. Data from observational studies suggest that cangrelor use in cardiogenic shock is associated with improved coronary flow, reduced ischaemic outcomes, and lower mortality, although these benefits may be offset by a numerical increase in major bleeding48. Of note, cangrelor can provide an additive platelet inhibitory effect when used in MI patients on top of crushed ticagrelor and can be safely used without the risk of drug-drug interactions51. In contrast, clopidogrel and prasugrel are subject to drug-drug interactions and thus need to be administered at the end of the cangrelor infusion52. The 2021 joint position statement from the ESC Working Group on Thrombosis gives a consensus recommendation that parenteral antithrombotic therapy should be considered to cover the period before the onset of action of oral P2Y12 inhibitors in critically ill patients53. Furthermore, in the recent DAPT-SHOCK-AMI trial, 605 patients with acute MI and cardiogenic shock were randomised to receive IV cangrelor versus crushed ticagrelor (ClinicalTrials.gov: NCT03551964)54. The trial results presented at the ESC Congress 2025 (Madrid, Spain) demonstrated that the primary lab endpoint of platelet reactivity <50% at the end of primary PCI was achieved in 100% of patients in the cangrelor group versus 22.1% in the ticagrelor group. The rate of all-cause death, MI, or stroke at 30 days was similar between groups, with some secondary clinical endpoints favourably influenced55.
Finally, bridging with cangrelor may be considered in selected cases for maintaining platelet inhibition in patients with recent coronary stents who require temporary discontinuation of oral P2Y12 inhibitors prior to surgery, although cangrelor is not approved for this indication56. The use of cangrelor in this context is supported primarily by a single randomised controlled trial – the BRIDGE trial – in which 210 patients awaiting CABG were enrolled57. The study demonstrated that cangrelor infusion maintained platelet inhibition without increasing major bleeding prior to CABG; however, it was associated with a higher incidence of minor bleeding events and showed no reduction in ischaemic outcomes. While the trial supports the pharmacodynamic rationale for bridging, it was underpowered to detect meaningful differences in clinical events. Moreover, the evidence for cangrelor bridging in the setting of non-cardiac surgery is even more limited and largely based on observational data58. Further research is warranted to confirm the safety, efficacy, and cost-effectiveness of this strategy across diverse surgical settings.
Novel parenteral agents for high-risk patients
Significant advancements in reperfusion strategies, including shorter door-to-balloon times, have contributed to a decline in ischaemic complications, especially in high-risk ACS patients presenting with STEMI1. However, despite these improvements, timely care remains a challenge, with only 17% of patients who require interhospital transfer to a PCI-capable centre receiving reperfusion therapy within the recommended timeframe59. Importantly, risk-adjusted in-hospital mortality is significantly higher among patients who experience delays in reperfusion, compared with those treated within guideline-directed target times, underscoring the urgent need for optimised prehospital strategies59. As discussed above, randomised trials have failed to demonstrate a clear clinical benefit of pretreatment with oral P2Y12 inhibitors in STEMI patients to enhance early platelet inhibition1536. However, meta-analyses and observational studies suggest that early and prehospital administration of P2Y12 inhibitors might be associated with better reperfusion and reduction of ischaemic events compared with no pretreatment6061. Despite these findings, the pharmacokinetics of oral P2Y12 inhibitors limit their efficacy in the acute phase of STEMI. Even ticagrelor and prasugrel, which exhibit a rapid onset of action (~30 minutes) in stable coronary artery disease, require approximately 4-6 hours to achieve full platelet inhibition in STEMI patients due to delayed gastrointestinal absorption and increased platelet reactivity in the acute setting62. This delayed onset leaves a critical window of vulnerability where patients remain at high risk for thrombotic complications, particularly during emergency transport and pre-PCI management. In addition, a trial of crushed versus uncrushed prasugrel demonstrated better platelet reactivity but no difference in TIMI flow of the infarct-related artery in STEMI patients, suggesting that alternative strategies to achieve faster and more potent platelet inhibition are needed63.
Parenteral agents, including P2Y12 inhibitors and glycoprotein IIb/IIIa inhibitors, provide a more rapid and effective strategy for achieving early platelet inhibition, including in prehospital settings. Selatogrel, a novel subcutaneous P2Y12 inhibitor, has been recently tested for self-injection in patients at very high ischaemic risk, who could hypothetically benefit from early administration at the first symptoms of ischaemia64. A single-dose subcutaneous administration of selatogrel induced profound, rapid, and dose-dependent antiplatelet effects in patients presenting with acute MI. The proportion of responders at 30 minutes post-dose was 91%, achieving platelet inhibition levels comparable to those observed with ticagrelor or prasugrel only after several hours, underscoring the potential of selatogrel to bridge the time gap between symptom onset and PCI64. Moreover, selatogrel was well tolerated, with no significant increase in major bleeding complications. The ongoing SOS-AMI trial (Table 4), a global double-blind RCT enrolling 14,000 patients, is evaluating the efficacy of selatogrel in reducing early ischaemic events in patients with STEMI and NSTEMI (ClinicalTrials.gov: NCT04957719)65.
On the other hand, the role of GPIs during PCI has evolved primarily into a bailout strategy for thrombotic complications rather than routine use. However, the emergence of next-generation compounds may offer new potential applications. The On-TIME 2 trial, which investigated prehospital intravenous administration of tirofiban in STEMI patients, demonstrated improved ST-segment elevation resolution and clinical outcomes post-PCI66. In the ADMIRAL trial, STEMI patients received abciximab in the mobile intensive care unit before arrival at the hospital, in the emergency department or in the catheterisation lab, and they were found to have lower rates of death, reinfarction, or urgent revascularisation when compared with placebo67. Nevertheless, guidelines do not recommend routine pretreatment with GPIs in STEMI patients, likely due to concerns that the potential ischaemic benefits are offset by an increased bleeding risk5. Other limitations of first-generation GPIs include a relatively long half-life, the risk of thrombocytopaenia, and the requirement for intravenous administration, which may be logistically challenging. The need for simpler alternatives has led to the development of zalunfiban (RUC-4), a second-generation glycoprotein IIb/IIIa inhibitor specifically designed for subcutaneous administration, making it a promising candidate for prehospital use6869. Zalunfiban’s pharmacodynamic profile includes rapid platelet inhibition within 15 minutes and a short half-life of approximately 1 hour70. By displacing the Mg²âº ion at the metal ion-dependent adhesion site of the glycoprotein IIIa subunit, zalunfiban effectively prevents fibrinogen binding and platelet aggregation68. Unlike traditional intravenous GPIs such as eptifibatide and tirofiban, zalunfiban does not induce receptor conformational changes, thereby significantly reducing the risk of thrombocytopaenia6871. Preliminary analyses have shown that pretreatment with zalunfiban is associated with greater coronary and myocardial perfusion and a lower thrombus burden in STEMI patients undergoing primary PCI72. The CELEBRATE trial, a phase 3, randomised, placebo-controlled trial (Table 4) that enrolled 2,467 STEMI patients, assessed the impact of a single prehospital subcutaneous injection of zalunfiban on early reperfusion, infarct size, and clinical outcomes when administered at first medical contact; it showed an improvement in the primary efficacy endpoint with zalunfiban (adjusted odds ratio 0.79; 95% confidence interval: 0.65-0.98; p=0.028). Severe bleeding was similar between the two groups, with increased mild to moderate bleeding observed in the zalunfiban group7374.
Table 4. Summary of current trials of novel parenteral agents for high-risk patients.
| Selatogrel65 | Zalunfiban74 | |
|---|---|---|
| Pharmacology | Potent, highly selective, and reversible P2Y12 inhibitor | Small molecule inhibitor of the platelet glycoprotein IIb/IIIa receptor |
| Trial name (ClinicalTrials.gov) | SOS-AMI trial (NCT04957719) | CELEBRATE trial (NCT04825743) |
| Study design | Studying the efficacy of selatogrel when self-administered upon occurrence of symptoms suggestive of AMI in participants at risk of recurrent AMI. | Studying the efficacy of zalunfiban in STEMI patients when administered by paramedics during ambulance transport to hospital. |
| Major inclusion criteria: Symptomatic Type 1 AMI (STEMI or NSTEMI) within 4 weeks Diagnosis of multivessel CAD during qualifying AMI event and presence of at least 1 additional risk factor | Major inclusion criteria: STEMI patient, presenting with ischaemic chest pain and new ST-segment elevation, in whom the total duration of symptoms is a maximum of 4 hours | |
| Primary outcome | Clinical status as assessed by a 6-point ordinal scale: 1) Death (all-cause) within 7 days; (2) AMI with compromised haemodynamics within 2 days; (3) STEMI within 2 days; (4) high risk NSTEMI within 2 days; (5) NSTEMI with peak troponin levels greater than 10 times the upper limit of normal within 2 days; (6) none of the above | Clinical status assessed by a 7-point scale: 1) Death (all-cause) at 30 days; (2) stroke at 30 days; (3) recurrent MI (Type 1 to 4 MI) at 30 days; (4) acute stent thrombosis at 24 hours post-PCI/angiography; (5) new onset heart failure or rehospitalisation for heart failure at 30 days; (6) MI with high-sensitivity cardiac troponin levels greater than 10 times the upper limit of normal at 24 hours post-PCI/angiography; (7) none of the above |
| AMI: acute myocardial infarction; CAD: coronary artery disease; MI: myocardial infarction; NSTEMI: non-ST-segment elevation myocardial infarction; PCI: percutaneous coronary intervention; STEMI: ST-segment elevation myocardial infarction | ||
Conclusions
While pretreatment with oral antiplatelet agents offers the theoretical benefit of protection from ischaemic effects, the data to support its use have been conflicting. Over the years, the guidelines have also shifted in their recommendations; routine pretreatment is not recommended for NSTE-ACS, while the benefit of pretreatment in STEMI remains unclear (Central illustration). In the current era, there has been an evolution in the management and pharmacotherapy of patients with ACS such that more patients are undergoing early invasive management, which limits the full potential of oral antiplatelet agents. When taking into account the evidence and data from the clinical trials described in this review, it is important to consider the median time to coronary angiography, as variability in time to the catheterisation lab can influence whether there is benefit from pretreatment. Real-world practice may also involve treatment delays that are longer than those reported in randomised controlled trials, and pretreatment may have a role if the time to invasive evaluation is prolonged. Newer antiplatelet drugs such as cangrelor also offer ways to achieve a fast antiplatelet effect with ability to optimise periprocedural thrombotic risk such that pretreatment with oral agents may no longer be necessary. Ongoing trials of novel parenteral agents will provide more information on the effectiveness and safety of therapeutics that could be administered early for high-risk patients.
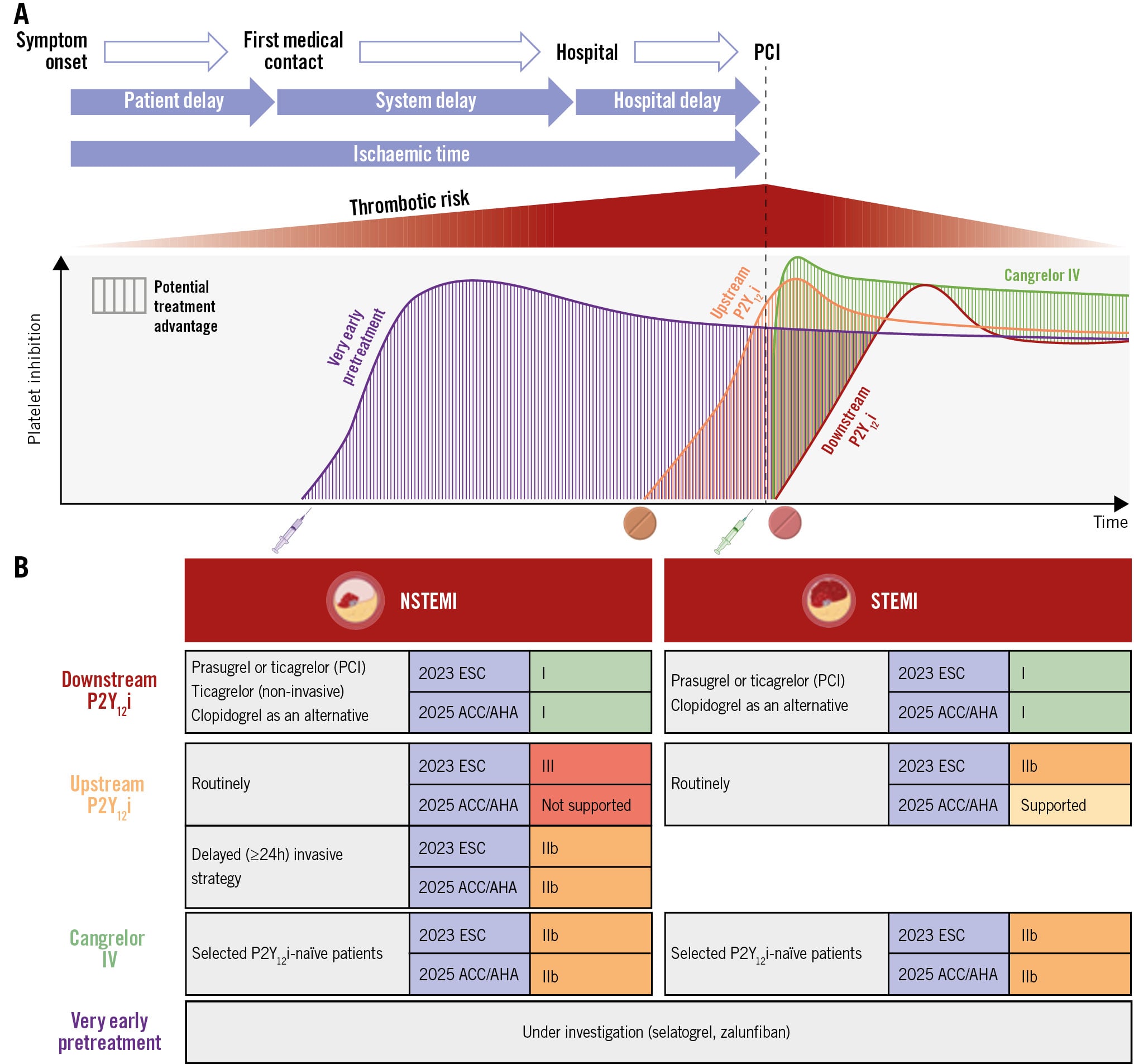
Central illustration. Overview of time-related treatment effects of downstream, upstream, and parenteral antiplatelet agents in ACS within the framework of current guidelines. In patients with ACS, multiple factors, including clinical presentation (e.g., STEMI vs NSTEMI) and delays associated with first medical contact and healthcare system logistics, influence the timing of PCI, potentially impacting the thrombotic risk (red gradient). A) An illustration of the dynamic process of platelet inhibition across different antiplatelet treatment strategies: very early pretreatment with investigational agents (selatogrel and zalunfiban; purple); upstream P2Y12 inhibitors administered before angiography (orange); downstream P2Y12 inhibition initiated at PCI (red); periprocedural intravenous (IV) cangrelor (green). B) A summary of current recommendations for the timing of P2Y12 inhibitor administration from the 2023 ESC Guidelines and 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the management of acute coronary syndromes. ACC: American College of Cardiology; ACS: acute coronary syndrome; ACEP: American College of Emergency Physicians; AHA: American Heart Association; ESC: European Society of Cardiology; NAEMSP: National Association of EMS Physicians; NSTEMI: non-ST-segment elevation myocardial infarction; P2Y12i: P2Y12 inhibitor; PCI: percutaneous coronary intervention; SCAI: Society for Cardiovascular Angiography and Interventions; STEMI: ST-segment elevation myocardial infarction
Conflict of interest statement
D.L. Bhatt discloses the following relationships - advisory board: AngioWave, Antlia Bioscience, Bayer, Boehringer Ingelheim, CellProthera, Cereno Scientific, E-Star Biotech, High Enroll, Janssen, Level Ex, McKinsey, Medscape Cardiology, Merck, NirvaMed, Novo Nordisk, Repair Biotechnologies, Stasys, SandboxAQ (stock options), Tourmaline Bio, and Viatris; board of directors: American Heart Association New York City, AngioWave (stock options), Bristol-Myers Squibb (stock), DRS.LINQ (stock options), and High Enroll (stock); consultant: Alnylam, Altimmune, Broadview Ventures, Corcept Therapeutics, Corsera, GlaxoSmithKline, Hims, SERB, SFJ, Summa Therapeutics, and Worldwide Clinical Trials; data monitoring committees: Acesion Pharma, Assistance Publique-Hôpitaux de Paris, Baim Institute for Clinical Research, Boston Scientific (Chair, PEITHO trial), Cleveland Clinic, Contego Medical (Chair, PERFORMANCE 2), Duke Clinical Research Institute, Mayo Clinic, Mount Sinai School of Medicine (for the ABILITY-DM trial, funded by Concept Medical; for ALLAY-HF, funded by Alleviant Medical), Novartis, Population Health Research Institute, and Rutgers University (for the NIH-funded MINT Trial); honoraria: American College of Cardiology (Senior Associate Editor, Clinical Trials and News, ACC.org; Chair, ACC Accreditation Oversight Committee), Arnold and Porter law firm (work related to Sanofi/Bristol-Myers Squibb clopidogrel litigation), Baim Institute for Clinical Research (AEGIS-II executive committee funded by CSL Behring), Belvoir Publications (Editor-in-Chief, Harvard Heart Letter), Canadian Medical and Surgical Knowledge Translation Research Group (clinical trial steering committees), CSL Behring (AHA lecture), Duke Clinical Research Institute, Engage Health Media, HMP Global (Editor-in-Chief, Journal of Invasive Cardiology), Medtelligence/ReachMD (CME steering committees), MJH Life Sciences, Oakstone CME (Course Director, Comprehensive Review of Interventional Cardiology), Philips (Becker’s Webinar on AI), Population Health Research Institute, WebMD (CME steering committees), and Wiley (steering committee); other: Clinical Cardiology (Deputy Editor, unpaid), Progress in Cardiovascular Diseases (Deputy Editor, unpaid), and Added Health (Editorial Board; stock options); patent: Sotagliflozin (named on a patent for sotagliflozin assigned to Brigham and Women’s Hospital who assigned to Lexicon; neither he nor Brigham and Women’s Hospital receive any income from this patent); research funding: Abbott, Acesion Pharma, Afimmune, Alnylam, Amarin, Amgen, AstraZeneca, Atricure, Bayer, Boehringer Ingelheim, Boston Scientific, CellProthera, Cereno Scientific, Chiesi, Cleerly, CSL Behring, Faraday Pharmaceuticals, Fractyl, Idorsia, Janssen, Javelin, Lexicon, Lilly, Medtronic, Merck, MiRUS, Moderna, Novartis, Novo Nordisk, Pfizer, PhaseBio, Regeneron, Reid Hoffman Foundation, Roche, Sanofi, Stasys, and 89Bio; royalties: Elsevier (Editor, Braunwald’s Heart Disease); site co-investigator: Cleerly. R. Mehran reports institutional research payments from Abbott, Alleviant Medical, Beth Israel Deaconess Medical Center, Concept Medical, CPC Clinical Research, Cordis, Elixir Medical, Faraday Pharmaceuticals, Idorsia Pharmaceuticals, Janssen, MedAlliance, Mediasphere Medical, Medtronic, Novartis, Protembis GmbH, RM Global Bioaccess Fund Management, and Sanofi US Services, Inc.; personal fees from Elixir Medical, IQVIA, Medtronic, Medscape/WebMD Global, and Novo Nordisk; equity <1% in Elixir Medical, Stel, and ControlRad (spouse); no fees from SCAI (Women in Innovations Committee Member), Faculty Cardiovascular Research Foundation (CRF), and Women as One (Founding Director); honorarium from AMA - JAMA Cardiology (Associate Editor) and ACC (BOT Member, SC Member CTR Programme). The other authors have no conflicts of interest to declare.

