Abstract
Aims: The retrograde approach via septal or epicardial collaterals holds promise in improving the success rate of chronic total occlusion (CTO) recanalisation but is still viewed as an esoteric practice reserved to a selected group of operators using materials not available in Western countries. We sought to introduce the novel technique of retrograde CTO recanalisation to our institution using materials and resources available outside Japan.
Methods and results: Between April 2005 and 2007, all patients with a failed antegrade attempt at CTO recanalisation were considered and selected based on their appropriateness to have a retrograde procedure. Seventeen patients were included. Average age was 65.3 years. The mean occlusion duration was 33.3 months. Successful CTO recanalisation was achieved in 15 of 17 (88.2%) patients. A retrograde wire was successfully advanced into the distal occluded vessel and facilitated successful CTO recanalisation in 13 (76.5%) patients. Final retrograde wire crossing was achieved in seven cases, while in the remaining cases, the CART technique (n=4) and ‘knuckle’ technique (n=2) were successful. In two cases (11.8%), the retrograde wire did not reach the distal occlusion because of septal tortuosity but a repeat antegrade attempt, aided by contralateral visualisation, was successful. In two (11.8%) patients, the procedure was unsuccessful. Cardiac magnetic resonance imaging performed in five of the successfully treated retrograde cases (45.5%) showed variable degrees of fibrosis in the territory of the occluded artery with no significant change after the procedure with the exception of one case of contained contrast extravasation within the septum.
Conclusions: The novel method of retrograde recanalisation of CTOs was performed with a high procedural success in this series and was applicable in 37.1% of previously failed antegrade procedures. This technique can also be successfully employed despite newly created dissections immediately following a failed antegrade attempt.
Introduction
The retrograde approach to the recanalisation of chronic total occlusions (CTOs) is an established method in peripheral revascularisation (i.e., popliteal puncture to open an occluded superficial femoral artery)1 and has been adopted through surgical grafts to open occluded proximal main epicardial vessels. It is the merit of Japanese interventional cardiologists, pioneered by Osamu Katoh from the Toyohashi Heart Centre, to introduce an alternative percutaneous strategy for patients not suitable, or after failure of a recanalisation procedure using the conventional antegrade approach. The controlled antegrade and retrograde sub-intimal tracking (CART) technique has been in use for over a year and a select group of interventionalists have gained significant experience and proficiency. However, with the exception of a few live demonstrations from Japanese physicians in Western laboratories, CART is viewed outside Japan as an esoteric method still under development with no current practical applications as a main stream procedure. This perception can be explained by the steep learning curve associated with its ground-breaking concept, the requirement for dedicated material not available outside Japan and the fear of untoward complications when dealing with fragile tiny collateral vessels. We set out to introduce this technique to our institution by inviting its main champion to train and guide us through the intricacies of this revolutionary method. In this manuscript we report both the results of three sessions of elective cases under proctorship, and other independently performed, unplanned procedures with the aim of showing that this approach can be safely and successfully applied using approved material also in Western countries.
Methods
Study population
All patients with one or more unsuccessful attempts to recanalise a CTO using an antegrade approach in the past two years were assessed regarding their suitability for a retrograde approach. Inclusion criteria were: 1. Prior failed attempt(s) using a conventional antegrade approach; 2. Ongoing symptoms or evidence of viable myocardium/inducible ischaemia in the territory of distribution of the occluded vessel assessed with cardiac magnetic resonance (CMR) or, if contraindicated because of claustrophobia or pacemaker, with scintigraphy; 3. Presence of a suitable collateral channel from a contra-lateral patent coronary artery.
CMR procedure
Patients who underwent a pre-intervention CMR scan (Siemens 1.5T) had baseline morphology evaluated with subsequent viability assessed with injection of gadolinium-DTPA. Late myocardial enhancement (LGE) was detected using an inversion recovery preparation with segmented gradient echo readout (GRE) and evaluated by applying the recommended 17-segment myocardial model for tomographic imaging of the heart2. A second CMR scan was performed following the procedure with the same protocol and settings used as in the pre-intervention examination. Two blinded observers measured the amount of late gadolinium enhancement, expressed as a percentage of the total myocardial mass and assessed its location. Pre- and post- PCI global left ventricular function was assessed.
Clinical definitions and intervention medications
Coronary CTOs were defined as thrombolysis in myocardial infarction (TIMI) grade 0 flow with a duration >3 months, estimated from clinical events such as acute myocardial infarction in the same target vessel territory, sudden onset or worsening of the symptoms, or proven by previous angiography, or undetermined. Angiographic success was defined as restoration of TIMI flow grade 2-3 with stent implantation in the occluded segments and residual stenosis <20% by visual estimate. Procedural success was defined as angiographic success in the absence of death, Q-wave or non-Q wave myocardial infarction (MI) (>3 fold increase of creatine-kinase MB), stroke and pericardial effusion requiring pericardiocentesis. All patients were pretreated with aspirin and clopidogrel. Heparin was used to maintain an activated clotting time (ACT) ≥250 seconds. Upstream use of glycoprotein IIb/IIIa inhibitor therapy or bivalirudin was avoided for the potential increased risk of bleeding in case of coronary perforation.
Technique
The retrograde approach, including retrograde wire crossing or CART technique require the insertion of a guidewire and balloons through collateral branches supplying chronically occluded vessels3,4. A 6, 7 or 8 Fr guiding catheter is positioned at the ostium of both the occluded and the donor vessel. Where possible, 85 or 90 cm guiding catheters are preferred to standard 100 cm guides to cannulate the collateral donor artery. A soft polymer-coated wire (Fielder Asahi Intecc, Whisper Guidant, Choice-PT or PT2 BSC) is used to engage collateral which can be followed angiographically from the donor to the occluded vessel. In case multiple collaterals are present, septal collaterals are preferred to epicardial collaterals because of the less serpiginous path and smaller risk of perforation into the pericardium. Collaterals with straighter course and no acute angulations with the donor or recipient artery are preferentially selected. The wire manipulation is directed by the acquisition of a sub-selective injection via an over the wire catheter (in this series Transit, Cordis, Johnson & Johnson, 2.5 Fr distal outer diameter, 0.021” inner lumen) used as a road map (Figure 1C).
After wire passage, a dilatation with a 1.25 or 1.50 mm balloon at 2-3 atm of the collateral channel is performed and an OTW balloon with a diameter matching the size of the distal occluded vessel is advanced near the distal end of the occlusion and inflated at 10-12 atm to provide support to the attempts of retrograde wire probing. With the balloon inflated to this pressure, we did not encounter difficulties with wire manipulation but this could potentially occur due to compression of the wire lumen by the inflated balloon. If this occurs however, this may be overcome by inflating to a lower pressure. The soft hydrophilic wire is replaced with a 3.0 Miracle wire (Asahi Intecc, Japan), used to cross the occlusion from the distal true lumen. The principle is that the occlusion is often softer at the distal end, allowing the wire to be advanced into the proximal true lumen and then to proceed into the guiding catheter positioned at the ostium of the occluded vessel or into the aorta. At this point the retrograde balloon is advanced and inflated in the occlusion, re-establishing patency and allowing easy crossing of a soft wire antegradely. If retrograde crossing is unsuccessful, the CART technique is applied using the retrograde wire which is still advanced to create a sub-intimal dissection, enlarged through the inflation of the balloon into the sub-intima. An antegrade wire is then advanced and steered sub-intimally at the level of the occlusion to connect with the retrograde wire. Wire advancement is followed in multiple angiographic views to ensure alignment. Following this, the antegrade wire can be advanced into the distal true lumen. Should the retrograde balloon not be able to be inflated in the false lumen, the ‘knuckle’ technique can be applied using a polymer coated wire with its tip knuckled in the false lumen retrogradely followed by advancement of an antegrade wire into the false lumen to make a connection. If successful, the procedure ends with balloon dilatation performed at the occlusion site and drug-eluting stents implanted to cover the entire occluded segment. This is usually performed after having swapped the antegrade wire to a regular workhorse wire (Balance Middleweight Universal Guidant or Prowater Asahi Intecc in this series) which minimises the risk of distal vessel perforation.
Statistical analysis
Descriptive analyses were used. Results are quoted as percentages for categorical data or as mean ± standard deviation for continuous variables.
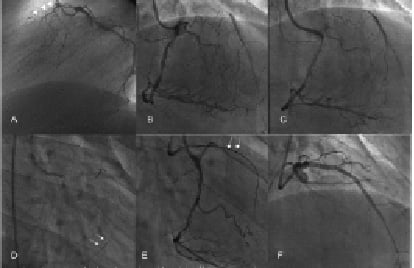
Figure 2. An example of the retrograde approach in a 51 year old man with an occluded left anterior descending artery (occlusion duration 4 years) and confirmed inducible ischaemia in the anterior wall and apex. A previous antegrade attempt had been unsuccessful.
A: Lateral view demonstrating the occluded left anterior descending artery (LAD, arrowheads); B: Right caudal view from the patent right coronary artery demonstrated collateral filling of the LAD. The complex collateral network is apparent; C: Fielder guidewire (Asahi-Intecc, Japan) through a Transit catheter (Cordis, Johnson & Johnson, USA) is used to negotiate the collateral with the straightest path to the LAD. This is best determined by selective contrast injection through the micro-catheter; D: The retrograde wire is seen at the level of the LAD and a 1.25 mm balloon is seen dilated gently to 2 atm in the collateral channel (arrows); E: The retrograde wire is now changed to an intermediate type (Miracle 3, Asahi-Intecc, Japan) and is used to initially probe the distal end of the occlusion. The wire was successfully negotiated through the occlusion (arrows) and is now seen within the aorta; F: Final outcome in the cranial view following pre-dilatation, stent implantation (Cypher Select, Cordis, Johnson & Johnson, USA) and high pressure post-dilatation.
Results
Baseline clinical characteristics
Between April 2005 and 2007, 137 patients underwent a percutaneous attempt to recanalise a CTO at our institution. Of these, 35 (25.4%) were unsuccessful using one or more conventional antegrade procedure(s). Following review of the angiogram to detect feasibility of a retrograde approach, 13 potential patients were identified with technically suitable coronary anatomy (37.1%). Twelve of those patients with persistent symptoms and/or demonstrable ischaemia agreed to a new retrograde approach and signed the dedicated informed consent approved by our local ethics committee. In addition to these 12 patients with prior failed attempt(s), five patients were treated immediately following a failed antegrade attempt.
Table 1 lists the clinical characteristics of the patients.

Average age of patients was 65.3 years (94.1% males) with 6 of 17 (35.3%) diabetics. The mean duration of CTO occlusion was 33.3 months with a range of 14 to 82 months. In five cases (29.4%), the age of the occlusion was indeterminate. Eight patients (47%) had a previous myocardial infarction. In all these patients, residual ischaemia was confirmed with imaging tests (nuclear perfusion or stress echocardiography).
CMR analysis
CMR scanning was performed in five patients with a successful retrograde procedure (45.5%) with variable degree of fibrosis present only in the territory of the occluded vessel. No significant change in the fibrotic area, both in absolute terms and % of the total myocardial mass was observed (Table 2).
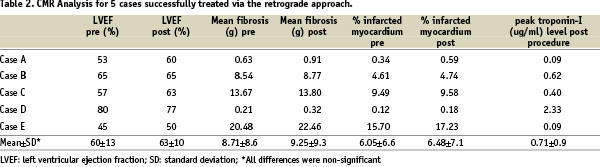
Furthermore, there were no new areas of fibrosis appearing post procedure with the exception of one case. In that patient, a new focal small area of sub-endocardial gadolinium uptake surrounded by oedema was seen within the septum two days after PCI which corresponded to a localised area of contrast staining angiographically after balloon dilatation of an angulated junction between septal collateral and posterior descending artery. A follow-up CMR scan two months following the PCI showed recovery with complete disappearance of the peri-lesional oedema initially seen5.
Procedural characteristics and outcomes
Table 3 lists the procedural characteristics of the treated cases.
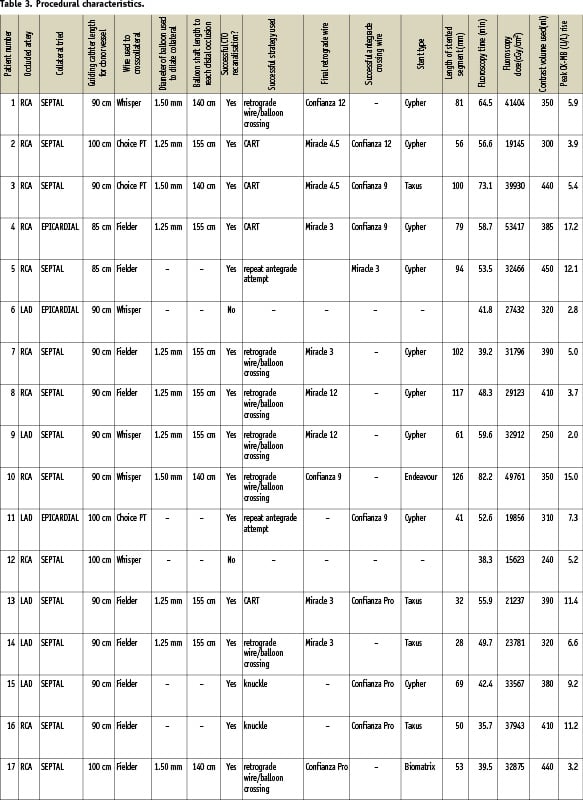
In 11 out of 17 cases (64.7%) the right coronary artery (RCA) was the target occluded vessel. A septal channel from the LAD was used in 10 cases with the last case approached from an epicardial collateral (atrial branch) of the left circumflex artery after an unsuccessful approach through a septal. The remaining six cases (35.3%) involved an occluded LAD with the retrograde wire past in four cases from the RCA into a septal branch and in the other two from an epicardial collateral surrounding the apex, arising from the distal posterior descending artery.
Successful CTO recanalisation was achieved in 15 of 17 (88.2%) patients. A retrograde wire was successfully advanced into the distal occluded vessel and facilitated CTO recanalisation in 13 (76.5%) of patients. Final retrograde CTO crossing was achieved in seven cases, while in the remaining cases, the CART technique (n=4) and ‘knuckle’ technique (n=2) were successful (Figure 1).
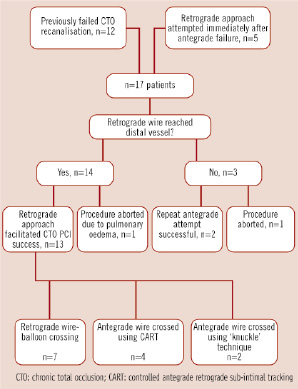
Figure 1. Flow-chart to indicate the outcome of PCI in the 17 patients studied.
In two cases (11.8%), the retrograde wire did not reach the distal occlusion because of septal tortuosity but a repeat antegrade attempt was successful, aided by using the parallel wire technique and optimal visualisation of the distal vessel via sub-selective collateral injection. In two cases (11.8%) the procedure was unsuccessful: in one case, the calcific occlusion was impenetrable, both retrogradely and antegradely and one further patient with underlying ischaemic cardiomyopathy (LVEF 30%) developed acute pulmonary oedema requiring cessation of the procedure. In this case, the retrograde wire and balloon were already advanced to the distal end of the occluded proximal LAD without complication via an epicardial RCA collateral when the procedure ended. There were no cases of in-hospital death, myocardial infarction (> 3 times CK-MB limit) or pericardial effusion.
Sirolimus-eluting (Cypher-Select, Cypher-Select Plus, Cordis J&J), paclitaxel-eluting (Taxus Liberte, BSC), zotarolimus-eluting (Endeavour, Medtronic) or biolimus-eluting (Biomatrix, Biosensors) stents were implanted in the occluded segment.
Discussion
Great caution must be used when drawing on the clinical implications of this preliminary experience in selected patients. Still, it is obvious that the retrograde approach is not a curiosity or a gimmick to show operative skills in live demonstrations. It can benefit patients with clinical indications to recanalise total occlusions and previous failure of a conventional percutaneous approach and represents a new therapeutic alternative to surgery. Although the technique is conceptually simple, its intricate and demanding requirements discourage its application without adequate proctorship. The steep learning curve arises not only from the unaccustomed route used to approach the occlusion but also from the specific procedural requirements of material and techniques. Sadly, most interventionalists in the UK and Europe are still unfamiliar with double coronary injection, over the wire balloons and stiff dedicated CTO wires which are standard of care in the most advanced centres in Southeast Asia and the USA. As the technique has been born in Japan, many of the procedural components adopted for the typical Japanese patient’s body habitus cannot be automatically transferred to the average Caucasian patient. A short 85 cm guide is normally used in Japan for the donor vessel but we have found this length insufficient for most of our patients forcing us to start with a slightly longer guide (90 cm) as first choice (Vista Brite-Tip, Cordis, Johnson & Johnson, Miami, Florida, USA). The ultimate solution to the challenge represented by the long and tortuous path the collateral takes to the occlusion will be the development of very long (>150 cm), flexible and kink resistant OTW balloons. An ingenious OTW super-flexible long catheter which can be advanced through particularly tortuous paths or across resistant CTOs (Tornus Asahi Intecc, Japan) has just received FDA approval and is expected to receive CE Mark by early 2008. We also changed our routine practice of using 6 Fr guides because 7 Fr guides offer greater support when tackling the occlusion or the tortuous track through collaterals and permit the use of the parallel wire technique, both with OTW support.
In identifying an appropriate collateral channel to the occluded vessel, we have found that super-selective contrast injection through a large lumen micro-catheter is imperative. Typical OTW balloon catheters have a too small inner lumen to properly delineate the collateral and have a stiff tip which does not track over the distal floppy part of the wire the often acute bend between donor vessel and collateral branch. A 0.021” inner lumen catheter with a single distal marker allows easy injection of undiluted contrast at the ostium of the collateral. We also learned that full angiographic continuity of the collateral up to the occluded vessel is a desirable feature but is not indispensable. Short segments of discontinuity of the septal from donor to recipient vessel simply indicate a collateral diameter below angiographic visibility (<100-200 µm) but which can still be crossed with a 0.014” (360 µm) lubricious wire. Although there was one case in whom super-sensitive imaging techniques allowed visualisation of a contained perforation embedded in the myocardium, the septum appears to offer a remarkable resistance to wall damage and development of bleeding complications.
The importance of the careful handling of the final guidewire to be used for antegrade/retrograde crossing of CTO cannot be sufficiently stressed. Japanese interventionalists spearheaded the development of a number of dedicated wires with optimal steerability and torqueability improving the chances of successful CTO crossing. No operator should apply a retrograde approach before having proved a high success rate in crossing CTOs antegradely using these devices. Interestingly, when a sufficiently large balloon (2.0 or 2.5 mm) is inflated in the vessel immediately distal to the occlusion to provide support, an intermediate Miracle 3 wire (Asahi Intecc, Japan) is often sufficient to penetrate the occlusion. Following the advice of the inventor of this approach, if an intermediate wire is unsuccessful in reaching the proximal true lumen, before trying stiffer wires (e.g. Miracle-12 or Confianza Pro, Asahi Intecc) retrogradely, the operator can attempt to proceed by advancing the balloon retrogradely to create a dissection and then advanced stiff wires antegradely.
The easiest part of the procedure is the final stent implantation and DES offer high chances of long-term patency, with the potential, if the indications for revascularisation were appropriate, of long lasting ischaemia relief6,7, improvement in regional wall motion8,9 and ultimately reduction of ‘hard’ clinical events (myocardial infarction, death)10,11. The retrograde approach has the potential to shorten the length of the stented segment because the sub-intimal track is limited to the occluded segment and is unlikely to extend to a long segment distal to the occlusion as in the antegrade parallel wire technique12. This small series has only partially confirmed this advantage because in most cases the disease distal to the occluded segment was so severe or, distal dissections were created by the balloon inflated to gain support, to require long stents anyway. The small percentage (37.1%) of patients selected for this approach from unsuccessful, previously attempted CTOs mainly reflects our fear to approach cases with too tiny or tortuous collaterals, especially if epicardial. The availability of dedicated material and improvement in operator’s skills may lower the threshold to apply this method. Still, the widespread use of this approach is made unlikely by the already good success rate of the antegrade approach. We have witnessed Japanese operators directly going to a retrograde approach to CTO in anatomically suitable cases, especially if there was no proximal occlusion stump. Our policy at this stage of experience is still to make prolonged attempts at crossing antegradely. We were favourably surprised by the three cases in which we could successfully switch to a retrograde approach immediately after failure of an antegrade approach, despite a long dissection distal to the occlusion. The feasibility to use a retrograde approach immediately after a failed antegrade attempt, without waiting weeks/months to allow healing of the dissection, is another important advantage of the retrograde strategy.
Procedure duration and fluoroscopy time are several times higher than in the majority of procedures. This increased radiation burden, at an average of 66.3 years represents a negligible additional risk of late events such as neoplasias, far exceeded by the potential benefit of the procedure. For operators often doing CTOs, a careful application of all the safety measures to reduce exposure is imperative. Proper scheduling, allowing sufficient laboratory time, ideally with two senior operators available, is also key to encourage a wider application of this technique. Besides availability and refinement of dedicated materials, a key element to apply the technique in the UK and Europe in a safe and highly successful fashion is to abandon the policy of “everybody doing everything” in the catheterisation laboratory and switch to the identification of CTO centres where the concentration of cases allows selected operators to acquire sufficient proficiency following sound proctorship schemes.
Contrast use, despite bilateral injection, can be minimised by experienced operators when a wire proximal / distal to the occlusion serves as a marker. None of these patients had a post-procedure creatinine rise, but in patients with renal impairment, generous peri-procedural hydration, acetyl-cysteine administration, and use of iso-osmolar contrast are recommended. The only complete failure in CTO crossing was observed in a patient with very low ejection fraction in whom stenting of a long sub-occlusive lesion was performed before the CTO was approached. Although the development of pulmonary oedema interrupted the procedure while wire and balloon were passed retrogradely, we interpreted this failure as a too ambitious plan in a frail patient hypersensitive to contrast and prolonged immobilisation lying flat rather than a consequence of the flow impairment / ischaemia secondary to collateral engagement.
Conclusions
The retrograde technique is safely applicable in more than a third of CTOs failed antegradely, possibly in the same session of the failed antegrade approach despite long and newly created dissections. With appropriate coaching, and if operators have sufficient experience of modern CTO material/technique, this preliminary consecutive series suggests that the retrograde approach yields a high success rate.

